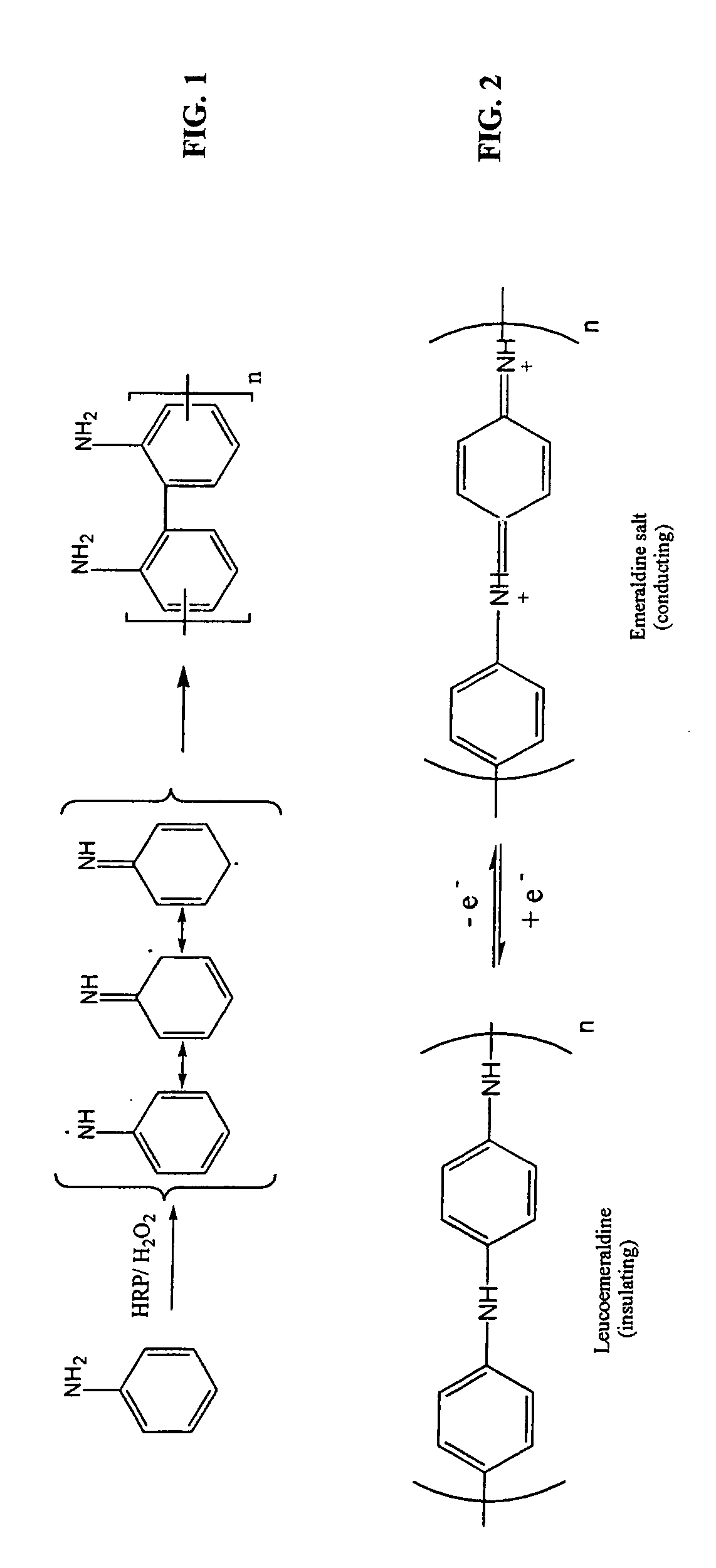
Enzymatic template polymerization
a template and polymerization technology, applied in the direction of non-metal conductors, conductors, organic conductors, etc., can solve the problems of limited application possibilities of polymers, achieve stable desired electronic and optical properties, reduce parasitic branching, and improve the electrical and optical properties of the final polymer complex
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0068] A. Materials and Methods
[0069] Horseradish peroxidase (HRP) (enzyme classification number (EC) 1.11.1.7), phosphate and Tris-HCl buffers were obtained from Sigma Chemicals Company, St. Louis, Mo. Aniline, sulfonated polystyrene (SPS) and hydrogen peroxide (30%) were obtained from Aldrich Chemicals, Inc., Milwaukee, Wis. All the chemicals were used as received.
