Fluorescent PCR (Polymerase Chain Reaction) detection kit for African swine fever virus, preparation method and application method
A technology of African swine fever virus and detection kit, which is applied in the direction of biochemical equipment and methods, microbial measurement/inspection, etc., can solve the problems of large error in detection results, unsuitable early diagnosis, and susceptibility to environmental influences, etc., to reduce The effects of missed detection, good repeatability, excellent sensitivity and specificity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0072] The embodiment of the present invention also provides a preparation method of the above-mentioned African swine fever virus fluorescent PCR detection kit, comprising:
[0073] S110, select the sequence of the highly conserved region in the p72 gene of the ASFV genome, design multiple sets of primers and probes, respectively amplify the multiple sets of primers and probes, and screen the amplification efficiency, probe signal-to-noise ratio and amplification A group with the best curve shape as the primer and the probe of the ASFV-reaction solution;
[0074] S120, synthesize the recombinant cloning plasmid numbered pUC-p72, the recombinant cloning plasmid contains the target nucleotide sequence of the amplified product of the primer in the ASFV-reaction solution, and use a protein and nucleic acid analyzer to analyze the recombinant cloning plasmid The concentration is quantified, and the quantified recombinant cloning plasmid is diluted by TE buffer solution to obtain m...
Embodiment 1
[0100] The preparation method of embodiment 1 kit
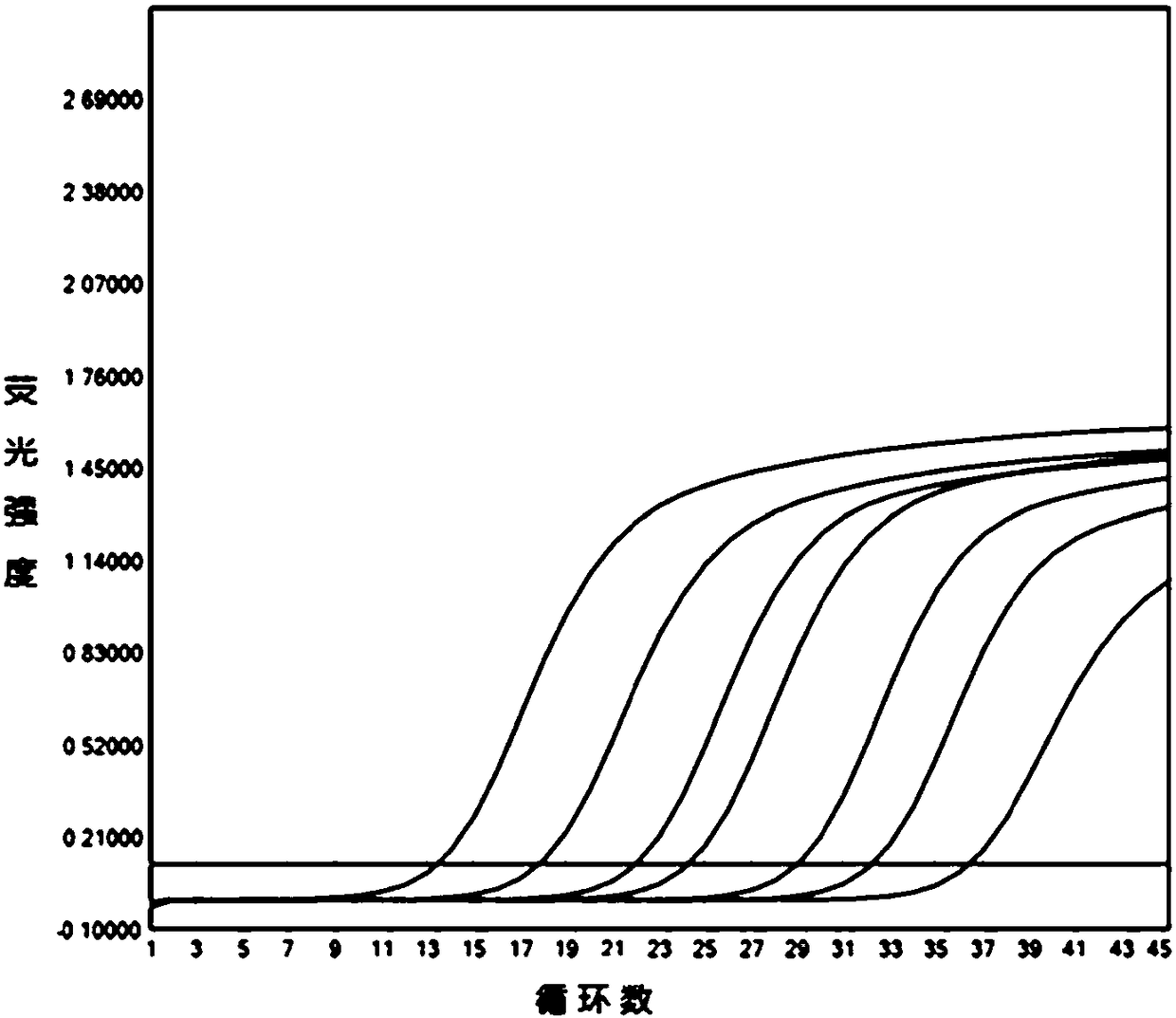
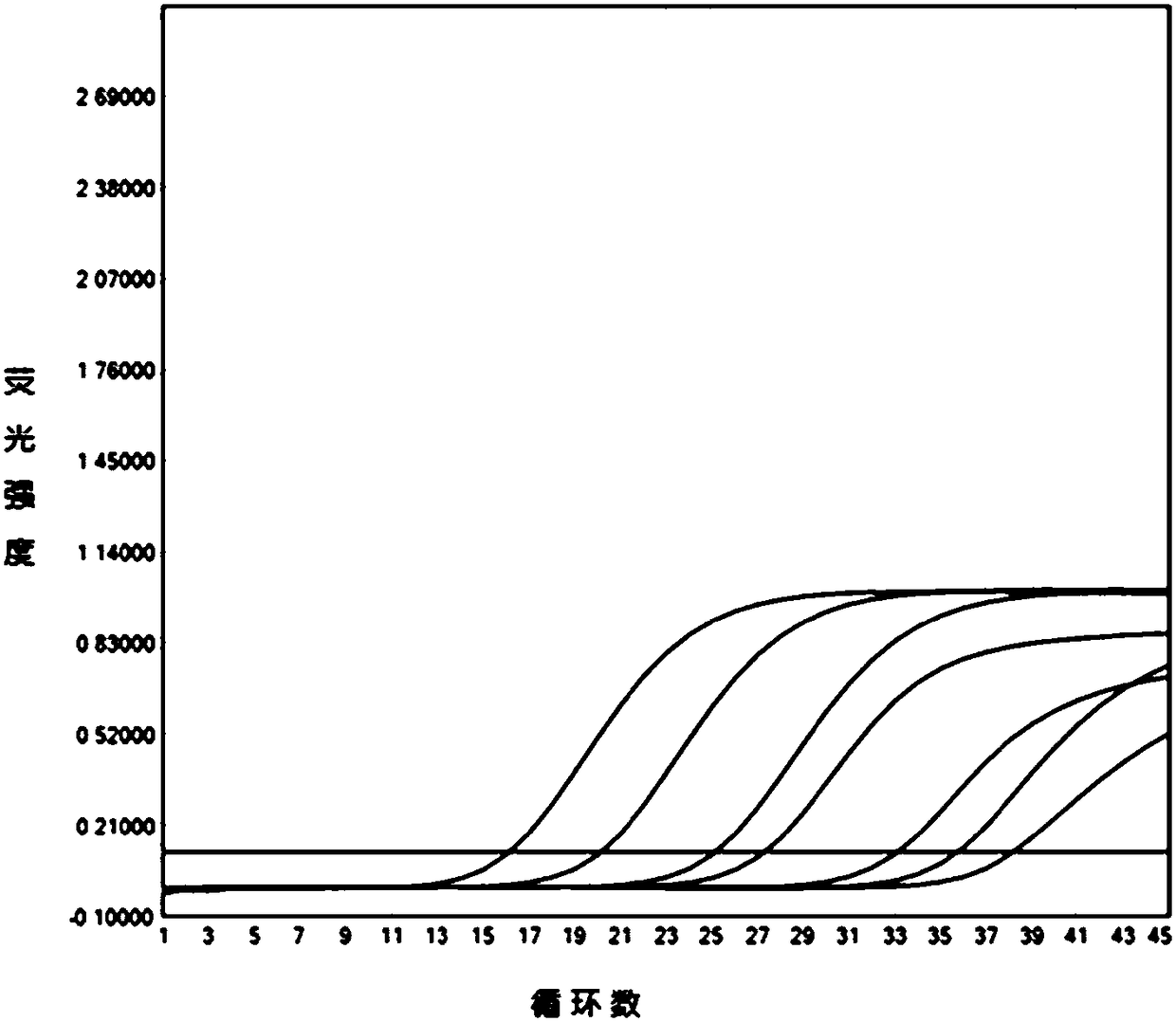
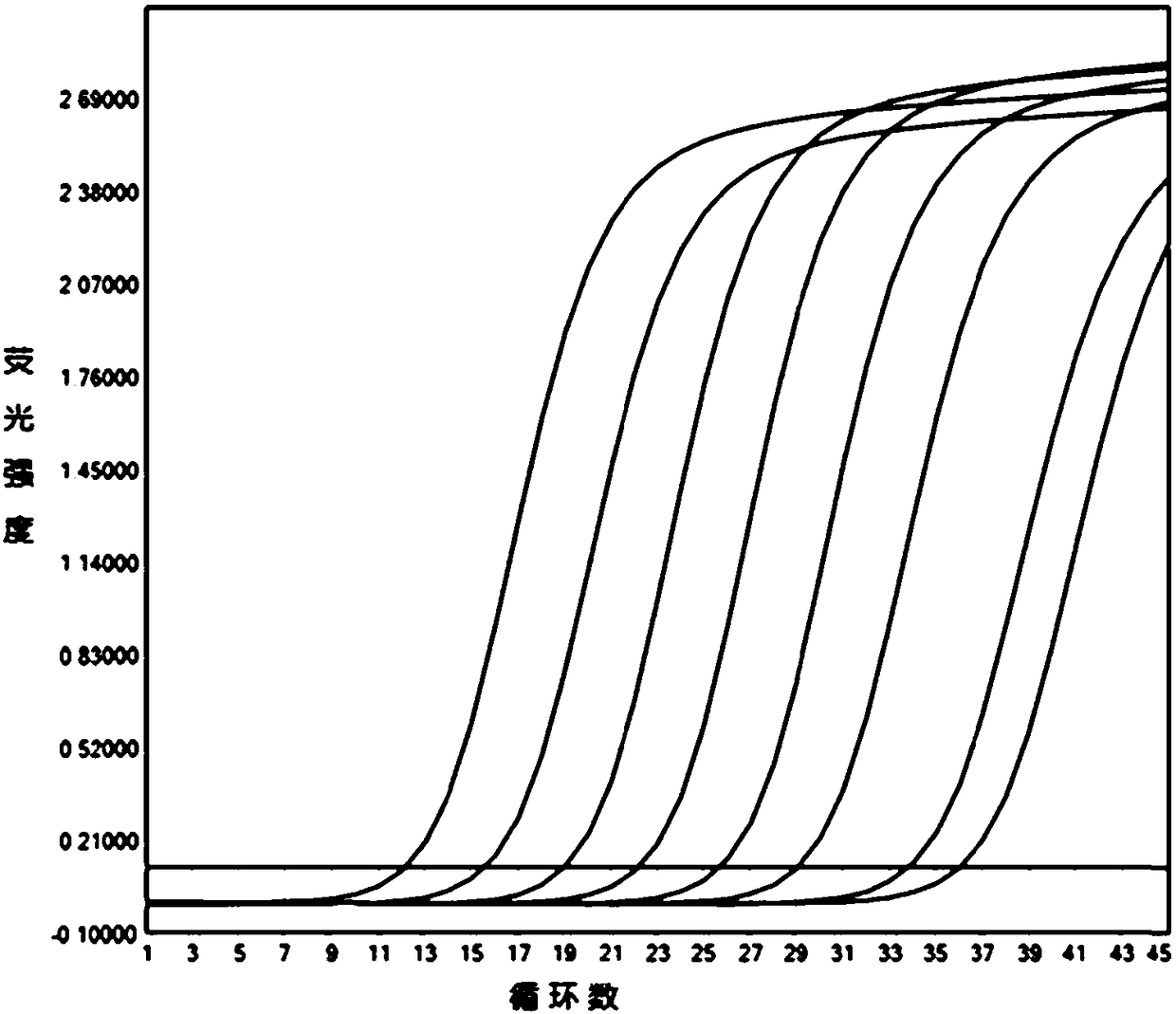
[0101] Analyzed and compared the p72 gene sequence information of 23 ASFV strains registered in NCBI, and selected highly conserved region sequences to design three sets of specific primers and probe sequences. Please refer to Table 1 for the sequences and marker information of each set. see Figure 1 to Figure 3 Amplification tests were carried out on the above three sets of primers and probes respectively, and the indicators such as amplification efficiency, probe signal-to-noise ratio, and amplification curve shape of each group were obtained. The curve shape and sensitivity of the third group are the best, and there is no mismatch between the probe sequence and some current ASFV strain sequences, so it is selected as the best primer and probe set.
[0102] The cloned plasmid pUC-p72 was synthesized according to the amplified product of the target gene of the ASFV primer obtained by the above screening, and its sequence w...
Embodiment 2
[0129] The usage method of embodiment 2 kit
[0130] Sample collection: dissect sick pigs, collect lungs, spleen, liver, lymph nodes and other tissues; or collect blood and tonsils from living organisms.
[0131] Sample pretreatment: Serum and plasma samples do not need to be processed and can be directly used for detection; other tissue samples need to be processed, take about 1g and grind it in a grinding device, add 1.2mL normal saline to grind until homogenized, transfer to a 1.5mL centrifuge tube, and centrifuge at 8000g After 5 minutes, the supernatant was diluted 10 times with normal saline for detection.
[0132] Amplification reagent preparation: Take out all the components in the packaging box except the DNase mixture, place them at room temperature, shake and mix them after they are completely dissolved; 2μL / portion+ASFV-built-in reference 0.5µL / portion) Take the corresponding amount of reagents, mix well to form PCR-Mix, and centrifuge briefly.
[0133] Nucleic a...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More