Application of juglone to preparation of medicament for treating autoimmune and inflammatory diseases
An autoimmune and juglone technology, applied in allergic diseases, blood diseases, sensory diseases, etc., can solve problems such as antigen signal stimulation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0021] 1.1 Construction method of EAE mouse model
[0022] (1) Experimental animals and sources
[0023] Thirty female wild-type C57BL / 6 pathogen-free (SPF grade) mice, 6-8 weeks old, weighing 18-20g, were purchased from Beijing Weitong Lihua Experimental Animal Technology Co., Ltd. and bred in the Experimental Animal Center of Tianjin Medical University , The mouse feeding environment is room temperature 20-25 ℃, relative humidity 40%-60%.
[0024] (2) Preparation of Antigen Adjuvant Emulsion
[0025] 100 μg of MOG35-55 polypeptide and 500 μg of inactivated Mycobacterium tuberculosis (Mycobacterium tuberculosis, purchased from Difco) were completely mixed with 100 μl of normal saline and 100 μl of Freund’s adjuvant (purchased from SIGMA), and emulsified.
[0026] (3) Experimental grouping
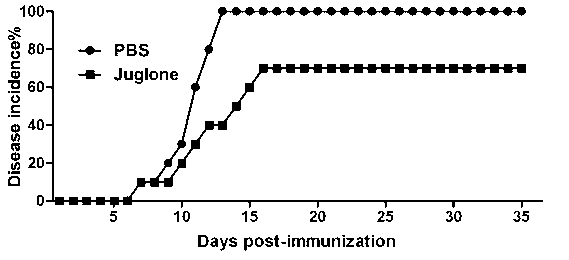
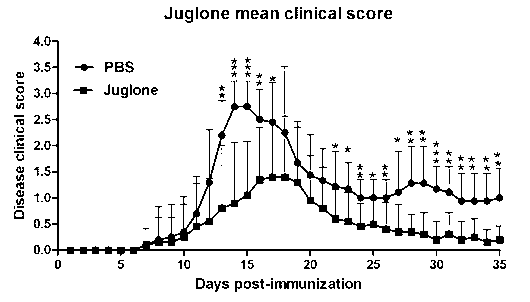
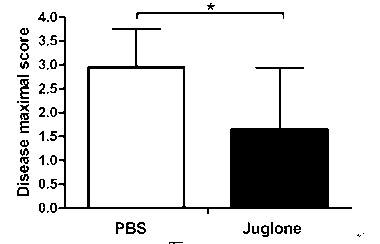
[0027] In order to study the drug effect of juglone in the mouse EAE animal model, the mice were randomly divided into four groups, with 8 mice in each group, two normal saline control ...
Embodiment 2
[0098] Mix 50g of juglone and 280g of starch evenly, make granules with starch slurry (take 220g of starch and make starch slurry with water), sieve, dry, and pack into capsules.
Embodiment 3
[0100] Mix 80g of juglone and 340g of starch evenly, make granules with starch slurry (take 210g of starch to make starch slurry with water), sieve, dry, add 6‰ magnesium stearate, mix well, press into tablets, and coat with film. .
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More