Non-metal bcn/g-c 3 n 4 Van der Waals heterojunction photocatalyst and preparation method and application thereof
A photocatalyst, g-c3n4 technology, applied in the direction of physical/chemical process catalysts, non-metallic elements, chemical instruments and methods, etc., can solve the problem of carbon nitride being difficult to be widely used, high carrier recombination efficiency, and low photocatalytic activity and other issues, to achieve the effect of facilitating large-scale production, improving the electron transmission rate, and low cost
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
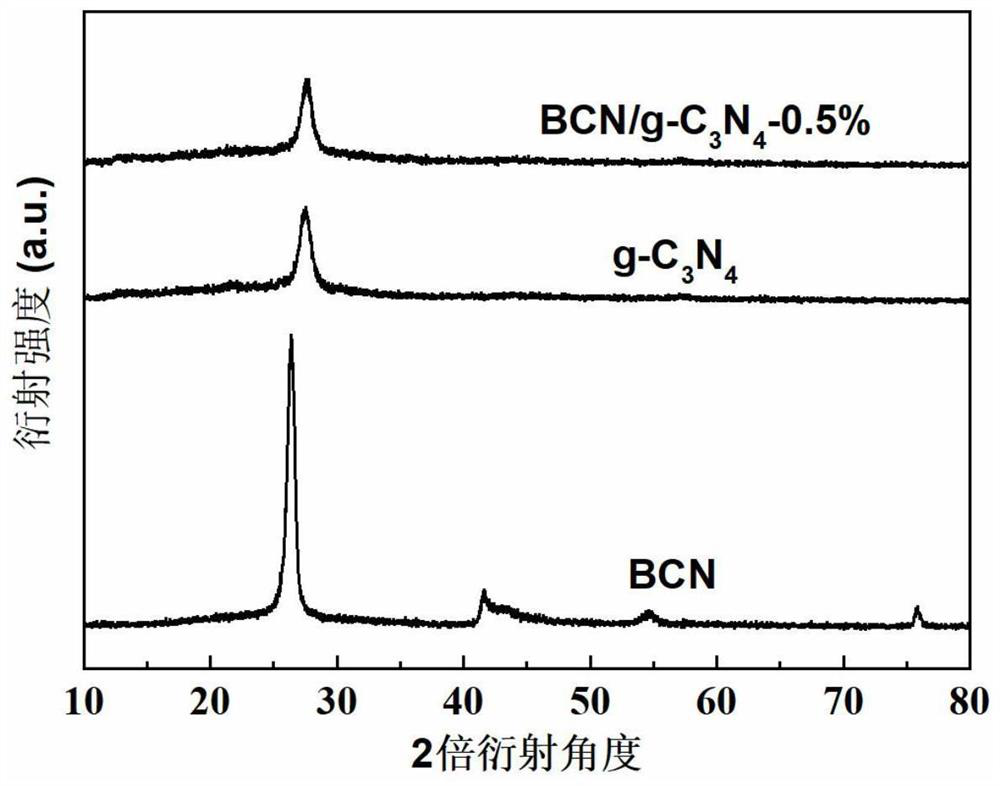
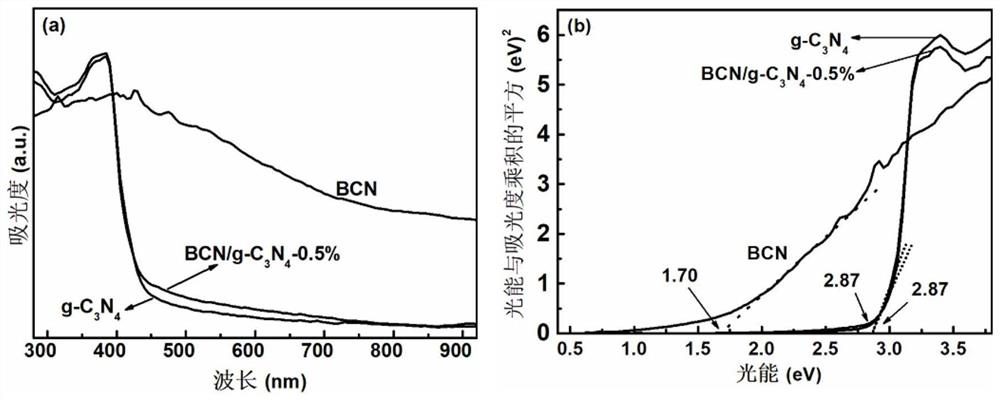
[0032] Example 1: Preparation of BCN / g-C 3 N 4 -0.1% van der Waals heterojunction photocatalyst and its photocatalytic hydrogen production
[0033] (1) Preparation of g-C 3 N 4
[0034] Weigh 100 g of urea, put it in a drying oven at 80 °C for 12 hours, take it out, grind it to powder, put it into a crucible, cover it, and put it in a muffle furnace for calcination at 550 °C for 4 hours, with a heating rate of 2.5 °C / min. After natural cooling, the calcined product was taken out and put into 300 mL of 1.5 M nitric acid solution, and stirred for 12 h. Then carry out washing and suction filtration, wash with deionized water until the pH value of the filtrate is the same as that of deionized water, and finally put it into a drying oven for drying at 80°C.
[0035] (2) Preparation of BCN nanosheets
[0036] Weigh 2g of boron oxide, 4g of urea, 0.6g of glucose into a quartz mortar and finely grind, mix well, then put the mixture into a porcelain boat, cover it, put it into a ...
Embodiment 2
[0041] Example 2: BCN / g-C 3 N 4 Preparation of -0.3% van der Waals heterojunction photocatalyst and photocatalytic hydrogen production
[0042] Steps (1)-(2) are the same as in Example 2.
