Biphenyl compound with anti-tumor activity and preparation method thereof
A technology of anti-tumor activity and compounds, applied in the field of biomedicine, can solve the problems of chemotherapy drug toxicity and side effects, and tumor chemotherapy can not meet expectations, and achieve the effect of cheap and easy-to-obtain reagents, mild reaction conditions, and easy-to-source sources
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
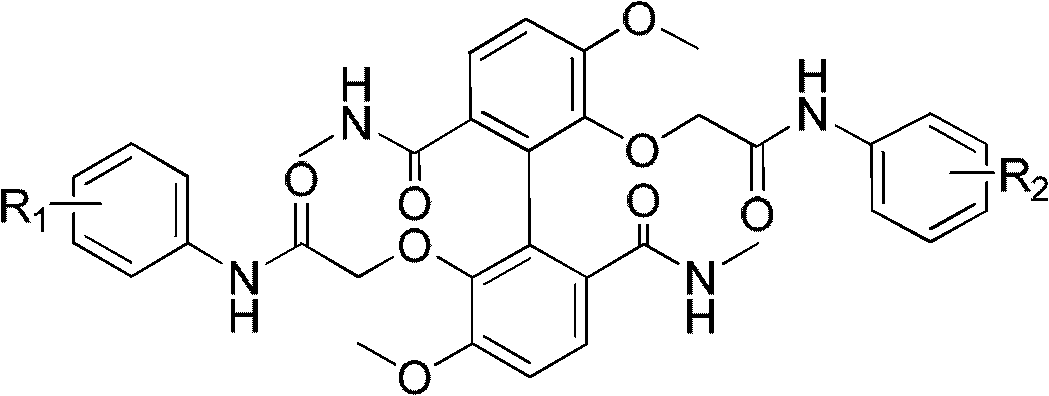
[0032] R in the structural formula of embodiment 1 1 , R 2 Compounds that are both 3-chloro-4-fluorohalogens, prepared by the following steps:
[0033] 2-Chloro-N-(3-chloro-4-fluorophenyl)acetamide
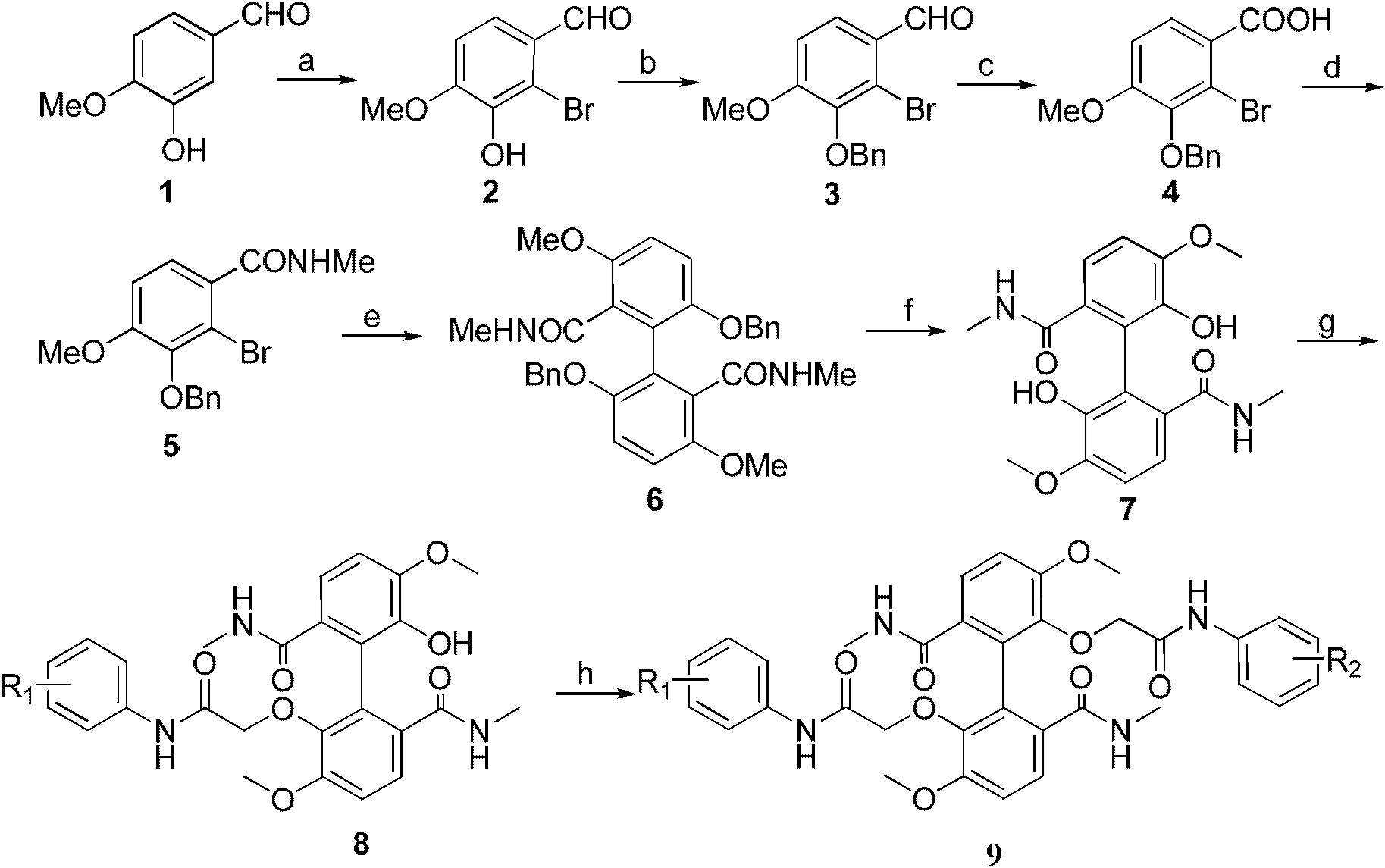
[0034] 1) Isovanillinization (1) Preparation of compound 2-bromoisovanillin (2) by bromination reaction
[0035] Put 20.0g (0.132mol) of isovanillin (1), 21.59g (0.263mol) of sodium acetate and 0.68g (0.012mol) of iron powder into a 500mL three-necked flask, add 120ml of glacial acetic acid, and stir at room temperature for 30min;
[0036] After the stirring is completed, add dropwise the solution prepared by mixing 7.0mL (0.14mol) liquid bromine and 30mL glacial acetic acid in advance under the condition that the temperature is controlled at 23-25°C. ;
[0037] Then add 250 mL of ice water, stir for 1 h; filter, evaporate the solid to dryness, and recrystallize from ethanol to obtain 24.59 g of an off-white solid product of 2-bromoisovanillin (2), with a yield of 81%;
[003...
Embodiment 2
[0069] R in the structural formula of embodiment 2 1 for hydrogen, R 2 Is the compound of 4-hydroxyl, prepared by the following steps:
