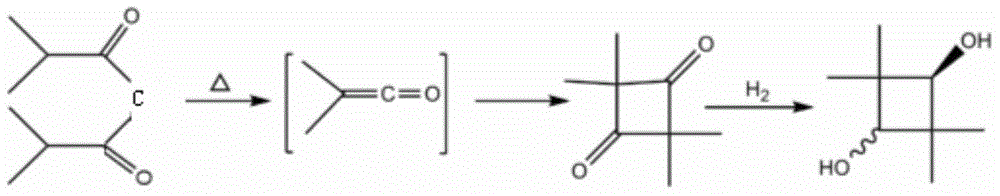
Synthetic method of 2, 2, 4, 4-tetramethyl-1, 3-cyclobutanediol
A synthesis method and technology of cyclobutanediol, applied in two fields, can solve problems such as complicated process and many side reactions, and achieve the effect of simple reaction process, simple composition and easy preparation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
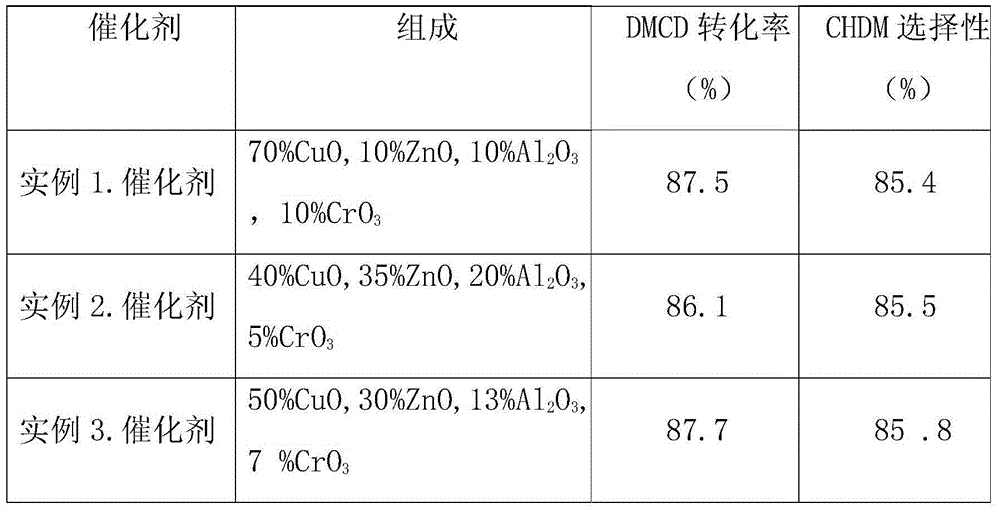
[0065] Put 212.5 grams of copper nitrate (Cu(NO 3 ) 2 ·3H 2 O), 36.5 grams of zinc nitrate (Zn(NO 3 ) 2 6H 2 O), 73.5 grams of aluminum nitrate (Al(NO 3 ) 3 9H 2 O) and 10 grams of chromic acid (CrO 3 ) was dissolved in 800 ml of distilled water. Under heating and stirring, use alkali solution (sodium hydroxide, sodium carbonate or ammonia water) to neutralize to PH = 7, and at the same time, precipitates are formed. After filtering and washing, dry at 120°C for 5 hours, bake at 400°C for 4 hours, and shape. Catalyst I was reduced in a hydrogen stream at 300°C for 4 hours before use.
Embodiment 2
[0067] Put 121.4 grams of copper chloride (CuCl 2 2H2O), 127.8 grams of zinc nitrate (Zn(NO 3 ) 2 6H 2 O), 147 grams of aluminum nitrate (Al(NO 3 ) 3 9H 2 O) and 5 grams of chromic acid (CrO 3 ) was dissolved in 800 ml of distilled water. Under heating and stirring, use alkali solution (sodium hydroxide, sodium carbonate or ammonia water) to neutralize to PH = 7, and at the same time, precipitates are formed. After filtering and washing, dry at 120°C for 5 hours, and bake at 400°C for 4 hours to shape. The catalyst code is DLW-66. Catalyst II was reduced in hydrogen flow at 300°C for 4 hours before use.
Embodiment 3
[0069] Put 151.8 grams of copper nitrate (Cu(NO 3 ) 2 ·3H 2 O), 109.5 grams of zinc nitrate (Zn(NO 3 ) 2 ·6H 2 O), 95.6 grams of aluminum nitrate (Al(NO 3 ) 3 9H 2 O) and 7 grams of chromic acid (CrO 3 ) was dissolved in 800 ml of distilled water, under heating and stirring, neutralized to PH=7 with alkaline solution (sodium hydroxide, sodium carbonate or ammonia water), and a precipitate was formed at the same time, after filtering and washing, it was dried at 120°C for 5 hours, Baking at 400°C for 4 hours to shape. Catalyst III was reduced at 300°C for 4 hours in a hydrogen stream before use.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com