Method for enhancing immune responses in mammals
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Production, Purification, and Characterization of the Immune System Inhibitor, Human sTNFRI
[0077] The sTNFRI used in the present studies was produced recombinantly either in E. coli (R&D Systems; Minneapolis Minn.) or in eukaryotic cell culture essentially as described (see U.S. Pat. No. 6,379,708, which is incorporated herein by reference). The construction of the eukaryotic expression plasmid, the methods for transforming and selecting cultured cells, and for assaying the production of sTNFRI by the transformed cells have been described (Selinsky et al., supra, 1998).
[0078] sTNFRI was detected and quantified in the present studies by capture ELISA (Selinsky et al., supra). In addition, the biological activity of recombinant sTNFRI, that is, its ability to bind TNF, was confirmed by ELISA. Assay plates were coated with human TNF a (Chemicon; Temecula Calif.), blocked with bovine serum albumin, and sTNFRI, contained in culture supernatants as described above, was added. Bound sTNF...
example 2
Production, Purification, and Characterization of TNFα Muteins
[0079] Briefly, TNFα muteins 1, 2, 3 and 4 were produced by expression of the respective cDNAs in E. coli. Genes encoding TNFα and TNFα muteins 1, 2, 3 and 4 were prepared using overlapping oligonucleotides having codons optimized for bacterial expression. Each of the coding sequences was fused in frame to that encoding the ompA leader to permit export of the recombinant polypeptides to the periplasm. Synthetic fragments were cloned into a pUC19 derivative immediately downstream of the lac Z promoter, and the resulting recombinant plasmids were introduced into E. coli. Recombinant bacteria were cultured to late-log, induced with isopropyl-β-D-thiogalactopyranoside (IPTG) for three hours, and harvested by centrifugation. Periplasmic fractions were prepared and tested by ELISA using polyclonal goat anti-human TNFα capture antibodies. After the addition of the diluted periplasms, bound TNFα and TNFα muteins 1, 2, 3 and 4 we...
example 3
Depletion of the Immune System Inhibitor, sTNFRI, from Human Plasma Using TNFα Mutein Absorbent Matrices
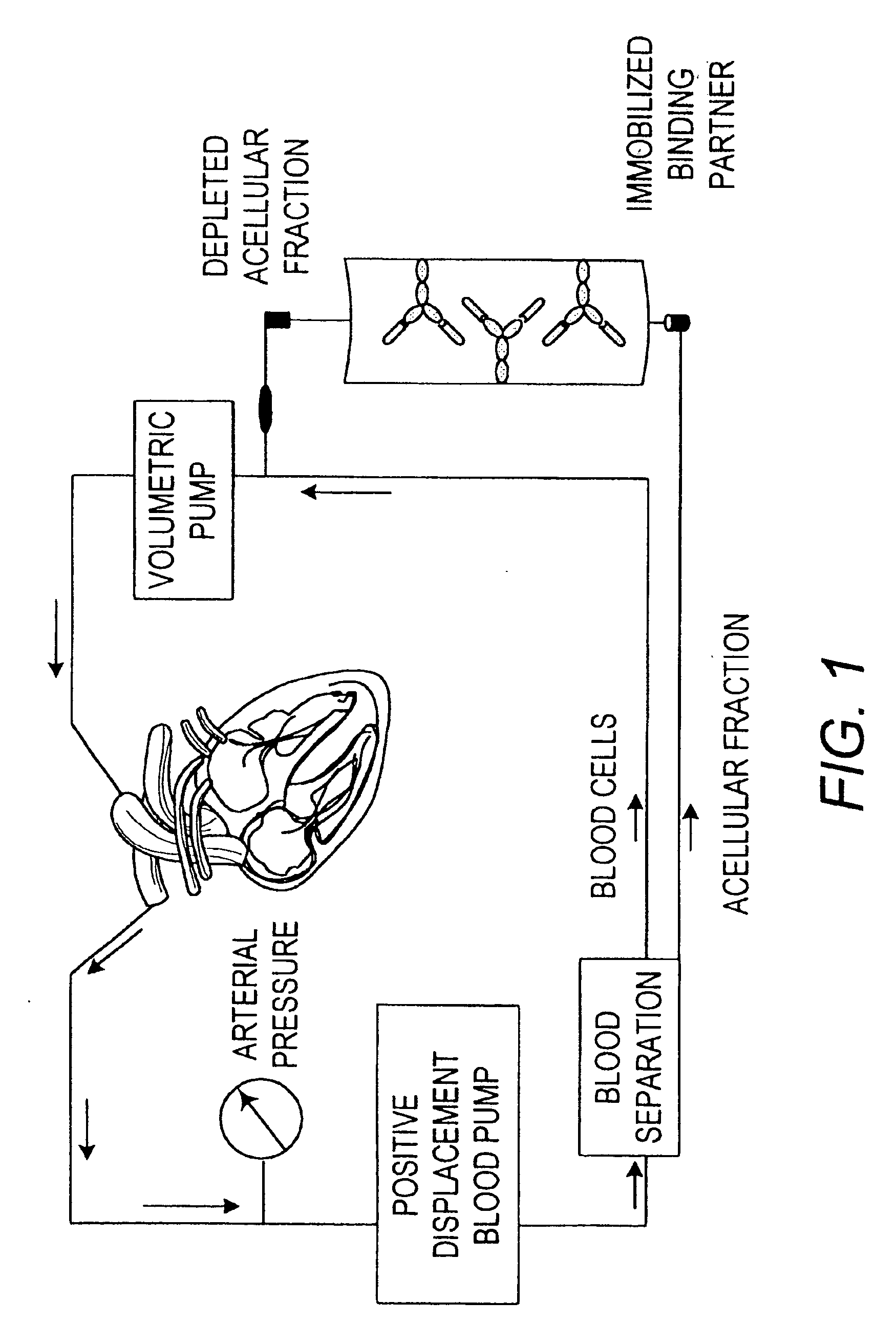
[0082] The TNFα mutein absorbent matrices were produced and tested for their ability to deplete sTNFRI from human plasma. Briefly, purified TNFα muteins 1, 2 and 4 each was conjugated to cyanogen bromide (CNBr) Sepharose™ 4B at a density of 0.5 mg per ml of beads, and the remaining CNBr groups were quenched with ethanolamine. The resulting matrices were packed in individual column housings and washed extensively with phosphate buffered saline prior to use.
[0083] Normal human plasma was spiked (33% v / v) with culture supernatant containing recombinant human sTNFRI (see Example 1) to a final concentration of 8 nanograms per milliliter and passed through the respective columns at a flow rate of one milliliter of plasma per milliliter of resin per minute. An additional column, with no immobilized protein and quenched with ethanolamine, was included to control for non-specific depleti...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Chemically inert | aaaaa | aaaaa |
| Solubility (mass) | aaaaa | aaaaa |
| Water absorption | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More