Cefathiamidine hydrate, preparation method thereof and application thereof
A technology of cefathiamidine and hydrate, which is applied in the field of medicine to achieve the effects of low toxicity, good dissolution performance, and convenient storage and transportation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0037] Example 1 Preparation of Cefathiamidine 0.25 Hydrate Suspend 8 g of 7-bromoacetyl ACA in 160 ml of dichloromethane, add 3 ml of triethylamine, stir to dissolve completely, add 0.2 g of activated carbon and stir for 30 minutes, filter, filter Wash the cake with 10ml of dichloromethane, combine the filtrate and washing liquid, add 3.8g of N,N'-diisopropylthiourea, stir, react at 35-40°C for 1.5 hours, cool to below 15°C, and slowly add Acetone 200ml, chloroform 20ml, stand for 3 hours, filter with suction, soak the crystals with a small amount of chloroform and acetone for 3 times, filter with suction, dissolve the obtained solid in 20ml of water, slowly add 120ml of acetone, 20ml of acetonitrile, place below 10°C, After the precipitate is fully separated, it is dried by suction filtration, and the solid is dried at about 40°C for about 5 hours to obtain 3.3 g of off-white crystals, melting point: 155°C decomposition (ELECTROTHERMALMELTING POINT APPARATUS, uncorrected), HP...
Embodiment 2
[0038] Example 2 Example 1 Preparation of Cefathiamidine 0.5 Hydrate Suspend 8 g of 7-bromoacetyl ACA in 180 ml of dichloromethane, add 3.2 ml of diisopropylamine, stir to dissolve completely, add 0.2 g of activated carbon and stir for 25 minutes , filtered, the filter cake was washed with 10ml of chloroform, the combined filtrate and washing liquid were added with 3.8g of N,N'-diisopropylthiourea, stirred, reacted