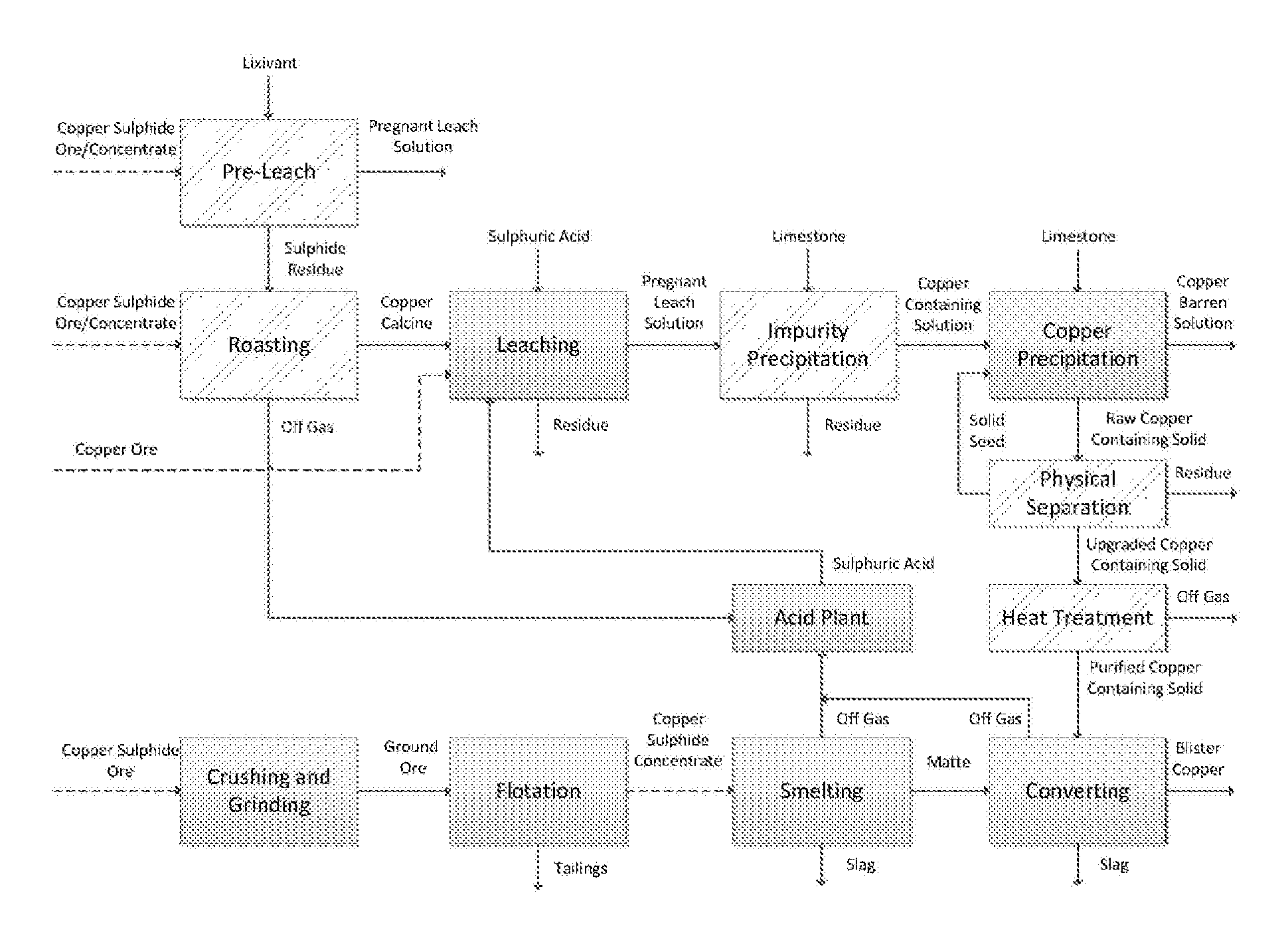
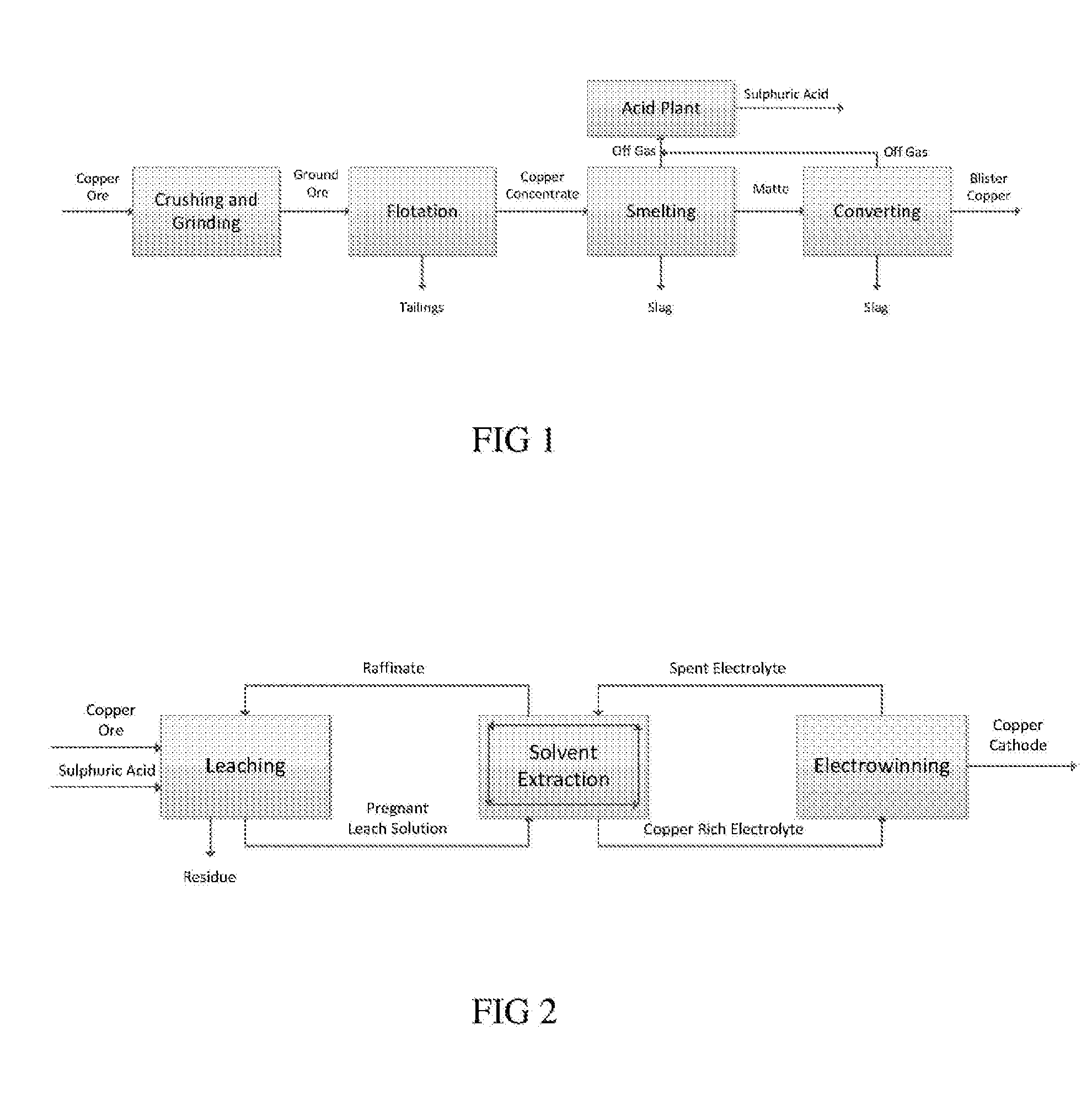
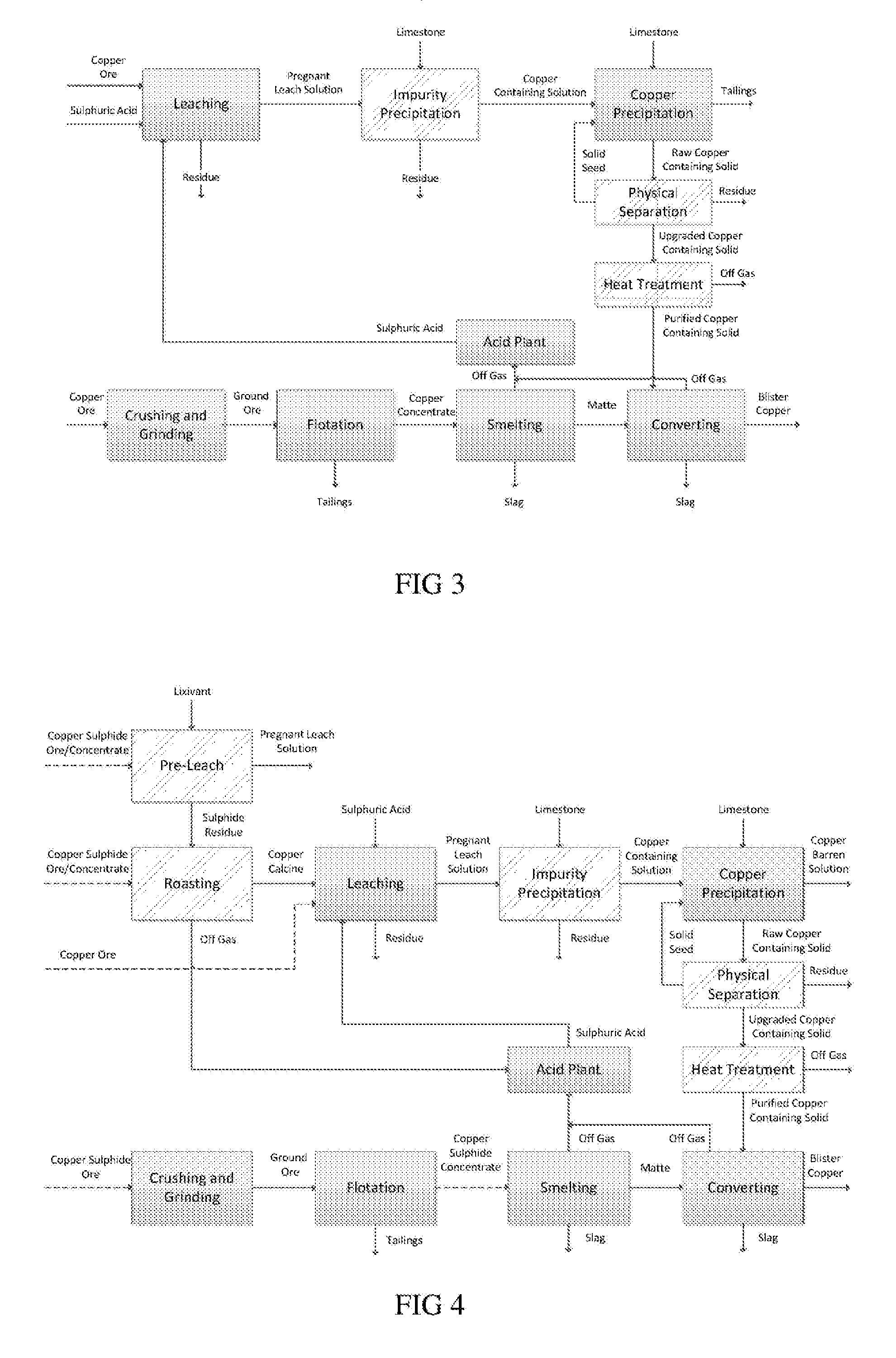
At the same time the identification of new high grade ore deposits is becoming increasingly challenging.
These two methods have some inherent advantages and particular disadvantages that become more profound as
lower grade ores are treated.
The
heat balance in these process units can be challenging to control and places limitations on the process.
One of the major energy consuming steps in this concentrate
smelting route is in the electrical power used for the
grinding of the ores.
The pyrometallurgical approach is thus economically limited in the grade of ore it can process.
This is becoming increasingly problematic as the copper content of newly discovered ores is steadily decreasing.
This creates significant problems with gas cleaning and disposal of arsenic-containing fume.
If the predominant copper minerals in an ore are not sulphides, the ore is difficult to concentrate by physical means and is unsuited to pyrometallurgical processing as the cost of heating the host rock is prohibitive in terms of energy and cost.
Further, if certain impurities (such as arsenic) in a sulphide ore and resulting concentrate surpass a critical concentration then that ore cannot be treated using the pyrometallurgical processes.
However, a handful of
key issues can make the process unsuitable for particular ore bodies.
The capital costs for the
solvent extraction and
