Preparation method of 1, 3-diamino guanidine hydrochloride
A technology of diaminoguanidine and hydrochloride, which is applied in the direction of organic chemistry, can solve the problems of unfavorable industrial production, high cost of cyanamide, and low yield, etc., so as to facilitate large-scale industrial production, reduce three wastes, and improve The effect of production profit
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
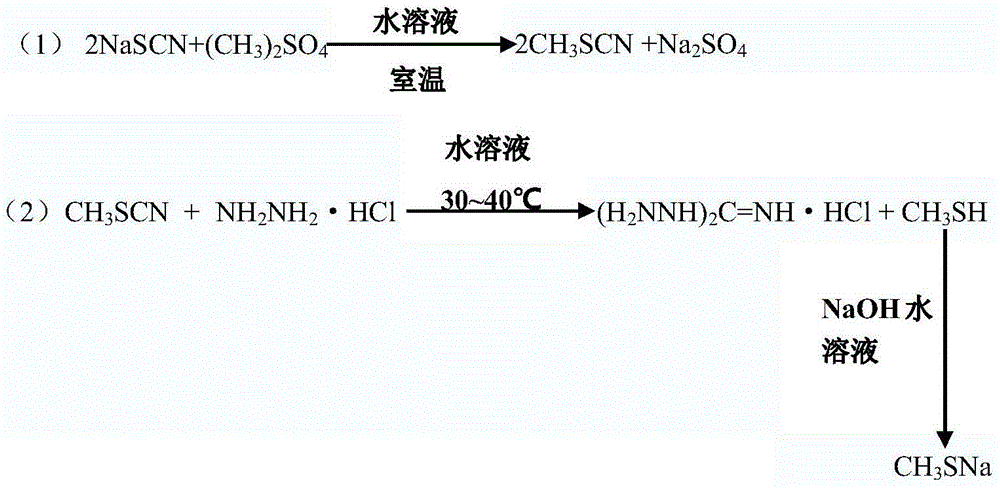
[0016] Embodiment 1: drop into sodium thiocyanate (1Kg) and aqueous solution (1Kg) in reaction kettle, then add dimethyl sulfate (0.780Kg), after room temperature reaction 5~6h, collect oil layer, obtain methyl thiocyanate ( 0.892Kg).
[0017] In another reaction kettle, add hydrazine hydrochloride (1.671g) and water (8Kg) to heat up to 30-40°C, then add methyl thiocyanate (0.892Kg), control the reaction temperature at 30-40°C, and react for 4-5 hours Then let it cool down. Filter, wash with cold water three times, and dry to obtain the white target product (1.435Kg, the total yield is 92.8%, and the purity by HPLC is 99.1%). The by-product methyl mercaptan was sequentially absorbed with saturated sodium hydroxide solution, 30% sodium hydroxide solution and 35% sodium bicarbonate solution to obtain sodium methyl mercaptan solution.
Embodiment 2
[0018] Example 2: Add sodium thiocyanate (10Kg) and aqueous solution (10Kg) into the reaction kettle, then add another raw material dimethyl sulfate (7.79Kg), react at room temperature for 5-6 hours, collect the oil layer to obtain thiocyanate Methyl ester (8.91Kg).
[0019] In another reaction kettle, add hydrazine hydrochloride (16.91Kg) and water (80Kg) to raise the temperature to 30-40°C, then add methyl thiocyanate (8.91Kg), use a water bath to control the reaction temperature at 30-40°C, and react 4 After ~5 hours, let it cool down. It was filtered, washed three times with cold water, and then dried to obtain the white target product (14.21Kg, the total yield was 91.7%, and the purity by HPLC was 98.3%). The by-product methyl mercaptan was sequentially absorbed with saturated sodium hydroxide solution, 30% sodium hydroxide solution and 35% sodium bicarbonate solution to obtain sodium methyl mercaptan solution.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More