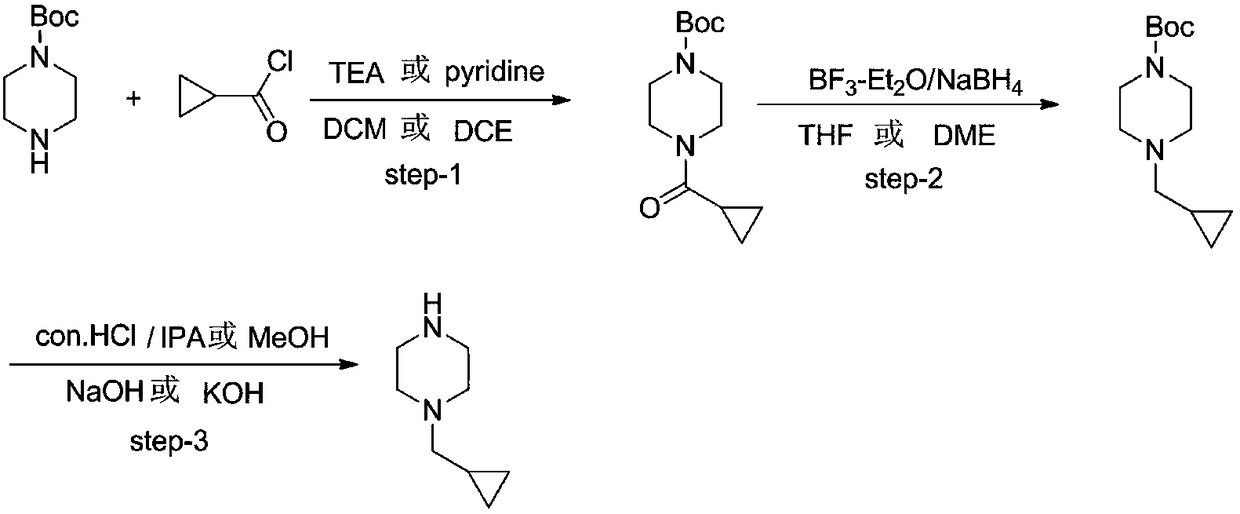
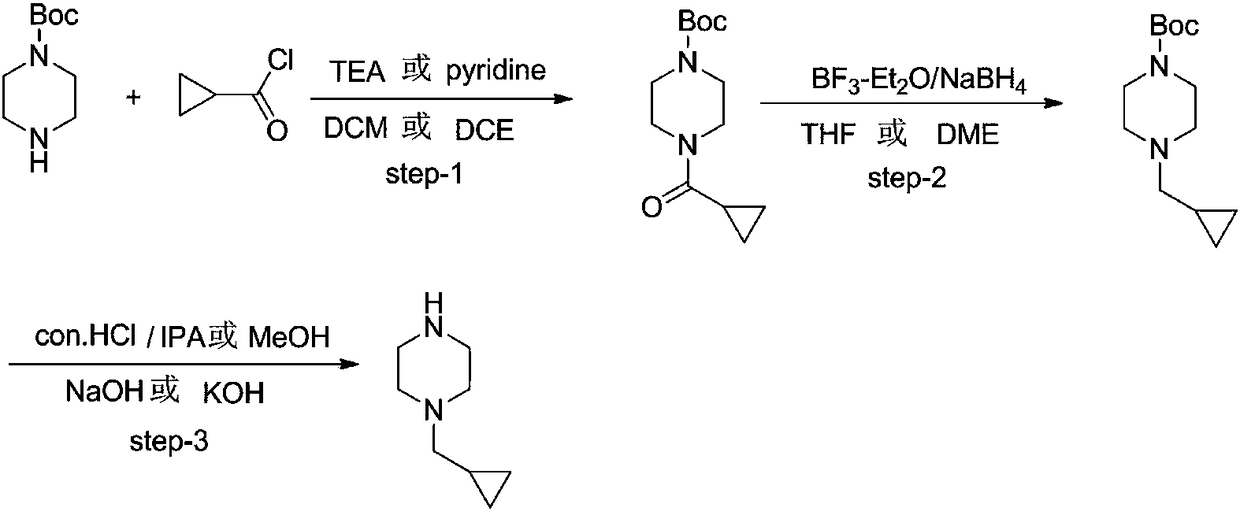
Preparation method of Volasertib intermediate 1-cyclopropyl methyl piperazine
A technology of cyclopropylmethylpiperazine and propylmethyl, which is applied in the field of drug synthesis, can solve the problems of low molecular molar utilization, poor activity, high cost, etc., and achieve the effect of simple method, convenient operation and good product quality
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0027] A preparation method of Volasertib intermediate 1-cyclopropylmethylpiperazine, comprising the following steps:
[0028] S1: Add N-Boc-piperazine (1.82kg, 9.77mol), triethylamine (1.48kg, 14.66mol), dichloromethane 5.46Kg in a 20L four-necked flask with mechanical stirring and a thermometer, and cool to At 0°C, cyclopropylformyl chloride (1.12kg, 10.75mol) was slowly added dropwise, and the temperature was controlled at 0°C-10°C. After the dropwise addition, the reaction was carried out at 10°C-20°C for 3 hours. Add 5kg of water, add sodium carbonate to adjust the pH=8-9, separate the liquid, collect the organic phase, add 1.50Kg to the water phase, extract once with dichloromethane, combine the dichloromethane phase, wash once with 2kg of 0.05M dilute hydrochloric acid, and then use Wash once with 2 kg of water, concentrate to remove dichloromethane to obtain 2.43 kg of tert-butyl 4-(cyclopropanecarbonyl)piperazine-1-carboxylate, yield 98%. The NMR is consistent with t...
Embodiment 2
[0032] A preparation method of Volasertib intermediate 1-cyclopropylmethylpiperazine, comprising the following steps:
[0033] S1: Add N-Boc-piperazine (1.82kg, 9.77mol), pyridine (1.16kg, 14.66mol), and 5.46Kg of dichloromethane into a 20L four-necked flask with mechanical stirring and a thermometer, and cool to 0°C. Cyclopropylformyl chloride (1.12 kg, 10.75 mol) was slowly added dropwise, and the temperature was controlled at 0°C-10°C. After the dropwise addition, the reaction was carried out at 10°C-20°C for 3 hours. Add 5kg of water, add sodium carbonate to adjust the pH=8-9, separate the liquid, collect the organic phase, add 1.50Kg to the water phase, extract once with dichloromethane, combine the dichloromethane phase, wash once with 2kg of 0.05M dilute hydrochloric acid, and then use Wash once with 2 kg of water, concentrate to remove dichloromethane to obtain 2.40 kg of tert-butyl 4-(cyclopropanecarbonyl)piperazine-1-carboxylate, yield 96.8%. The NMR is consistent w...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More