Method for determining content of piperazine ferulate
A technology of piperazine ferulic acid and a detection method, which is applied in the direction of measuring devices, instruments, scientific instruments, etc., can solve the problem of not examining the separation degree of starting material vanillin and ferulic acid, the inability to ensure the separation degree of the main peak and impurities, Issues such as specificity need to be considered to achieve the effect of good repeatability and accuracy, low instrument requirements, and controllable quality
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
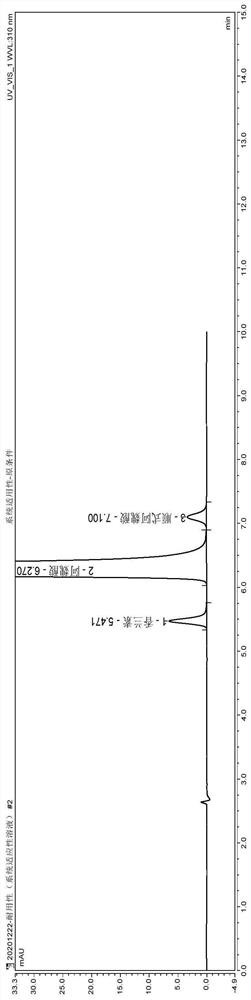
[0047] (1) Instrument and chromatographic conditions
[0048] High performance liquid chromatography: U3000 high performance liquid chromatography system and workstation;
[0049] Chromatographic column: Thermo Acclaim (4.6mm×250mm, 5μm) octadecylsilane bonded silica gel column;
[0050] Configure methanol-water-acetic acid (20:80:0.1) as mobile phase A, methanol-water-acetic acid (80:20:0.1) as mobile phase B, phase A-phase B (55:45), isocratic elution , flow rate 1.0ml / min; column temperature 35°C, detection wavelength 310nm;
[0051] (2) Experimental steps
[0052] Avoid light operation.
[0053] Take the appropriate amount of piperazine ferulate reference substance, impurity A, and impurity F reference substance respectively, and use solvent [water-methanol-acetic acid (60:40:0.1)] to make each 1ml of piperazine ferulate 0.1mg, other System suitability solution containing 1 μg of each impurity;
[0054]Take an appropriate amount of sample, add diluent [methanol-water-...
Embodiment 2
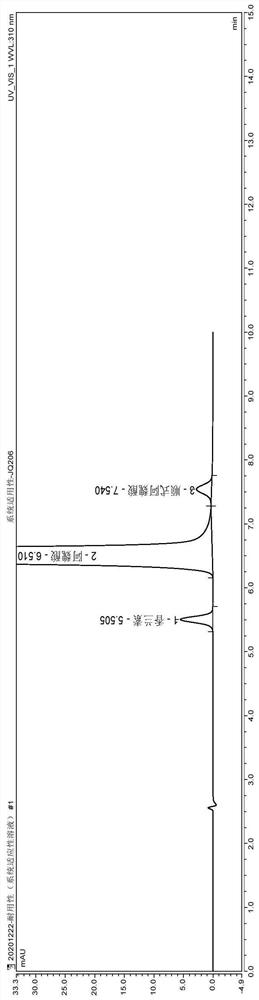
[0060] (1) Instrument and chromatographic conditions
[0061] High performance liquid chromatography: U3000 high performance liquid chromatography system and workstation;
[0062] Chromatographic column: Luna C18 (4.6mm×250mm, 5μm) octadecylsilane bonded silica gel column;
[0063] Configure 0.01M sodium acetate-methanol (80:20) as phase A, 0.01M sodium acetate-methanol (20:80) as phase B, phase A-phase B (55:45), isocratic elution, flow rate 1.0 ml / min; column temperature 35°C, detection wavelength 310nm;
[0064] (2) Experimental steps
[0065] Avoid light operation.
[0066] Take the appropriate amount of piperazine ferulate reference substance, impurity A, and impurity F reference substance respectively, and use solvent [0.01M sodium acetate-methanol (60:40)] to make each 1ml containing piperazine ferulate 0.1mg, other various System suitability solution containing 1 μg of each impurity;
[0067] Take an appropriate amount of sample, add diluent [methanol-water-acetic...
Embodiment 3
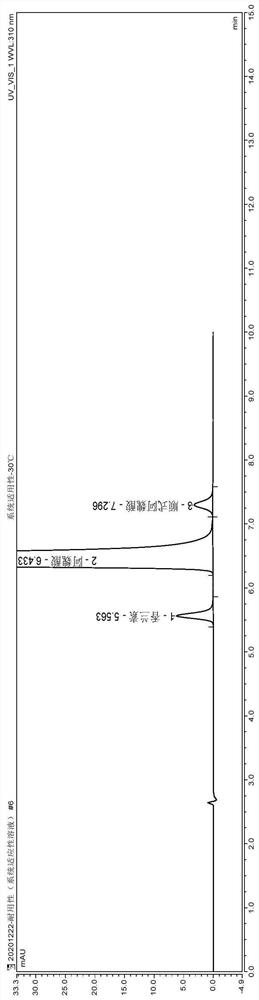
[0073] (1) Instrument and chromatographic conditions
[0074] High performance liquid chromatography: U3000 high performance liquid chromatography system and workstation;
[0075] Chromatographic column: Thermo Acclaim (4.6mm×250mm, 5μm) octadecylsilane bonded silica gel column;
[0076] Configure methanol-water-acetic acid (20:80:0.1) as mobile phase A, methanol-water-acetic acid (80:20:0.1) as mobile phase B, phase A-phase B (55:45), isocratic elution , flow rate 1.0ml / min; column temperature 30°C, detection wavelength 310nm;
[0077] (2) Experimental steps
[0078] Avoid light operation.
[0079] Take the appropriate amount of piperazine ferulate reference substance, impurity A, and impurity F reference substance respectively, and use solvent [water-methanol-acetic acid (60:40:0.1)] to make each 1ml of piperazine ferulate 0.1mg, other System suitability solution containing 1 μg of each impurity;
[0080] Take an appropriate amount of sample, add diluent [methanol-water...
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
| length | aaaaa | aaaaa |
| diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More