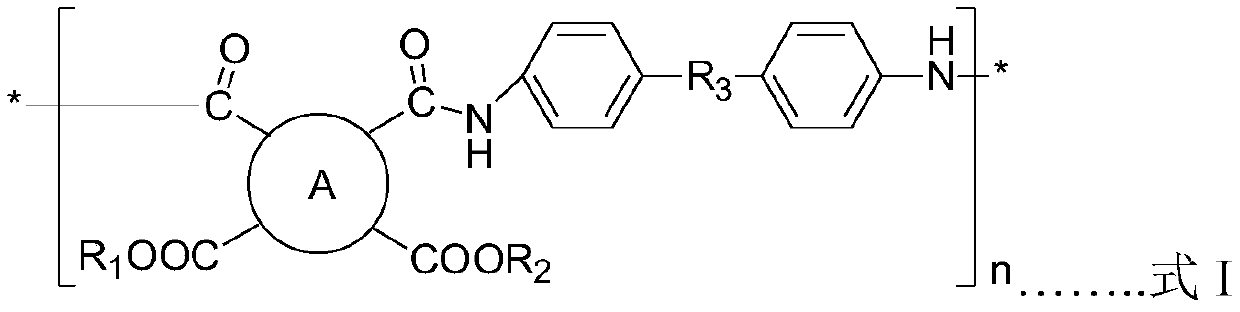
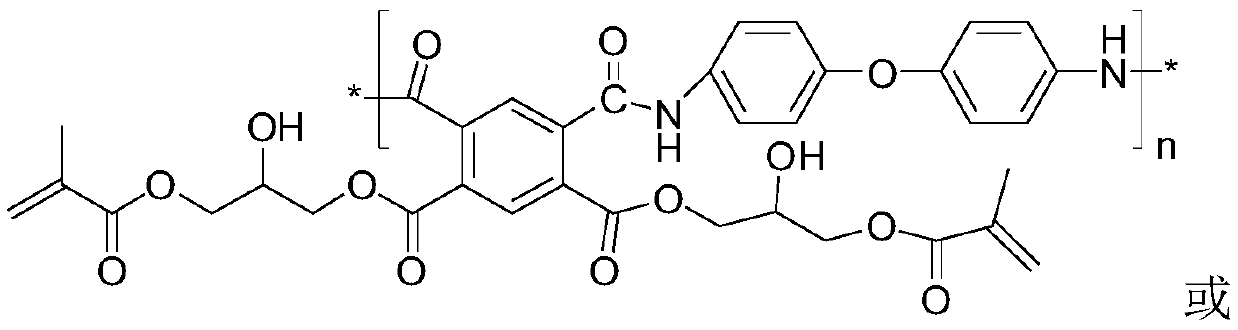
Polyimide precursor, preparation method and resin composition and application thereof
A technology of polyimide precursor and resin composition, which is applied in the field of resin composition, polyimide precursor and preparation, can solve the problem that the main chain flexibility and aromatic ring side groups are not compatible, cracking, carboxyl group pair amide group autocatalytic cracking and other problems, to achieve the effect of easy self-degradation, prolonged storage time, and inhibition of self-degradation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0058] This embodiment prepares the polyimide precursor through the following steps
[0059]
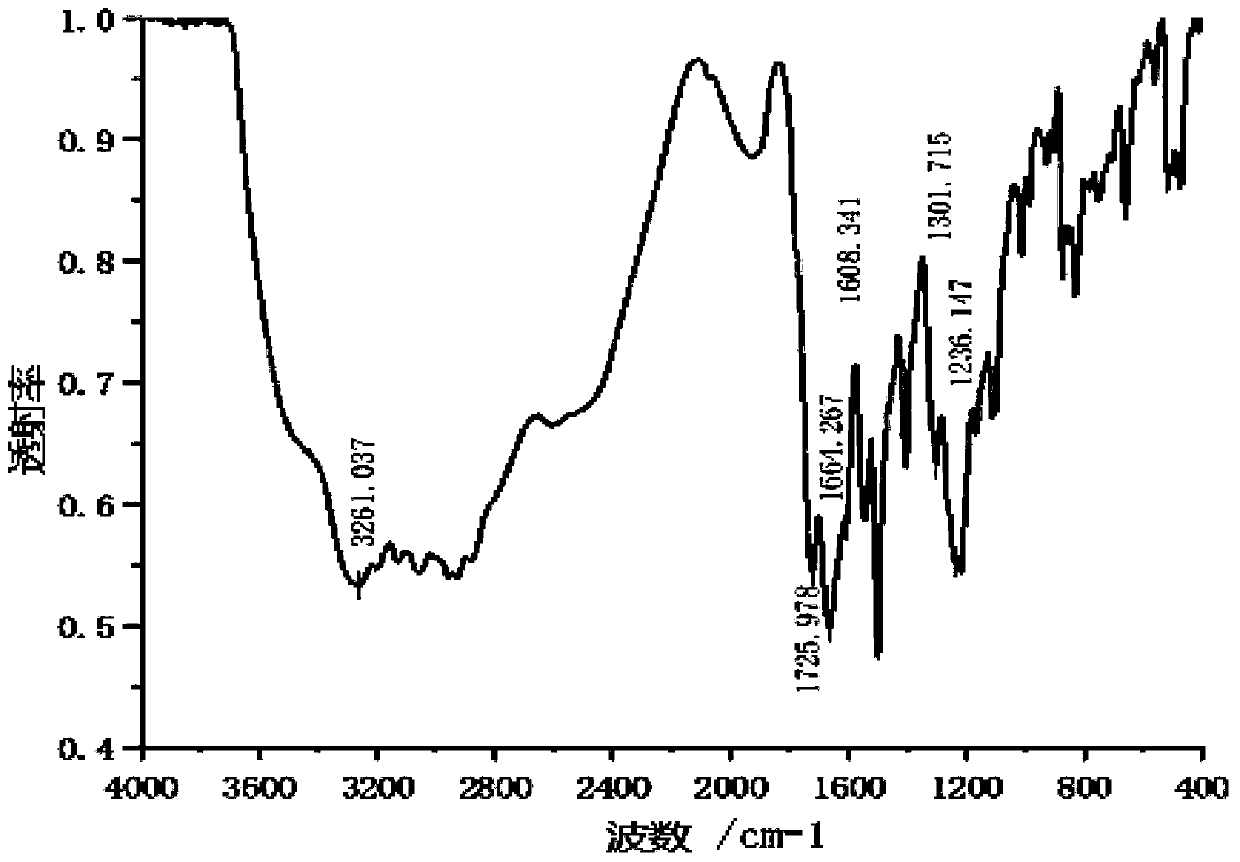
[0060] (1) 43.624 g (200 mmol) of pyromellitic dianhydride, 57.002 g (401 mmol) of glycidyl methacrylate and 0.220 g of hydroquinone dried in a dryer at 160°C for 24 hours were dissolved in 394 g of N-methylpyrrolidone In , after adding a catalytic amount of 1,8-diazabicycloundecene, it was stirred at room temperature (25° C.) for 24 hours to perform esterification, thereby obtaining a pyromellitic acid-glycerol methacrylate solution.
[0061] (2) Add 195.564 g of the solution obtained in step (1) to a 0.5 L flask equipped with a stirrer and a thermometer, and then add 25.9 g of thionyl chloride dropwise using a dropping funnel under ice cooling to keep the temperature of the reaction solution below 10°C. g (217.8 mmol). After the dropwise addition of thionyl chloride was completed, the reaction was carried out under ice cooling for 2 hours to obtain an acid chloride solution of ...
Embodiment 2
[0065] This embodiment prepares the polyimide precursor through the following steps
[0066]