[0043] (3) Preparation of BCN / g-C 3 N 4 -0.3% van der Waals heterojunction photocatalyst
[0044] Weigh 1 g of g-C prepared in step (1) 3 N 4 With 0.003g of BCN nanosheets, finely ground in a quartz mortar, transferred to a porcelain boat, put the porcelain boat into a tube furnace, and heated in N 2 Heated to 500 °C at a heating rate of 5 °C / min under gas protection, and calcined for 4 h to obtain BCN / g-C 3 N 4 -0.3% photocatalyst.
[0045] (4) BCN / g-C 3 N 4 -0.3% van der Waals heterojunction photocatalyst for photocatalytic hydrogen production
[0046] The photocatalytic reaction was carried out in a closed reaction system with a total volume of about 250 mL, and 50 mg BCN / g-C 3 N 4 -0.3% catalyst was uniformly dispersed in 100mL of 20vol% TEOA aqueous solution pH 11.4, then...
Embodiment 3
[0047] Example 3: BCN / g-C 3 N 4 Preparation of -0.5% van der Waals heterojunction photocatalyst and photocatalytic hydrogen production
[0048] Steps (1)-(2) are the same as in Example 2.
[0049] (3) Preparation of BCN / g-C 3 N 4 -0.5% van der Waals heterojunction photocatalyst
[0050] Weigh 1 g of g-C prepared in step (1) 3 N 4 With 0.005g of BCN nanosheets, finely ground in a quartz mortar, transfer to a porcelain boat, put the porcelain boat into a tube furnace, in N 2 Heated to 500 °C at a heating rate of 5 °C / min under gas protection, and calcined for 4 h to obtain BCN / g-C 3 N 4 -0.5% photocatalyst.
[0051] (4) BCN / g-C 3 N 4 -0.5% van der Waals heterojunction photocatalyst for photocatalytic hydrogen production
[0052] The photocatalytic reaction was carried out in a closed reaction system with a total volume of about 250 mL, and 50 mg BCN / g-C 3 N 4 -0.5% catalyst was uniformly dispersed in 100mL of 20vol% TEOA aqueous solution pH 11.4, then 3% H was adde...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More