[0070] B. Results and Discussion
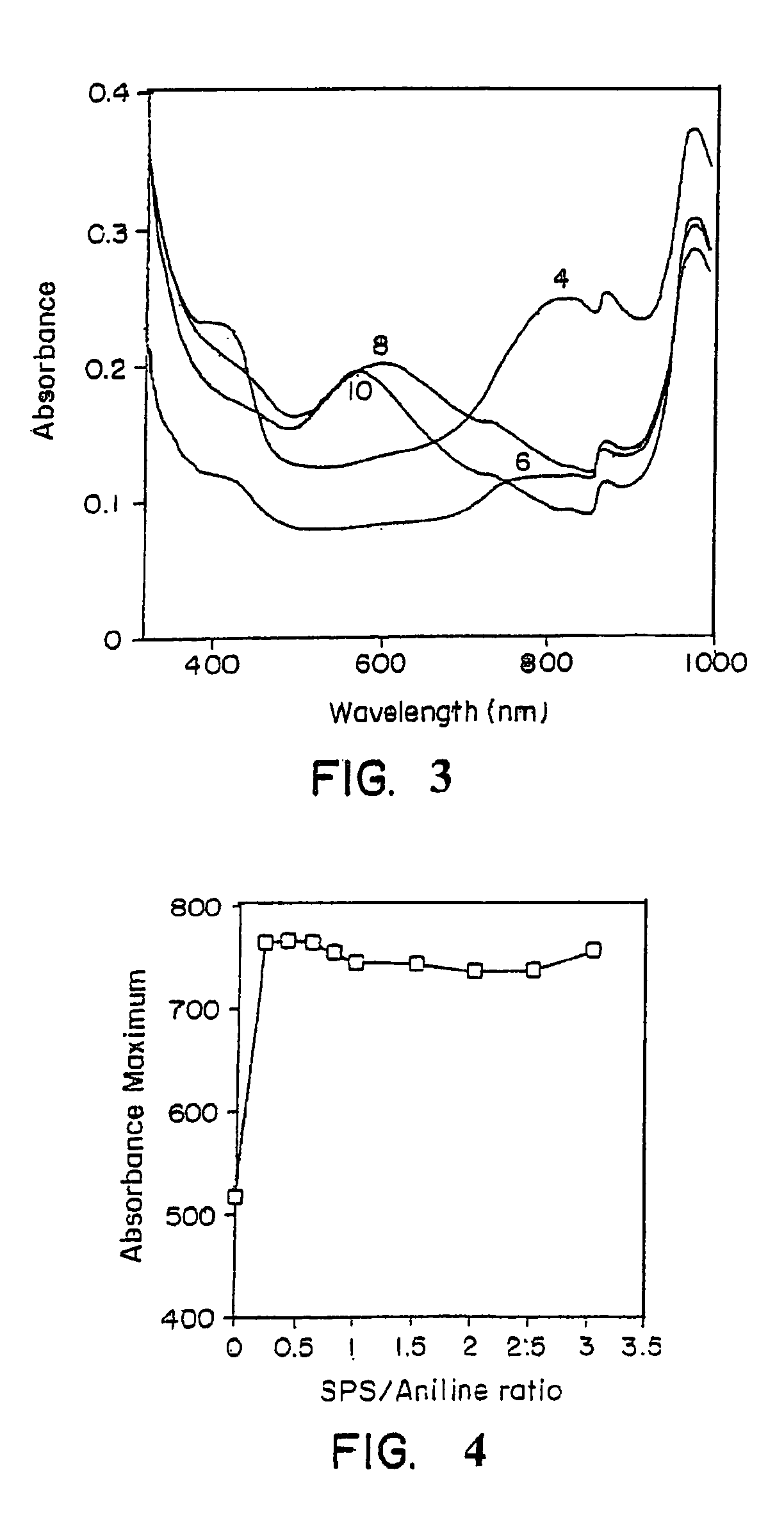
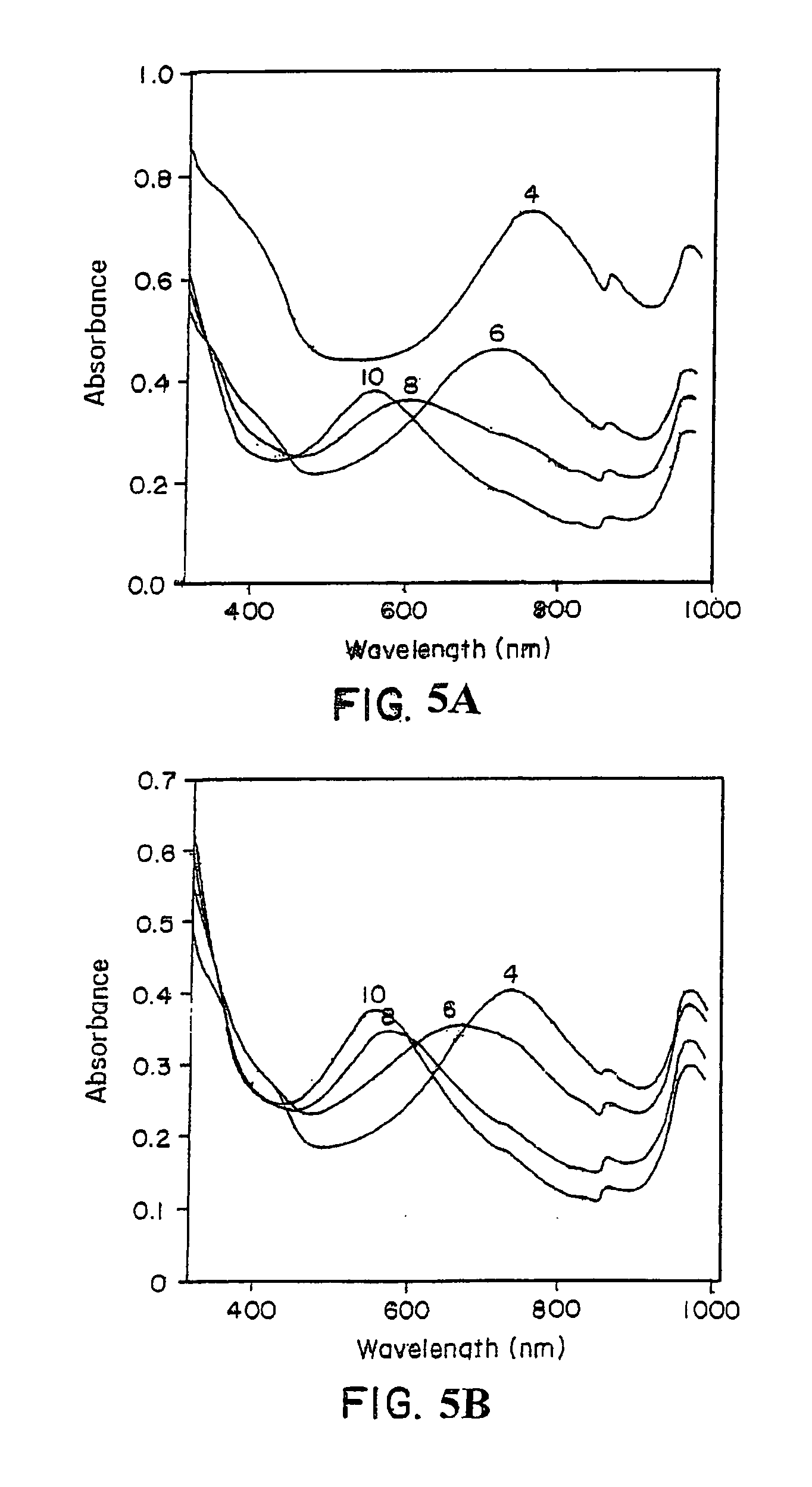
[0071] The progress of a template-assisted polymerization reaction of aniline in the presence of the polyelectrolyte, sulfonated polystyrene (SPS) in a 1:1 ratio, was monitored by the change in visible absorbance. A Perkin-Elmer Lambda-9® UV-Vis-near IR spectrophotometer was used for the spectral characterization of the polymer. FIG. 3 shows the visible absorption spectra of the sulfonated polystyrene / polyaniline (SPS / PA) complex prepared under various pH conditions of 4, 6, 8, and 10. As shown in FIG. 3, SPS / PA, prepared at a pH of 4, exhibited a strong absorbance maximum at approximat...
example 2
[0077] A. Materials and Methods
[0078] Horseradish peroxidase (HRP) (enzyme classification number (EC) 1.11.1.7), phosphate and Tris-HCl buffers were obtained from Sigma Chemicals Company, St. Louis, Mo. Phenol, sulfonated polystyrene (SPS) and hydrogen peroxide (30%) were obtained from Aldrich Chemicals, Inc., Milwaukee, Wis. All the chemicals were used as received.
[0079] B. Results and Discussion
[0080] The progress of a template-assisted polymerization reaction of phenol in the presence of the polyelectrolyte, sulfonated polystyrene (SPS) in a 1:1 ratio, was monitored by the change in visible absorbance. Perkin-Elmer Lambda-9® UV-Vis-near IR spectrophotometer was used for the spectral characterization of the polymer. FIG. 7A shows