[0067] (1) 58.844 g (200 mmol) of 3,3',4,4'-biphenyltetracarboxylic dianhydride, 57.002 g (401 mmol) of glycidyl methacrylate, and hydroquinone dried in a dryer at 160°C for 24 hours 0.220g was dissolved in 350g N,N-dimethylformamide, after adding a catalytic amount of 1,8-diazabicycloundecene, stirred at room temperature (25°C) for 24 hours to carry out esterification, thereby A 3,3',4,4'-biphenyltetracarboxylic dianhydride-glycerol methacrylate solution was obtained.
[0068] (2) Add 213.52 g of the solution obtained in step (1) to a 0.5 L flask equipped with a stirrer and a thermometer, and then add 26.5 g of thionyl chloride dropwise using a dropping funnel under ice cooling to keep the temperature of the reaction solution below 10°C. g (222.5 mmol). After the dropwise addition of thionyl chloride was completed, the reaction was carried out under ice cooling for 2.5 hours to...
Embodiment 3
[0071] This embodiment prepares the polyimide precursor through the following steps
[0072]
[0073] (1) Dissolve 43.624 g (200 mmol) of pyromellitic dianhydride, 12.802 g (401 mmol) of methanol, and 0.220 g of hydroquinone dried in a dryer at 160°C for 24 hours in 353 g of dimethyl sulfoxide, and add a catalytic amount The 1,8-diazabicycloundecene was stirred at room temperature (25° C.) for 24 hours to perform esterification, thereby obtaining a pyromellitic acid-methyl ester solution.
[0074] (2) Add 172.55 g of the solution obtained in step (1) to a 0.5 L flask equipped with a stirrer and a thermometer, and then add 24.8 g of thionyl chloride dropwise using a dropping funnel under ice cooling to keep the temperature of the reaction solution below 10°C. g (208.9 mmol). After the dropwise addition of thionyl chloride was completed, the reaction was carried out under ice cooling for 2 hours to obtain an acid chloride solution of PMDA. Next, using a dropping funnel, a sol...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Sensitivity | aaaaa | aaaaa |
| Sensitivity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com