PCR (Polymerase Chain Reaction) detection kit for cat and/or dog pathogens, detection method and application
A technology for detecting kits and pathogens, applied in the field of biomedicine, can solve the problems of low detection accuracy and low positive coincidence rate of pet pathogens, and achieve the effects of saving detection time, strong specificity, and saving human resources
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0141] Reagent test kit
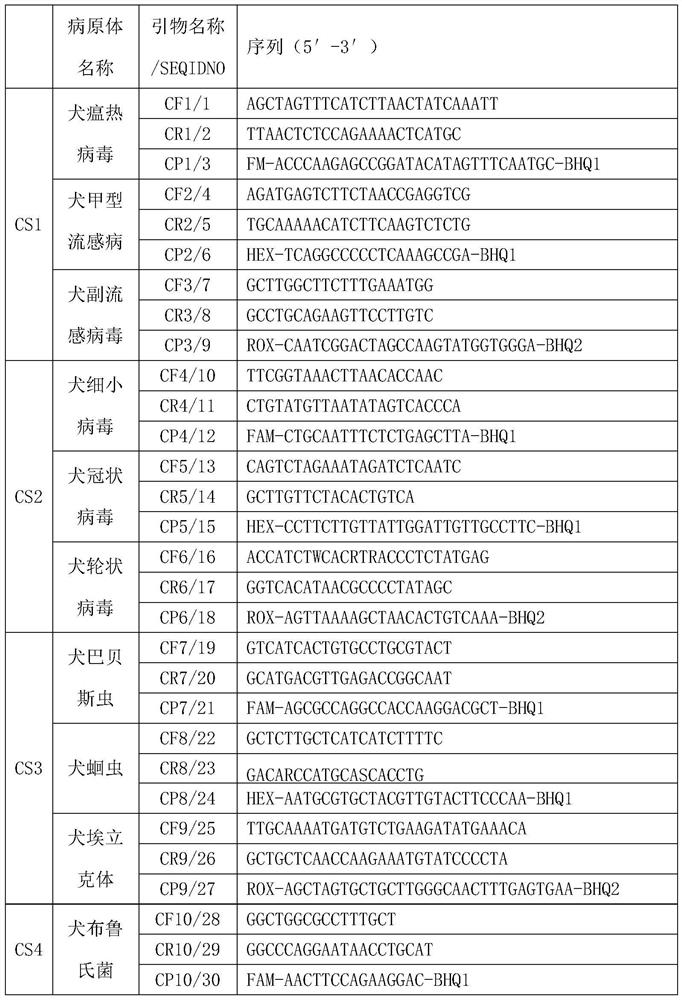
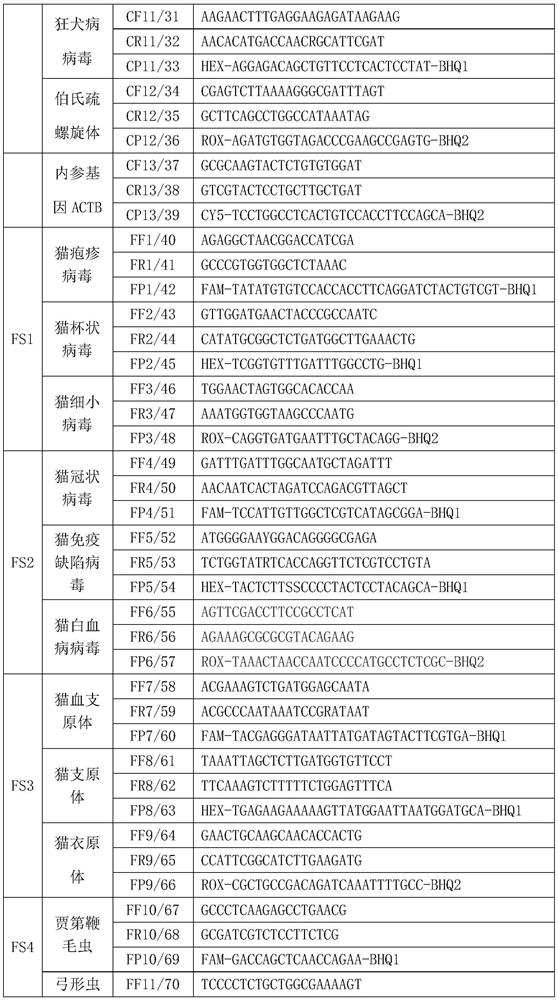
[0142] 1. Specific primers and probes, see the table below
[0143]
[0144]
[0145]
[0146] 2. The PCR reaction reagent is: TaqPath TM 1-Step Multiplex Master Mix (No ROX).
[0147] 3. Deionized water.
Embodiment 2
[0150] Collect 1ml of peripheral venous blood from cats and dogs according to routine medical procedures, and add EDTA for anticoagulation. Using Thermo's MagMAX TM CORE Nucleic Acid Purification Kit+MagMAX TM CORE Mastitis&PanbacteriaModule Extraction Kit, extract DNA and RNA.
Embodiment 3
[0152] PCR reaction system preparation
[0153] Prepare the PCR reaction system according to the following system:
[0154] A total of 8 reaction systems (the total reaction system is 20 μL), which are CS1-CS4 and FS1-FS4 in Table 1 and Table 2, respectively, were composed of mixed internal reference primers and probes.
[0155] PCR reaction reagent: 10 μL;
[0156] The concentration of each primer probe is 10M, the primer volume is 0.8 μL, and the probe volume is 0.4 μL;
[0157] Sample template: 2 μL;
[0158] Make up 20 μL with deionized water.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More