A kind of verapamil thermosensitive sustained-release preparation and its preparation method and application
A slow-release preparation and temperature-sensitive gel technology, which is applied in the field of medicine, can solve problems such as interference with normal physiological functions, and achieve the effects of alleviating the washing effect, obvious slow-release effect, and high encapsulation rate
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
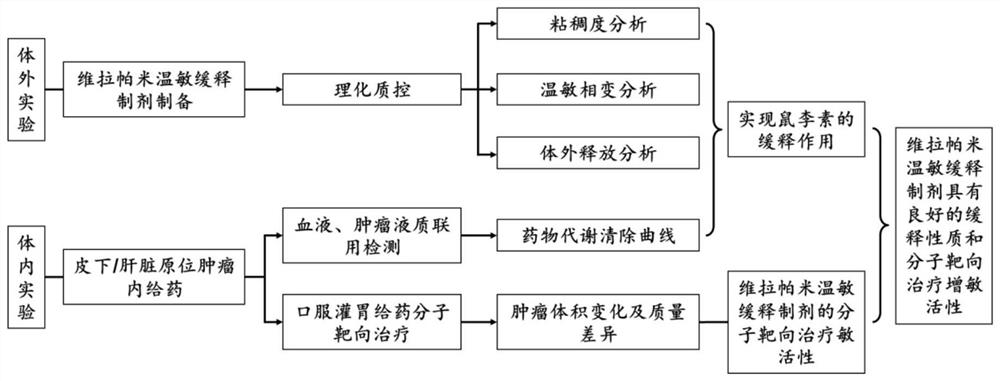
[0035] Example 1: Preparation of drug-loaded albumin nanoparticles-multilamellar liposomes-thermosensitive adhesive three-phase sustained release system
[0036] Research Methods and Experimental Means
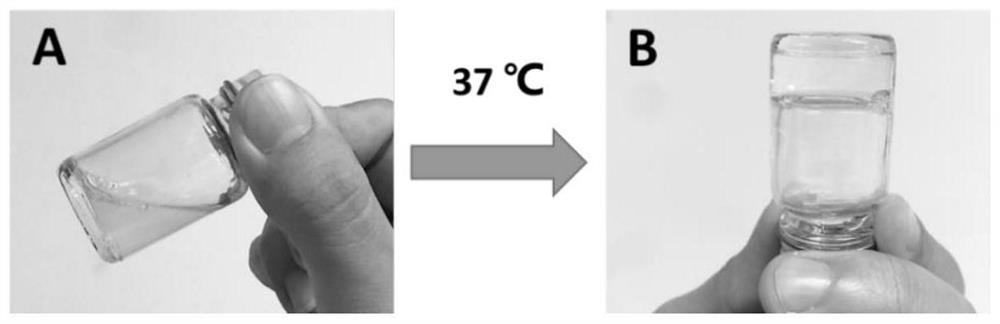
[0037] (1) Preparation process of drug-loaded albumin nanoparticles-multilamellar liposomes-thermosensitive adhesive three-phase sustained release system
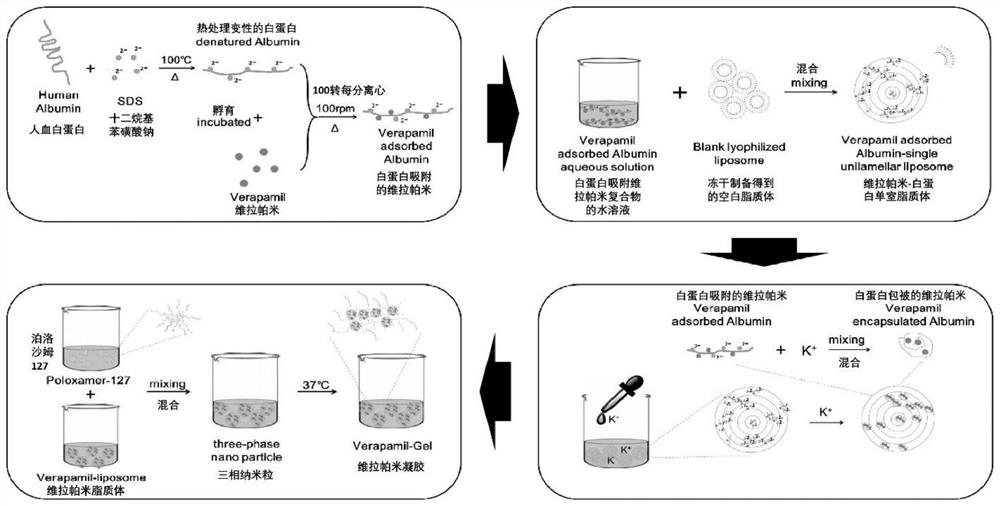
[0038] ①Preparation of drug-loaded denatured albumin: adding an appropriate amount of sodium dodecyl sulfate (SDS) to an aqueous solution of human serum albumin with a volume to mass ratio of 5%, boiling for 10 minutes to allow protein denaturation to complete, and then adding an aqueous solution of verapamil (Concentration is 10mg / mL, add 0.1-2mL), incubate at 100rpm, 37℃ for 6h, make the drug adsorbed on the hydrophobic domain of the protein, and form drug-loaded denatured albumin, the unadsorbed free drug is removed by dialysis ;
[0039] ②Formation of drug-loaded albumin nanoparticles-multilamellar liposome composite ...
Embodiment 2
[0043] Example 2: Establishment of HCC mouse liver orthotopic model and rat liver orthotopic tumor model
[0044] Research Methods and Experimental Means
[0045] ①Cultivate HCC cell lines HepG2 and MHCC-97H, etc., inoculate the cultured HCC cell lines subcutaneously in immunodeficiency mice, and after 3-4 weeks of growth, form a subcutaneous tumor model of HCC in immunodeficiency mice;
[0046] ② HCC cells were first inoculated subcutaneously in immunodeficient mice to form solid tumors, and small tissue masses with rich blood vessels on the surface of the solid tumors and in good condition were selected, and then inoculated into the livers of immunodeficient mice. After 4-6 weeks of growth, cancer nests with clear outlines can be formed in the liver of immunodeficient mice, which is the orthotopic HCC model (cancer nests) in the liver of immunodeficient mice;
[0047] ③Cultivate rat liver cancer cells Walker-256 cells, inoculate the cultured HCC cell line into SD rats subcu...
Embodiment 3
[0051] Example 3: Detection of metabolic properties of verapamil temperature-sensitive sustained-release preparations
[0052] Research Methods and Experimental Means
[0053] ①Establish and configure verapamil aqueous solution (use DMSO and other organic solvents to dissolve verapamil first, and then dilute with normal saline), verapamil solubilization solution (DMSO or solubilized solution of verapamil) , Blank nano-sustained-release preparation as a control;
[0054] ②Using the subcutaneous tumor model of immunodeficiency mice established above, solvent control, verapamil solubilization solution (solubilization preparation), and verapamil temperature-sensitive sustained-release preparation were injected into the subcutaneous tumor respectively. time to collect tumor specimens;
[0055] ③Using the SD rat subcutaneous tumor model established above, the verapamil aqueous solution was injected through the tail vein, or the solvent control, verapamil solubilization solution (s...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More