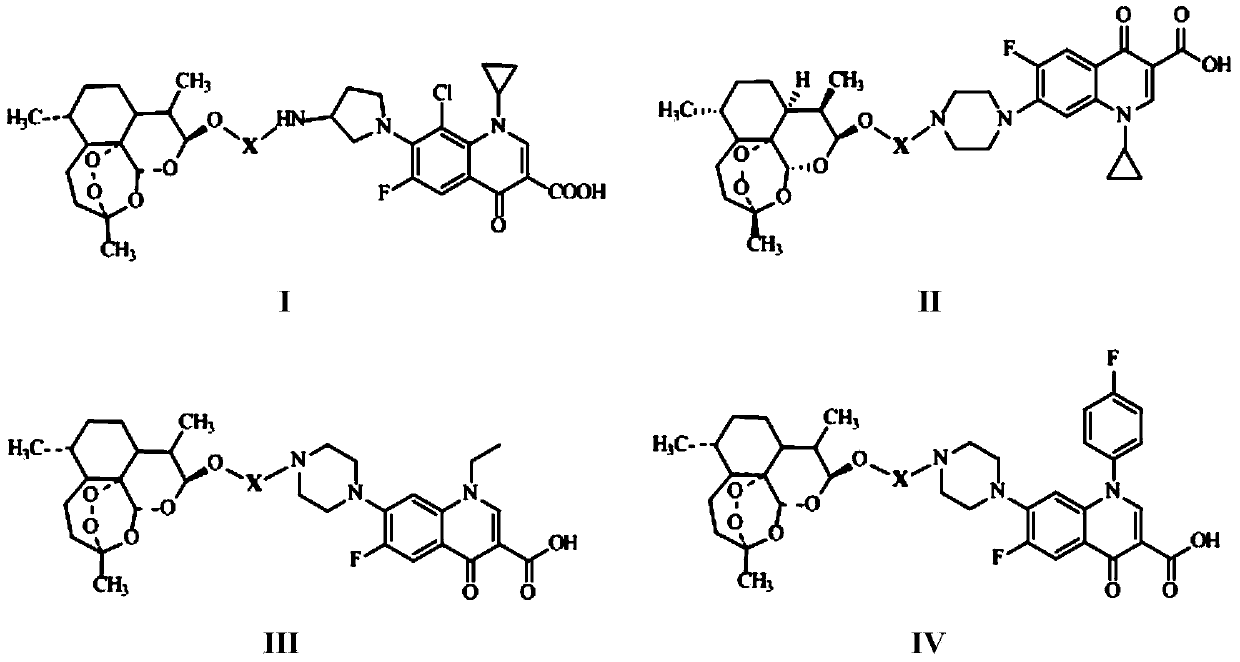
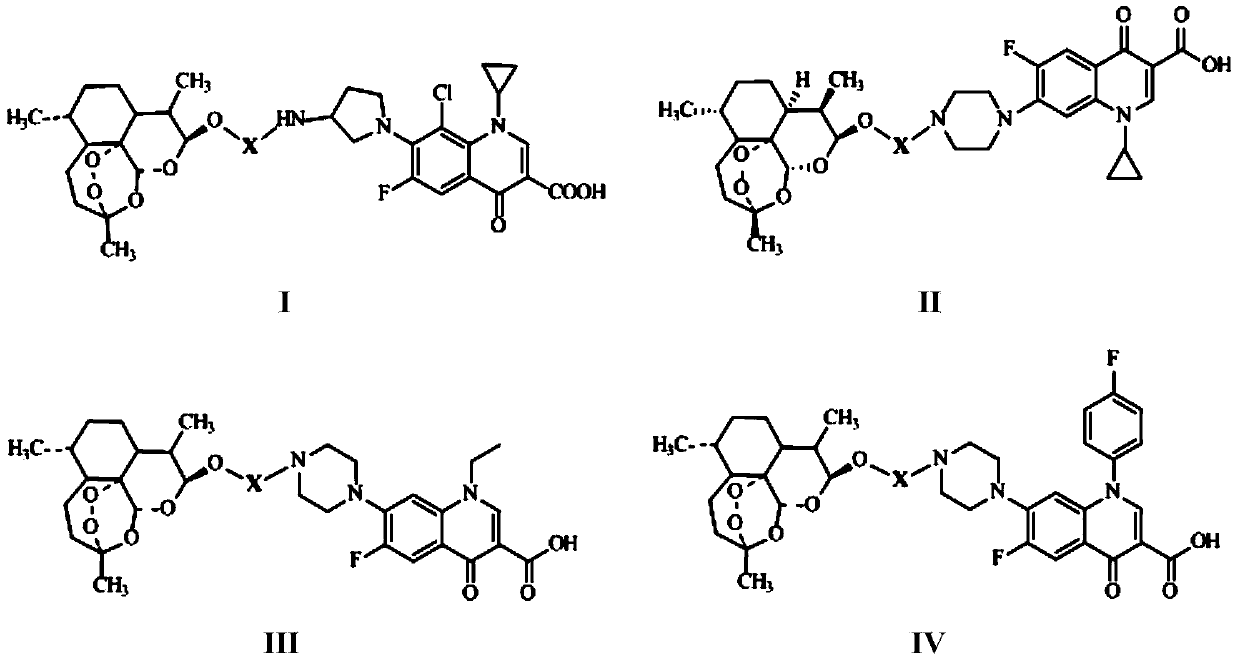
Conjugates of dihydroartemisinin and quinolones compounds as well as preparation method and application thereof
A technology of dihydroartemisinin and quinolones, which is applied in the chemical field, can solve the problems of low bioavailability and fast excretion in the body, and achieve the effect of simple preparation method
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0023] Embodiment 1, the synthesis of intermediate IM1
[0024]
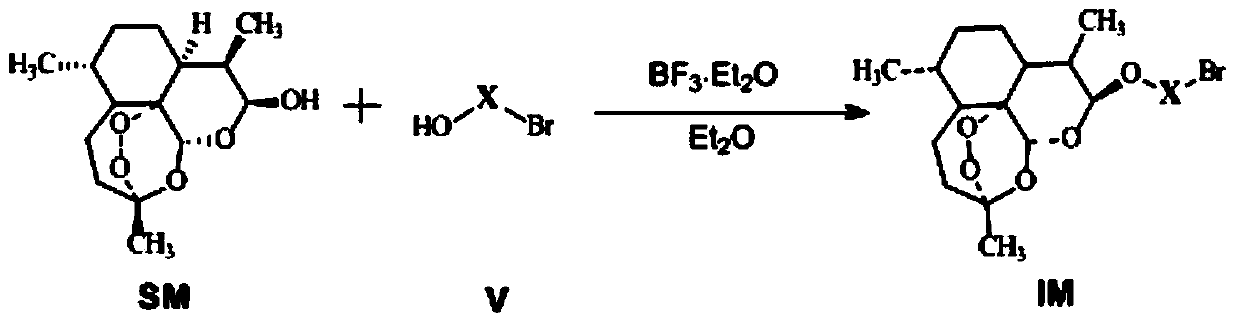
[0025] In a 250mL round bottom flask, add SM, Et 2 O and 2-bromoethanol, add BF under ice bath cooling 3 ·Et 2 O, continue cooling and stirring the reaction in an ice bath, and monitor the progress of the reaction by TLC (thin layer chromatography). After the reaction is complete, add saturated NaHCO 3 The reaction was terminated, the layers were left to stand, the aqueous layer was extracted with ethyl acetate (EtOAc), the organic phases were combined, washed with saturated brine, anhydrous MgSO 4 Drying, suction filtration, the filtrate was rotary evaporated under reduced pressure to remove the solvent to obtain a crude product, which was recrystallized with petroleum ether-EtOAc mixed solvent to obtain a pure product, namely intermediate IM1. The specific synthesis conditions and results are shown in Table 1.
[0026] IM1 12β-(2-bromoethoxy)dihydroartemisinin: white solid; m.p.:162.0-162.8℃; (c=1mg / ...
Embodiment 2
[0029] Embodiment 2, the synthesis of target compound TM1
[0030]