the visible absorption of polyphenol without SPS, versus phenol monomer. As shown, there was a significant absorption maximum in the visible spectrum upon polymerization, indicating formation of polyphenol. However, with time the polymer began to preci...
example 3
[0082] A. Preparation of DNA-Polyaniline Complex
[0083] Horseradish peroxidase (HRP, EC 1.11.1.7) type II, (150-200 units / mg) solid was purchased from Sigma Chemical Co. (St. Louis, Mo.). Calf Thymus DNA was purchased from Worthington Biochemical Corporation (Freehold, N.J.). Aniline monomer (purity 99.5%) and hydrogen peroxide (30% by weight) were purchased from Aldrich Chemicals, Inc., Milwaukee, Wis., and were used as received. All other chemicals were of reagent grade or better. All glassware and plasticware were sterilized by autoclaving for 30 minutes.
[0084] A stock solution of calf Thymus DNA (19.9 mg) was dissolved in 40 ml of sterile, 10 mM sodium citrate buffer maintained at pH 4. The solution was stored in the refrigerator for 48 hours before reaction. The concentration of DNA was determined by the UV absorbance at 258 nm. The molar extinction coefficient (ε) at 258 nm was taken as 6420 1 mol cm−1, as reported by Sprecher, et al., Biopolymers (1979), 18:1009. The reactio...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Molecular weight | aaaaa | aaaaa |
| Electrical conductor | aaaaa | aaaaa |
| Optical properties | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More