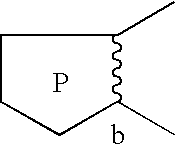
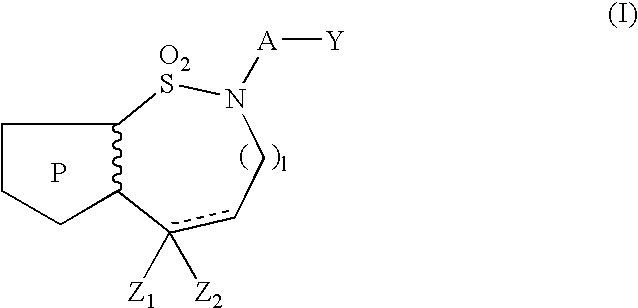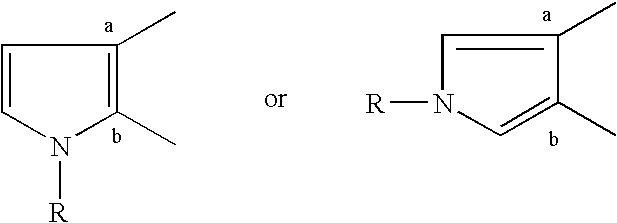Pyrrolothiazine and pyrrolothiazephine compounds having serotonin-2 receptor antagonistic and alpha-1-blocking action
a technology of serotonin-2 receptor and pyrrolothiazephine, which is applied in the field of pyrrolesulfonamide derivatives, can solve the problems of many problems accompanied by the antagonistic action of the serotonin-2 receptor, and achieve the effects of strong serotonin-2 receptor antagonistic action, low toxicity and less side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
referential example 1
[0135] Synthesis of sodium 3-pyrrolesulfonate (Compound 1)
[0136] A mixture consisting of 30.0 g (447 mmol) of pyrrole, 75.0 g (471 mmol) of sulfur trioxide-pyridine complex and 250 ml of 1,2-dichloroethane was refluxed for 16 hours. The top layer of the reaction mixture was removed by decantation. To the residue, 150 ml of water and 30 g of sodium carbonate were added successively. After the resulting mixture was boiled, the solvent was distilled off under reduced pressure. Ethanol-water (9:1 v / v, 500 ml) was added to the residue, followed by reflux for 1 hour. The reaction mixture was subjected to hot filtration, and the filtrate was allowed to cool down. Precipitated crystals were collected, washed with chilled ethanol and diethyl ether, and then dried under reduced pressure, whereby 17.0 g of powdery crystals were obtained.
referential example 2
[0137] Synthesis of Benzyl 2-(3-pyrrolesulfonamide)acetate (Compound 2)
[0138] A suspension of 16.9 g (100 mmol) of Compound 1 and 22.9 g (110 mmol) of phosphorus pentachloride in 750 ml of diethyl ether was stirred at room temperature for 2 hours, and was then refluxed for 4 hours. After the reaction mixture was allowed to cool down, it was filtered. The filtrate was washed successively with ice water (twice), a chilled, saturated aqueous solution of sodium hydrogencarbonate, ice water and a chilled, saturated aqueous solution of sodium chloride. The organic layer was dried over anhydrous sodium sulfate and then concentrated under reduced pressure, whereby 11.2 g of 3-pyrrolesulfonyl chloride were obtained as crude crystals.
[0139] After a mixture consisting of the whole amount of the thus-obtained crude crystals, 32.6 g (96.6 mmol) of glycine benzyl ester p-toluenesulfonate, 19.6 g (193 mmol) of triethylamine and 250 ml of tetrahydrofuran (hereinafter called “THF”) was refluxed f...
referential example 3
[0140] Synthesis of Benzyl 3-(3-pyrrolesulfonamide)propionate (Compound 3)
[0141] A mixture consisting of 1.66 g (10 mmol) of 3-pyrrolesulfonyl chloride obtained by the process of Referential Example 2, 7.03 g (20 mmol) of β-alanine benzyl ester p-toluenesulfonate, 4.05 g (40 mmol) of triethylamine and 100 ml of THF was refluxed for 16 hours. The reaction mixture was concentrated under reduced pressure, and ethyl acetate was added to the residue. The organic layer was washed successively with a saturated aqueous solution of sodium hydrogencarbonate, water, a 10% aqueous solution of citric acid, water and a saturated aqueous solution of sodium chloride, dried over anhydrous sodium sulfate, and then concentrated under reduced pressure. The residue was purified by column chromatography on silica gel (Merck & Co. Inc. No. 9385) (the same silica gel were used in the subsequent examples) (eluent: ethyl acetate / hexane=1 / 1), whereby 2.82 g of the title compound were obtained-(yield: 92%)....
PUM
| Property | Measurement | Unit |
|---|---|---|
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More