at 35-40°C for 1.5 hours, cooled to below 10°C, Slowly add 200ml of acetone and 20ml of ethyl acetate, let stand for 3 hours, filter with suction, soak the crystals with a small amount of acetone and acetonitrile for 3 times, filter with suction, dissolve the obtained solid in 20ml of water, slowly add 120ml of acetone, 20ml of ethanol, iso Propyl ether 10ml, place below 10°C, wait until the precipitate is fully separated, filter to dryness, and dry the solid at about 35°C for about 5 hours to obtain 3.0g off-white crystals, melting point: 155°C decomposition (ELECTR...
Embodiment 3
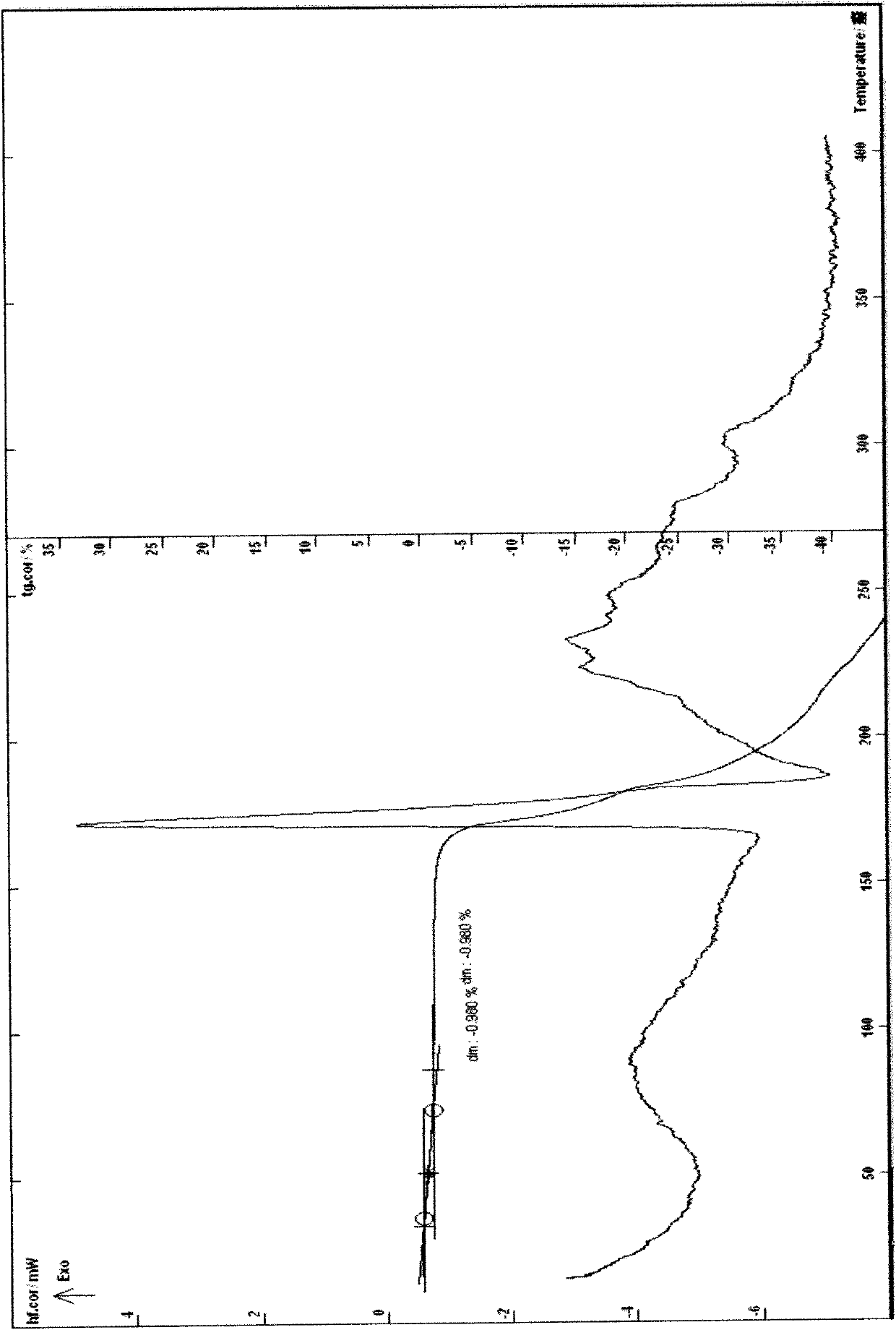
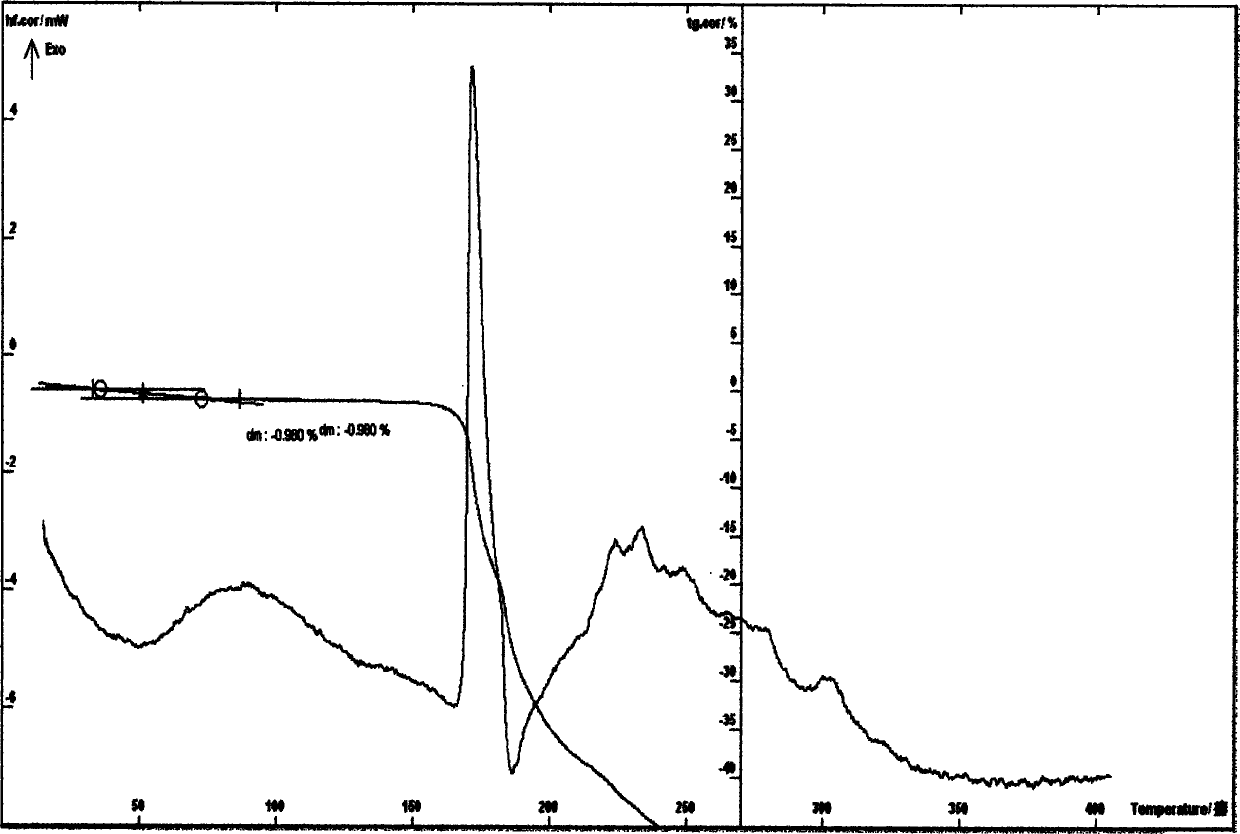
[0040] Example 3 Put 5 g of cefathiamidine in a container and add 10 ml of water to dissolve it, put the solution in a freeze-drying box, lower the temperature of the liquid to -40°C, and pre-freeze for 4 hours; lower the temperature of the condenser to -45°C, Vacuumize, control the degree of vacuum and control the temperature of the plate layer to rise below -15°C, freeze and sublimate, and sublimate and dry for about 17 hours; continue heating to gradually increase the temperature to about 30°C at a rate of 10°C per hour, and keep warm for about 3 hours. Close the hydrazine butterfly valve of the box for 3 minutes, and when the pressure of the box rises to within 5Pa, the entire freeze-drying process is ended, and the gas is released from the box to obtain cefathiamidine 0.5 hydrate; [α] D 20 : +140; Karl Fischer's method of measuring moisture is 1.96%, thermal analysis: platform weight loss of about 1.80%, which is within the error range with the result of the sample contai...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More