Fluorogenic quantitative detection test card for human C-reactive protein
A fluorescent quantitative detection and reaction protein technology, applied in the biological field, can solve problems such as insufficient risk prediction, and achieve the effect of simple operation and convenient mass production.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0042] Embodiment 1. Preparation of anti-human C-reactive protein hybridoma cell line
[0043] 1. Animal immunization
[0044] BALB / c female mice (purchased from Changzhou Cavens Experimental Animal Co., Ltd.) were immunized with C-reactive protein extracted from human plasma (purchased from HyTest Company) according to the general immunization procedure. For specific immunization conditions, please refer to the "Experimental Guidelines for Antibody Preparation and Use". The serum titer of immunized mice was tracked by indirect ELISA method, and the immunized mouse with the highest serum titer was selected for fusion experiment of mouse splenocytes and mouse myeloma cells.
[0045] 2. Cell Fusion
[0046] (1). Preparation of spleen cells
[0047] Take the immunized mice, remove their eyeballs, take blood, put them to death by breaking the cervical spine, soak them in 75% (v / v) alcohol for 10 minutes, take out their spleens in a sterile operating table, place them in a cell ...
Embodiment 2
[0057] Example 2. Determination of the variable region sequence of the hybridoma cell line antibody
[0058] The sequences of the antibody variable regions of the above-mentioned hybridoma cell lines M24 and M03 were determined.
[0059] a. Extraction of RNA: Extract the total RNA of the above-mentioned hybridoma cell lines M24 and M03 with reference to the instructions of the Total Cell RNA Extraction Kit (purchased from Roche Company) and perform reverse transcription immediately;
[0060] b. Reverse transcription of RNA into DNA: Refer to Thermo Scientific Reverted First strand cDNA Synthesis Kit (purchased from Thermo Company) to reverse transcribe the total RNA extracted in the previous step to obtain cDNA, and freeze it at -20°C for later use;
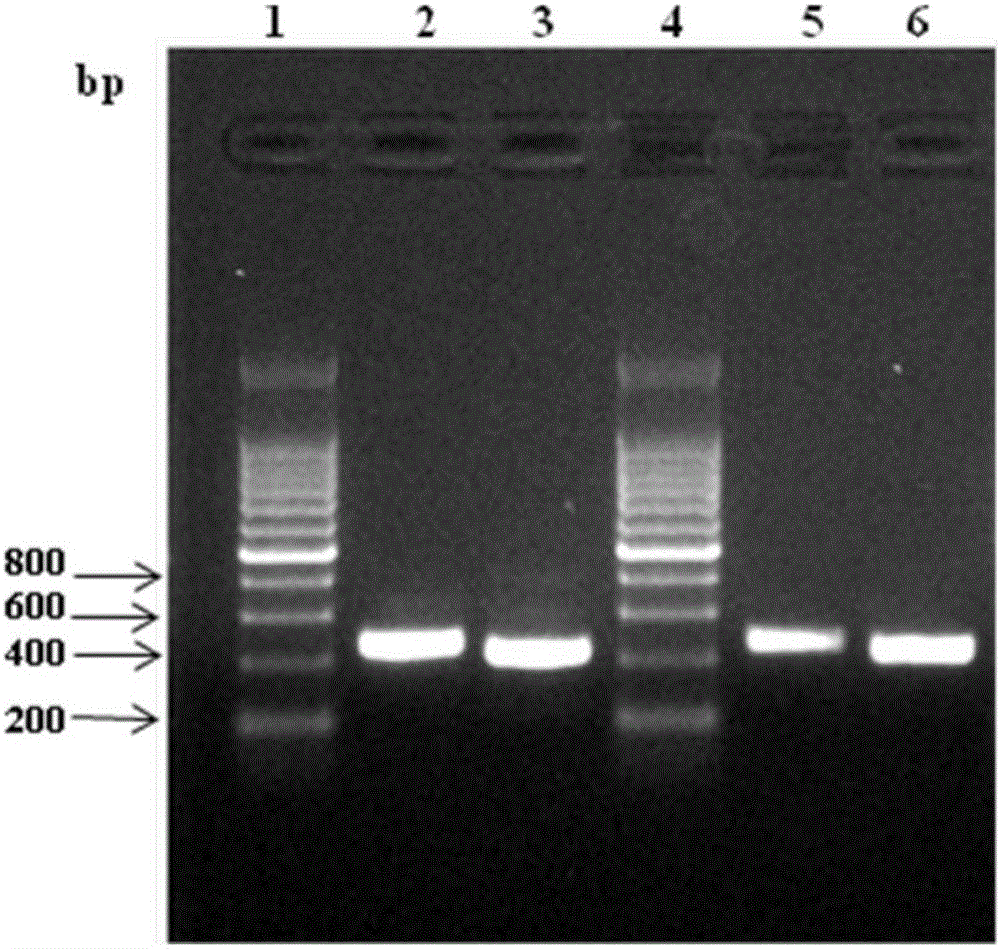
[0061] c. PCR amplification and recovery of the variable region sequence: the cDNA obtained in the above step is used as a template, and the variable region sequence of the heavy chain and light chain is sequenced with the genera...
Embodiment 3
[0065] Example 3. Recombinant expression and purification of single-chain antibody
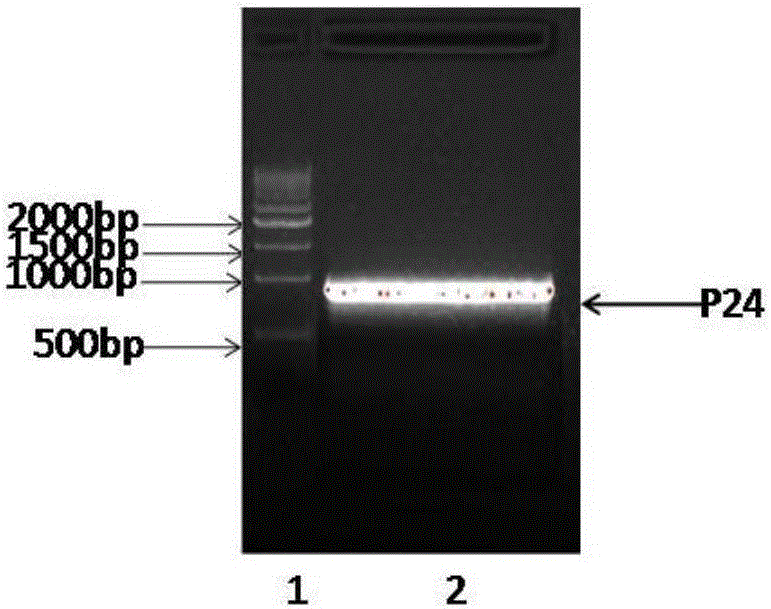
[0066] According to the sequencing results in Example 2, a linking peptide (GGGGS) was added between the heavy chain and light chain variable regions of the hybridoma cell lines M24 and M03 antibody respectively 3 , introduce six histidine tags SEQ ID NO: 17, and perform recombinant expression of single-chain antibodies by codon-optimizing the whole gene fusion histidine tags according to the preference of the Pichia pastoris expression system. The expressed antibodies were named as antibody P24 and antibody P03 respectively, and their structures and compositions are shown in the attached figure 2 shown. The recombinant expression of the above-mentioned single-chain antibody is specifically as follows:
[0067] a) Expression plasmid construction of fusion protein gene
[0068] The nucleotide sequence of the codon-optimized antibody P24 is shown in SEQ ID NO:18, and the amino acid sequence ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More