[0070]Steps 1) to 6) are the same as in Example 1, that is, from compound isovanillinization (1) to compound 5,5'-dimethoxy-6,6'-dihydroxybiphenyl-2,2'- The preparation steps of dimethylformamide (7) are the same; afterwards, in the different etherification processes of the two phenolic hydroxyl groups, the two phenolic hydroxyl groups react successively with chloroacetanilide and chloroacetyl 4-hydroxyaniline, specifically:
[0071] Dissolve 3.60g (10mmol) of compound 5,5'-dimethoxy-6,6'-dihydroxybiphenyl-2,2'-dicarboxamide in (7) 200mL of anhydrous acetone, add 4.15 g (30 mmol) of anhydrous potassium carbonate, stirred at room temperature for 30 minutes, added 1.86 g (11 mmol) of 2-chloro-N-(phenyl)acetamide, and heated at reflux at 56°C for 8 hours;
[0072] After the reaction is complete, evaporate the solvent under reduced pressure, d...
Embodiment 3
[0077] R in the structural formula of embodiment 3 1 is 3-chloro-4-hydroxyl, R 2 is 3-chloro-4-fluoro-5-amino compound, prepared by the following steps:
[0078] Steps 1) to 6) are the same as in Example 1, that is, from compound isovanillinization (1) to compound 5,5'-dimethoxy-6,6'-dihydroxybiphenyl-2,2'- The preparation steps of dimethylamide (7) are identical; afterwards, in the different etherification processes of the two phenolic hydroxyl groups, the two phenolic hydroxyl groups are successively reacted with 2-chloro-N-(3-chloro-4-hydroxyphenyl) acetamide and 2-Chloro-N-(3-chloro-4-fluoro-5-aminobenzene)acetamide reaction, specifically:
[0079] Dissolve 3.60g (10mmol) of compound 5,5'-dimethoxy-6,6'-dihydroxybiphenyl-2,2'-dicarboxamide in (7) 200mL of anhydrous acetone, add 4.15 g (30 mmol) of anhydrous potassium carbonate, stirred at room temperature for 30 minutes, added 2.42 g (11 mmol) of 2-chloro-N-(3-chloro-4-hydroxyphenyl)acetamide, and heated at reflux for 8...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More