Relative quantifying kit used for detecting copy number of survival genes of human motor neurons
A motor neuron and gene copy number technology, applied in the determination/testing of microorganisms, biochemical equipment and methods, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0068] 1. Nucleic acid extraction:
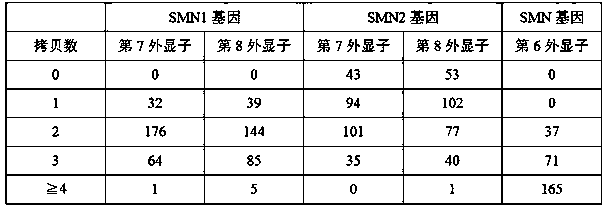
[0069] Twenty patients with a clinical diagnosis of SMA were included, as well as whole blood samples from 195 pregnant women. Use the nucleic acid extraction reagent produced by Dobtech Biotechnology (Xiamen) Co., Ltd. (record number: Minxia Machinery Equipment No. 20160080) to extract the whole blood sample collected by the EDTA anticoagulation tube, and use a micro-ultraviolet spectrophotometer to determine the nucleic acid after extraction Purity and concentration, the OD260 / OD280 should be between 1.6 and 2.0; dilute the genomic DNA concentration with sterilized double distilled water to 2ng / µL for later use.
[0070] 2. Serial dilution of the reference substance
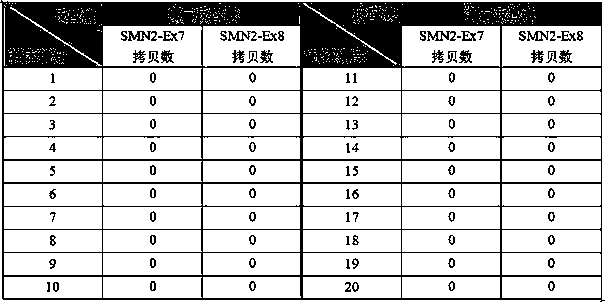
[0071]Take 10 µL of the first control, the second control, and the third control, respectively, and add them to 3 centrifuge tubes, then add 40 µL 1×TE solution, and mix well to obtain 3 kinds of 5-fold diluted reference substances; For the reference substance, take out 10...
Embodiment 2
[0089] 1. Reagent specificity verification: cross-reaction of common clinical pathogens
[0090] 1. Experimental samples
[0091] Take 13 specific samples to verify the specificity of the reagent, hepatitis B virus, hepatitis C virus, herpes simplex virus type 1, herpes simplex virus type 2, Epstein-Barr virus, human cytomegalovirus, human herpes virus type 6A, Human herpesvirus type 6B, human parvovirus B19, varicella-zoster virus type B, adenovirus type 2, JC polyomavirus, BK polyomavirus.
[0092] 2. Experimental process
[0093] Use the first reaction solution, the second reaction solution and the third reaction solution to detect the above 13 specific samples respectively, analyze the test results, and verify the specificity of the reagents.
[0094] 3. Experimental results
[0095] The 13 specific samples tested by the three reaction solutions were all Undetermined, indicating good specificity and no cross-reaction. The specific results are shown in the table below: ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More