Organic electroluminescence material and preparation method and application
An electroluminescent material and luminescent technology, applied in the direction of luminescent materials, organic chemistry, chemical instruments and methods, etc., can solve problems such as poor blue light color purity, achieve good blue light luminescence performance and stability, and reduce self-quenching Phenomenon, the effect of high blue light emission wavelength
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
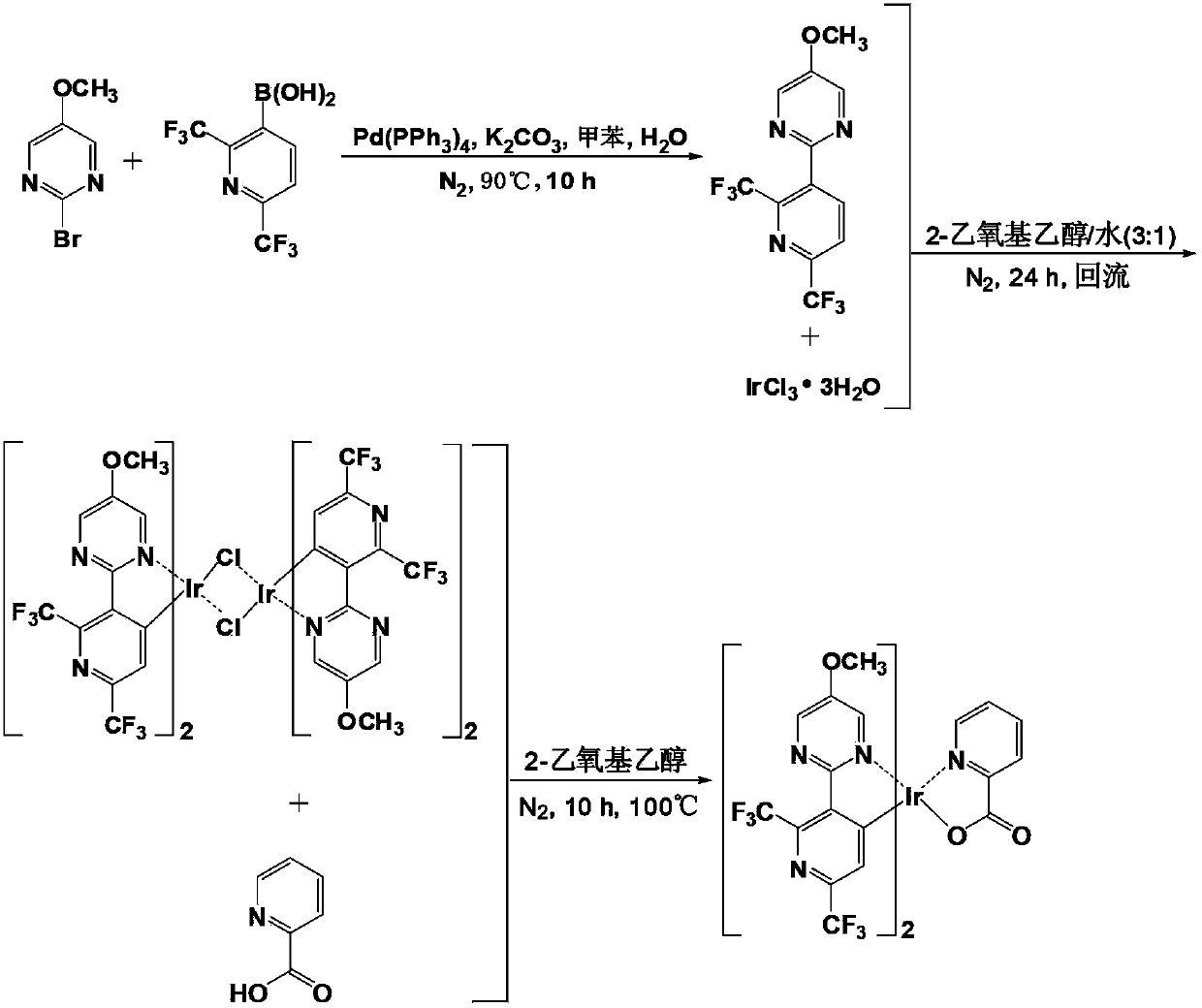
[0065] This example provides a bis(2-(2',6'-bis(trifluoromethyl)pyridin-3'-yl)-5-methoxypyrimidine-N,C 4' ) (2-pyridinecarbonyl) iridium complex, its chemical structural formula is as shown in P1:
[0066]
[0067] The preparation steps of the above P1 are as follows:
[0068] S10. Provide compound A1 (2-bromo-5-methoxypyrimidine) and compound B (2,6-bis(trifluoromethyl)pyridine-3-boronic acid) represented by the following structural formula:
[0069]
[0070] S20. Synthesis of compound C1 (2-(2',6'-bis(trifluoromethyl)pyridin-3'-yl)-5-methoxypyrimidine)
[0071] Under nitrogen protection, compound A1 (1.89g, 10mmol), compound B (3.11g, 12mmol) and Pd(PPh 3 ) 4 (0.58mg, 0.5mmol) was dissolved in 35mL of toluene, and then 15mL of an aqueous solution containing potassium carbonate (2.76g, 20mmol) was added, and the reaction was stirred at 90°C for 10 hours; Extract with methane, separate liquid, then wash with water until neutral, then dry with anhydrous magnesium sulf...
Embodiment 2
[0097] This example provides a bis(2-(2',6'-bis(trifluoromethyl)pyridin-3'-yl)-4-hexyloxypyrimidine-N,C 4' ) (2-pyridinecarbonyl) iridium complex, its chemical structural formula is as shown in P2:
[0098]
[0099] The preparation steps of above-mentioned P2 are as follows:
[0100] S10, providing compound A2 (2-bromo-4-hexyloxypyrimidine) and compound B (2,6-bis(trifluoromethyl)pyridine-3-boronic acid) represented by the following structural formula:
[0101]
[0102] S20, Synthesis of Compound C2 (2-(2',6'-bis(trifluoromethyl)pyridin-3'-yl)-4-hexyloxypyrimidine)
[0103] Under nitrogen protection, compound A2 (1.30g, 5mmol), compound B (1.29g, 5mmol) and Pd(PPh 3 ) 2 Cl 2 (0.14mg, 0.2mmol) was dissolved in 35mL of DMF, and then 15mL of an aqueous solution containing potassium carbonate (2.07g, 15mmol) was added, and stirred at 80°C for 12 hours; extraction, liquid separation, and then washed to neutrality, then dried with anhydrous magnesium sulfate and filtered, a...
Embodiment 3
[0126] This example provides a bis(2-(2',6'-bis(trifluoromethyl)pyridin-3'-yl)-5-eicosyloxypyrimidine-N,C 4' ) (2-pyridinecarbonyl) iridium complex, its chemical structural formula is as shown in P3:
[0127]
[0128] The preparation steps of above-mentioned P3 are as follows:
[0129] S10, providing compound A3 (2-bromo-5-eicosyloxypyrimidine) and compound B (2,6-bis(trifluoromethyl)pyridine-3-boronic acid) represented by the following structural formula:
[0130]
[0131] S20. Synthesis of compound C3 (2-(2',6'-bis(trifluoromethyl)pyridin-3'-yl)-5-eicosyloxypyrimidine)
[0132] Under nitrogen protection, compound A3 (0.91g, 2mmol), compound B (0.78g, 3mmol) and tetrakis(triphenylphosphine) palladium (0.07mg, 0.06mmol) were dissolved in 20mL of toluene, and then 10mL containing An aqueous solution of sodium carbonate (0.21g, 2mmol) was stirred at 90°C for 15 hours; after the reaction solution was cooled to room temperature, it was extracted with dichloromethane, separ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Sheet resistance | aaaaa | aaaaa |
| Maximum lumen efficiency | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More