[0031] Add quinolones (clenfloxacin, norfloxacin, ciprofloxacin or sarafloxacin), anhydrous K 2 CO 3 and N,N-dimethylformamide (DMF), stirred in a water bath at 60°C for 30 minutes (quinolones are slightly soluble), added intermediate IM1 (IM1 quickly and completely dissolved, and the reaction system gradually became a paste), and continued at 60°C The reaction was stirred in a water bath, and the progress of the reaction was monitored by TLC. After the reaction was completed, add water and stir, a large amount of solids were precipitated, extracted twice with 20mL EtOAc, the organic phases were combined, washed with 0.5N HCl and water successively, anhydrous Na 2 SO 4 Drying, suction filtration, the filtrate was rotary evaporated to remove the solvent to obtain a yellow viscous, which was purified by column chromatography, using dichloromethane-methanol (DCM-MeOH, volume ratio 90:1) as the eluent, coll...
Embodiment 3
[0038] Embodiment 3, the synthesis of intermediate IM2
[0039]
[0040] In a 250mL round bottom flask, add SM, Et 2 O and 3-bromo-1-propanol, add BF under ice bath cooling 3 ·Et 2 0, continue cooling and stirring the reaction in an ice bath, and monitor the progress of the reaction by TLC. After the reaction is complete, add saturated NaHCO 3 The reaction was terminated, the layers were left to stand, the aqueous layer was extracted with EtOAc, the organic phases were combined, washed with saturated brine, anhydrous MgSO 4 Dry, filter with suction, remove the solvent by rotary evaporation of the filtrate under reduced pressure to obtain a yellow oily mucus, purify by column chromatography, freeze the petroleum ether refrigerator to precipitate white crystals, filter with suction, freeze the mother liquor again to precipitate crystals, filter with suction, dry in vacuum, combine the crystals, and obtain Intermediate IM2. The specific synthesis conditions and results ar...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More