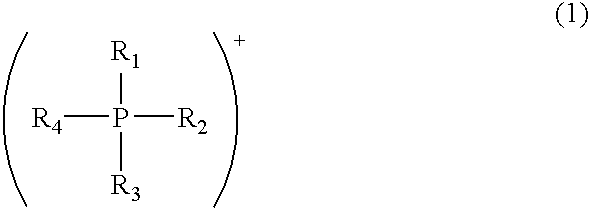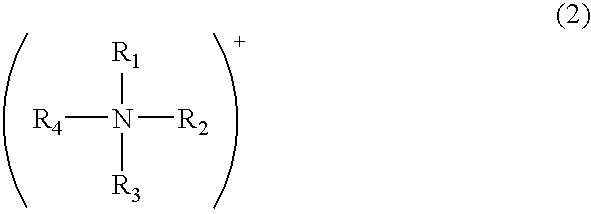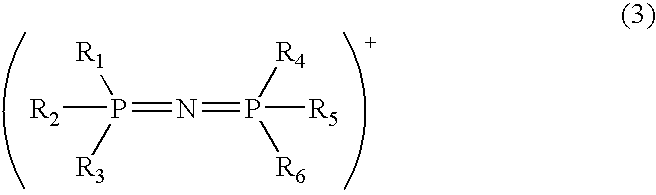Modified acidic ion-exchange resin and method for preparing bisphenol
a technology of acidic ion exchange resin and bisphenol, which is applied in the direction of organic compound/hydride/coordination complex catalyst, physical/chemical process catalyst, bulk chemical production, etc., can solve the problems of insufficient conversion of acetone, complex process, corrosion of apparatus used in reaction, etc., and achieve the effect of increasing the selective effect of bisphenol
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0045] (1) Preparation of a Modified Ion-Exchange Resin Catalyst
[0046] While vigorously stirring the water-swollen Amberlyst 31 (dry weight 4 g) with 80 ml of ion-exchange water, thereto was slowly added dropwise 40 ml of an aqueous n-tetrabutylammonium chloride solution having a concentration of 0.153 mole / L. After completing the dropwise addition, the mixture was further stirred for 5 hours, and was then repeatedly filtered and washed with ion-exchange water. Thereafter, it was dried under vacuum at 80° C. for 10 hours or longer to obtain Catalyst 1. The results of the measurement of an acid amount and of the elemental analysis in the Catalyst are shown in Table 1.
[0047] (2) Synthesis Reaction of Bisphenol A
[0048] 6.63 g of phenol, 0.37 g of acetone and 0.35 g of Catalyst 1 prepared in (1), were put into a 70 ml pressure-resistant reactor, and 3-mercaptopropionic acid was put thereto such that the concentration is 3000 ppm. Then, the pressure-resistant reactor was pressurized w...
example 2
[0049] In the same manner as in (1) of Example 1, except that an aqueous tetramethylammonium chloride solution having a concentration of 0.153 mole / L was used instead of an aqueous tetrabutylammonium chloride solution, the operation was performed to obtain Catalyst 2. The results of the measurement of an acid amount and of the elemental analysis in the Catalyst are shown in Table 1. In addition, under the same conditions as in (2) of Example 1, except that Catalyst 2 was used instead of Catalyst 1, the synthesis reaction of bisphenol A was performed. The results were shown in Table 2.
example 3
[0050] In the same manner as in (1) of Example 1, except that an aqueous n-dodecyltrimethylammonium chloride solution having a concentration of 0.153 mole / L was used instead of an aqueous tetrabutylammonium chloride solution, the operation was performed to obtain Catalyst 3. The results of the measurement of an acid amount and of the elemental analysis in the Catalyst are shown in Table 1. In addition, under the same conditions as in (2) of Example 1, except that Catalyst 3 was used instead of Catalyst 1, the synthesis reaction of bisphenol A was performed. The results were shown in Table 2.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More