electrowinning circuits are relatively high so the use of the hydrometallurgical process
route on some smaller or
short lifetime resources can be uneconomical.
Furthermore, the
electrowinning step is energy intensive and requires a significant source of electrical power.
As the cost of energy generation increases through increased demand and taxation, the cost of the required electrical power becomes even more prohibitive, especially in remote areas where the required infrastructure is not already installed.
A less obvious but much more technically challenging issue for hydrometallurgical processing is the huge dependence of the process on the
proton and sulphate balance.
The fact that the process regenerates sulphuric acid in the
electrowinning section is often purported as a major
advantage but it can also become a major problem in the situation where the leach circuit is also generating acid.
This is often the case in the leaching of sulphides and results in the need to neutralise a portion of the acid generated.
The required bleed to a neutralisation step results in extra
reagent costs, introduces a potential avenue of valuable
copper loss and the resulting residue requires environmentally sound storage and disposal.
These issues with the hydrometallurgical process has resulted in ore bodies which contain a
copper oxide cap over a more substantial sulphide deposit having the
oxide cap removed and discarded or stored rather than being processed and the copper value realised.
As the concentration of copper in these reserves decreases and the concentrations of
impurity elements increase the ores and concentrates are becoming increasingly difficult to treat using existing industrial process routes and technologies.
The impacts of these trends are in the form of decreasing productivity, increasing
energy consumption and costs, and increasing
capital investment and operating costs required to avoid adverse environmental impacts.
It is more difficult to effectively extract copper from such complex ores and concentrates.
For example, these complex ores often have relatively high levels of iron which must be removed at some stage.
Importantly, a percentage of the available copper will always partition into the
slag with the iron and greater quantities of
slag resulting from the higher levels of iron will inevitably result in a greater loss of copper to
slag.
It is clear that, using conventional pyre- and hydro-
metallurgy routes, as mean ore grades decrease the capital and operating costs of copper production will increase along with the electrical energy requirements and also the
greenhouse gas
impact, if the energy used is produced from fossil fuels.
In addition, arsenic is becoming even more of a problem for copper processing as its concentration in many copper ores is increasing while, at the other end, stricter limits are being placed on its environmental release.
Similar issues exist for other
impurity elements such as lead,
bismuth and a range of radioactive elements.
 Login to View More
Login to View More