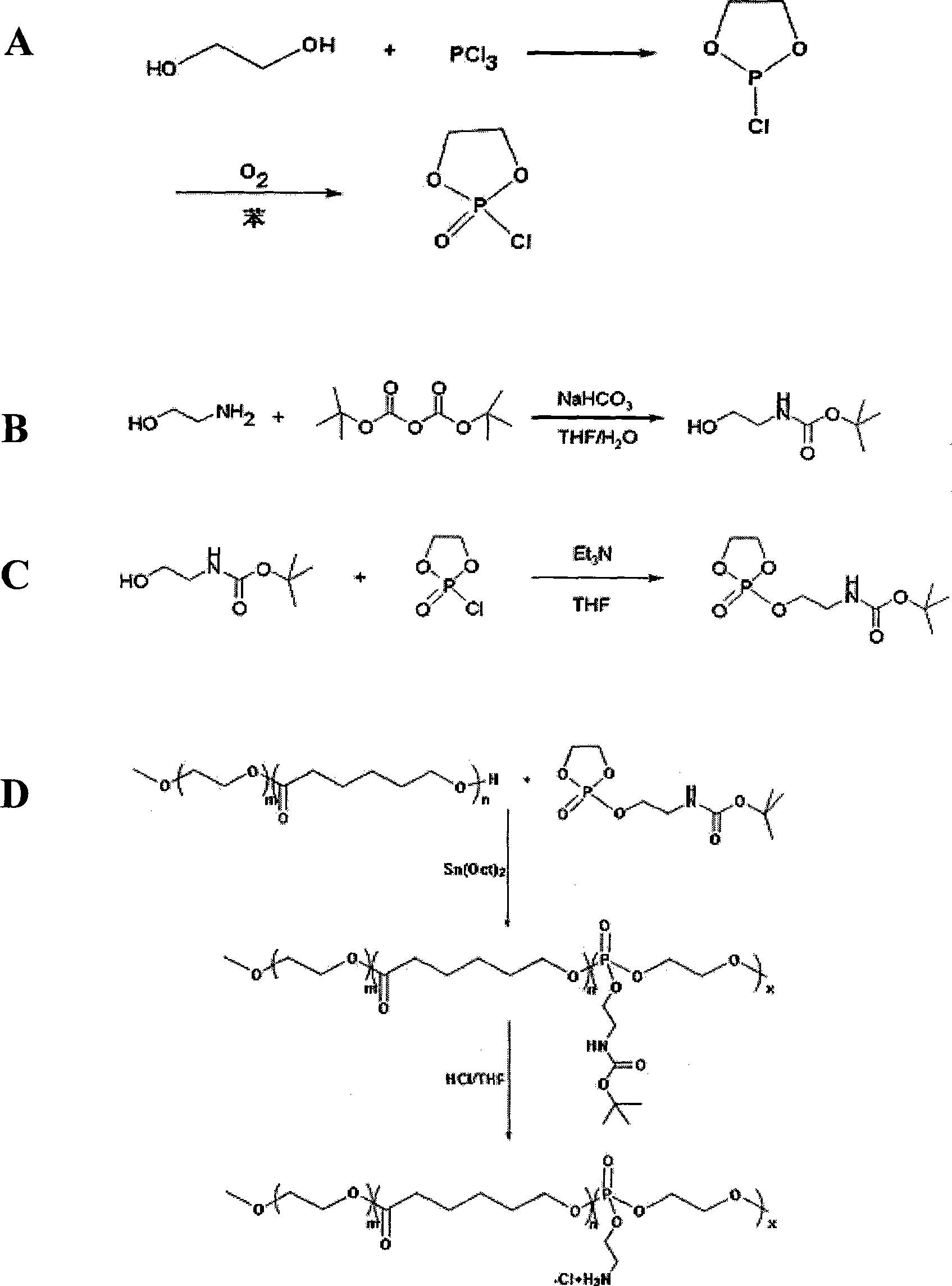Polyethylene glycol monomethyl ether-polycaprolactone-polyphosphate triblock copolymer and siRNA medicament carrier prepared thereby
A technology of polyethylene glycol monomethyl ether and polycaprolactone, which is applied in the field of siRNA drug carriers, can solve the problems of difficult process amplification and poor repeatability, and achieve good stability, high repeatability and good application prospects Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0063] Synthesis and characterization of embodiment 1, mPEG-PCL-PPEEA
[0064] 1. Synthesis and characterization of 2-(N-tert-butoxycarbonylamino)ethoxy-2-oxo-1,3,2-dioxaphospholane (PEEABoc) cyclic phosphate monomer
[0065] (1) Synthesis of 2-chloro-2-oxo-1,3,2-dioxaphospholane (COP)
[0066] COP synthesis route such as figure 1 (A) shown. The specific steps for synthesizing COP are: slowly add 301 mL of 3.25 mol / L ethylene glycol in dichloromethane to 300 mL of 3.26 mol / L phosphorus trichloride in dichloromethane. After the drop was completed, the reaction was continued for 0.5 hour, and the solvent was evaporated under reduced pressure. After distilling off the product under reduced pressure for two consecutive times, it was dissolved in benzene and passed through 3 days. 2 Until the reaction is complete, distill under reduced pressure (20Pa), collect the distillate at 72°C to obtain COP.
[0067] (2) Synthesis and characterization of N-tert-butoxycarbonylaminoethanol...
Embodiment 2
[0109] Embodiment 2, prepare nanoparticle with mPEG-PCL-PPEEA copolymer
[0110] 1. Preparation of nanoparticles
[0111] Amphiphilic block copolymers have hydrophobic interactions between hydrophobic blocks in aqueous solution, and nanoparticles can be formed as long as the copolymer concentration is higher than the critical aggregation concentration.
[0112] There are many ways to prepare nanoparticles, the most simple and common one is the solvent evaporation method, the specific method is: dissolve 10mg mPEG-PCL-PPEEA triblock copolymer in 1mL tetrahydrofuran, stir at room temperature for 1 hour, and then 10 mL of ultrapure water was added dropwise at a rate of 60 mL / h, and after stirring for two hours, the organic solvent was removed under reduced pressure, and the volume was adjusted to 10 mL to obtain a 1 mg / mL nanoparticle solution.
[0113] Prepare 1 mg / mL of mPEG separately by the above method 45 -PCL 45 -PPEEA 7 Nanoparticle solution and 1 mg / mL of mPEG 45 -PC...
Embodiment 3
[0130] Embodiment 3, the application of mPEG-PCL-PPEEA nanoparticle as siRNA carrier
[0131] siRNA is a double-stranded small molecule RNA composed of more than twenty nucleotides, with a negative charge, and can form a stable complex with positively charged mPEG-PCL-PPEEA nanoparticles through the interaction of positive and negative charges.
[0132] N / P refers to the ratio of the positive charge of the amine group of the nanoparticle to the negative charge of the siRNA phosphate group.
[0133] 1. Characterization of complexes of nanoparticles and siRNA
[0134] It is the mPEG of 5mg / mL that the method for preparing concentration according to embodiment 2 45 -PCL 45 -PEEP 7 Nanoparticle solution. Green fluorescent protein GFP siRNA, whose sequence is GCAAGCTGACCCTGAAGTTCAT, was purchased from Shanghai Gemma Pharmaceutical Technology Co., Ltd., 10D GFP siRNA was dissolved in 150 μL DEPC-treated water to obtain a 20 μM siRNA solution. The above-mentioned nanoparticles w...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More