Ranitidine hydrochloride combination for inhibiting generation of gastric acid
A technology of ranitidine hydrochloride and composition, which is applied in the field of drug ranitidine hydrochloride composition for inhibiting the production of gastric acid, which can solve the problems of toxic side effects and drug safety impact, and achieve good stability and low content of insoluble particles , good fluidity effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
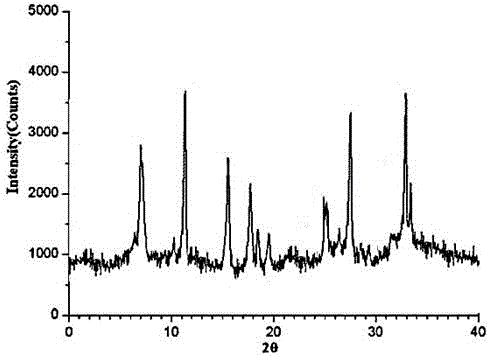
[0025] Embodiment 1: the preparation of ranitidine hydrochloride crystal
[0026] (1) Grind the crude ranitidine hydrochloride, pass through an 80-mesh sieve, then add it into ethanol whose volume is 5 times the weight of ranitidine hydrochloride, and stir at 130 rpm for 10 minutes;
[0027] (2) Add propanol with a volume 6 times the weight of ranitidine hydrochloride under stirring at 145 rpm, and raise the temperature to 40°C;
[0028] (3) After adding the solution, let it stand for 2 hours, and add dropwise a mixed solution of 2-pentanone and carbon tetrachloride whose volume is 10 times the weight of ranitidine hydrochloride at 10°C under the condition of stirring at 180 rpm , The volume ratio of 2-pentanone and carbon tetrachloride is 4:1, and the uniform dropwise addition is completed within 2 hours;
[0029] (4) After the dropwise addition was completed, the temperature was lowered to -10°C, and stirring was continued for 3 hours at a stirring rate of 85 rpm, and the c...
Embodiment 2
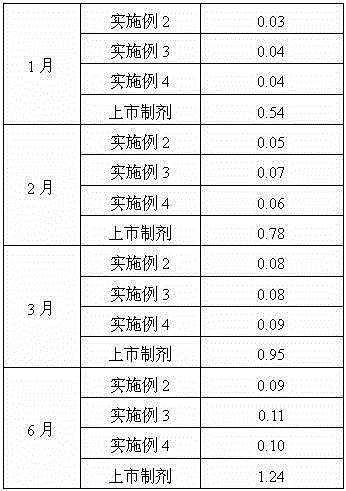
[0031] Embodiment 2: the preparation of ranitidine hydrochloride composition
[0032] The composition is: 1 part by weight of ranitidine hydrochloride crystal prepared in Example 1 of the present invention, and 0.4 part by weight of sodium chloride.
[0033] The preparation method is:
[0034] (1) Weigh ranitidine hydrochloride crystals and sodium chloride in proportion and mix them thoroughly;
[0035] (2) Dispense into sterilized vials and stopper them.
Embodiment 3
[0036] Embodiment 3: the preparation of ranitidine hydrochloride composition
[0037] The composition is: 1 part by weight of ranitidine hydrochloride crystal prepared in Example 1 of the present invention, and 0.5 part by weight of sodium chloride.
[0038] The preparation method is:
[0039] (1) Weigh ranitidine hydrochloride crystals and sodium chloride in proportion and mix them thoroughly;
[0040] (2) Dispense into sterilized vials and stopper them.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More