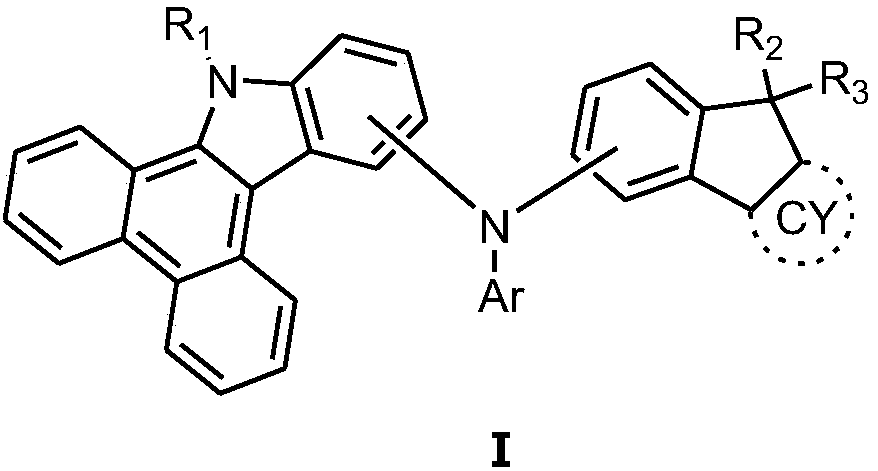
Fused ring compound and organic luminescent device thereof
A technology of organic light-emitting devices and compounds, applied in the field of organic photoelectric materials, to achieve low driving voltage, good electrical and optical properties, and high efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0063]
[0064] Preparation of intermediate A1
[0065] Tri-tert-butylphosphine (4.4 mL of a 1.0M solution in toluene, 1.48 g, 0.05 mmol), palladium acetate (0.4 g, 1.83 mmol) and sodium tert-butoxide (22.8 g, 238 mmol) were added to 12-bromo-9H - A solution of dibenzo[a,c]carbazole (63.3 g, 183 mmol) and iodobenzene (38.1 g, 185 mmol) in degassed toluene (1 L), and the mixture was heated at reflux for 2 hours. The reaction mixture was cooled to room temperature, diluted with toluene and filtered through celite. The filtrate was diluted with water and extracted with toluene, and the combined organic phases were evaporated under vacuum. The residue was filtered through silica gel and recrystallized. Intermediate A1 (54.0 g, 70% of theory) was obtained. Mass Spectrum m / z: 422.28 (calculated: 422.32). Theoretical element content (%)C 26 h 16 BrN: C, 73.94; H, 3.82; Br, 18.92; N, 3.32 Measured element content (%): C, 73.95; H, 3.81; Br, 18.92; N, 3.32. The above results ...
Embodiment 2
[0071]
[0072] In Example 1, 2-bromo-9,9-dimethylfluorene was replaced with equimolar 2-bromo-11,11-dimethyl-11H-indeno[b]anthracene, and the other steps were the same to obtain compound 4 . Mass Spectrum m / z: 676.87 (calculated: 676.84). Theoretical element content (%)C 51 h 36 N 2 : C, 90.50; H, 5.36; N, 4.14 Measured element content (%): C, 90.52; H, 5.37; N, 4.11. The above results confirmed that the obtained product was the target product.
Embodiment 3
[0074]
[0075] Compound 11 was obtained by replacing 2-bromo-9,9-dimethylfluorene in Example 1 with equimolar 3-bromo-9,9-diphenylfluorene, and the other steps were the same. Mass Spectrum m / z: 750.99 (calculated: 750.93). Theoretical element content (%)C 57 h 38 N 2 : C, 91.17; H, 5.10; N, 3.73 The measured element content (%): C, 91.15; H, 5.12; N, 3.73. The above results confirmed that the obtained product was the target product.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More