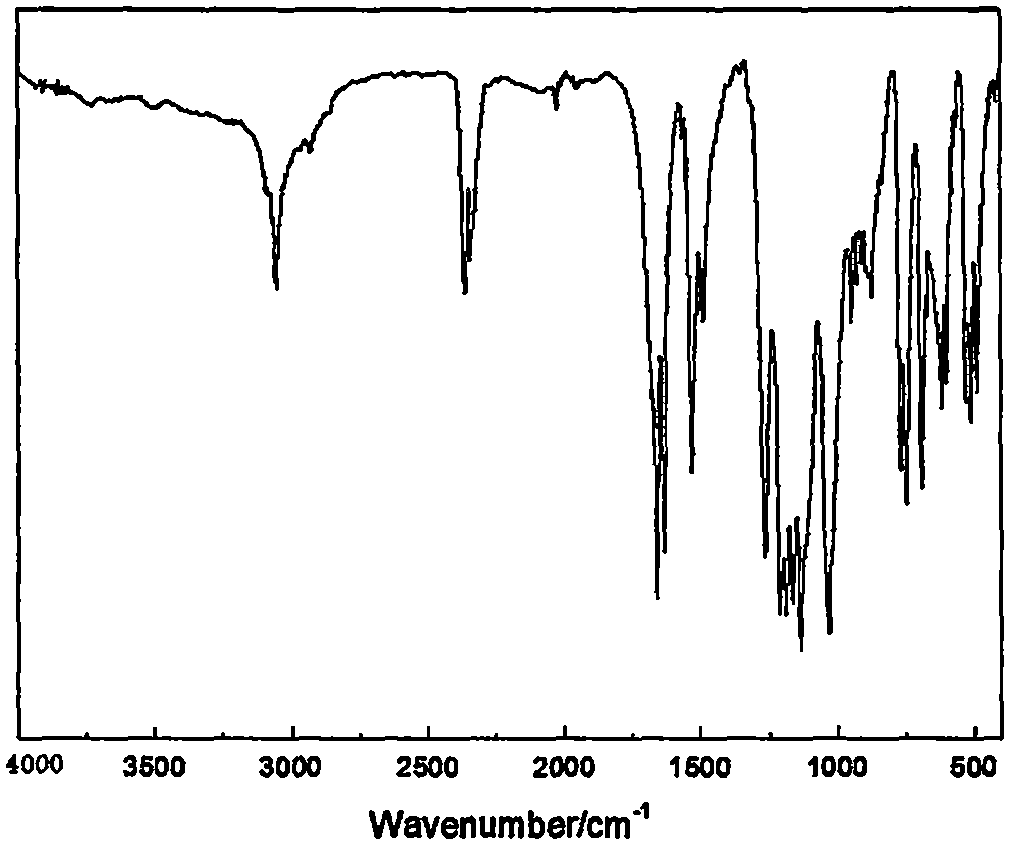
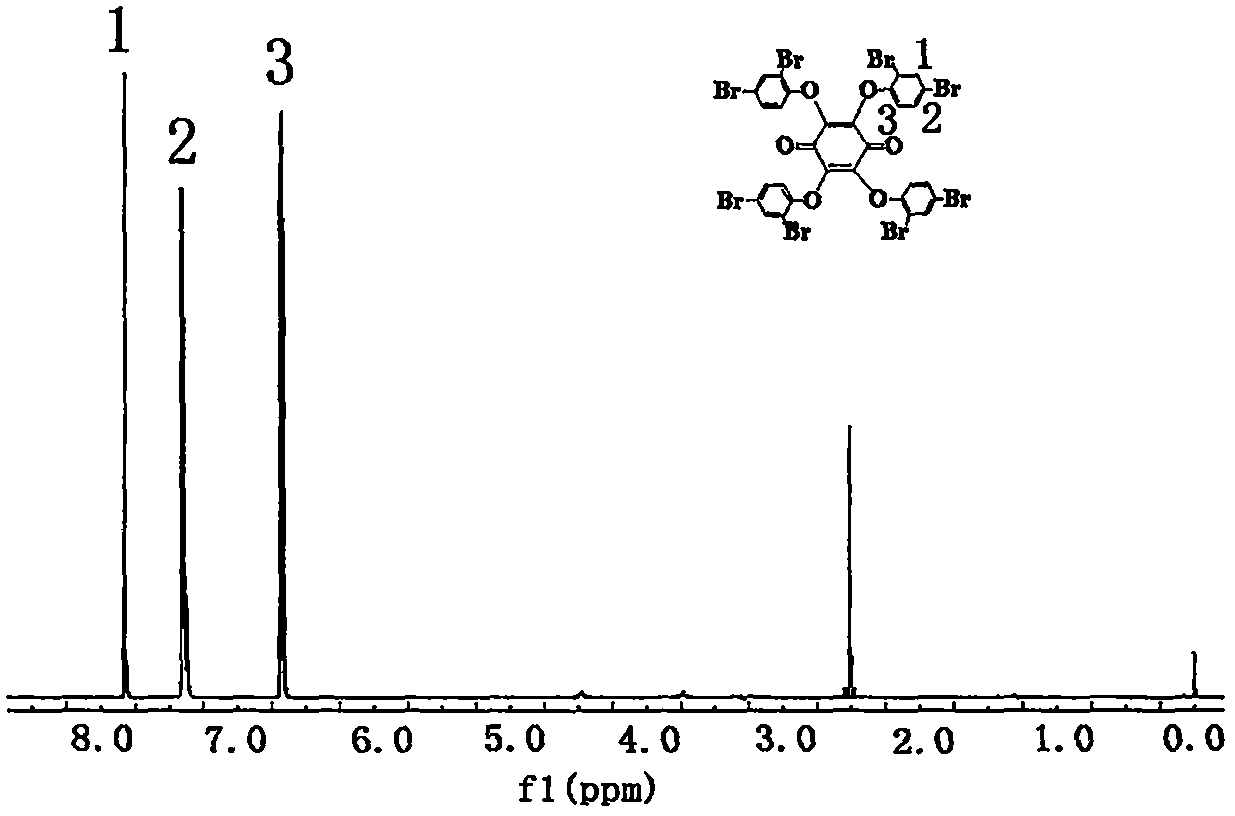
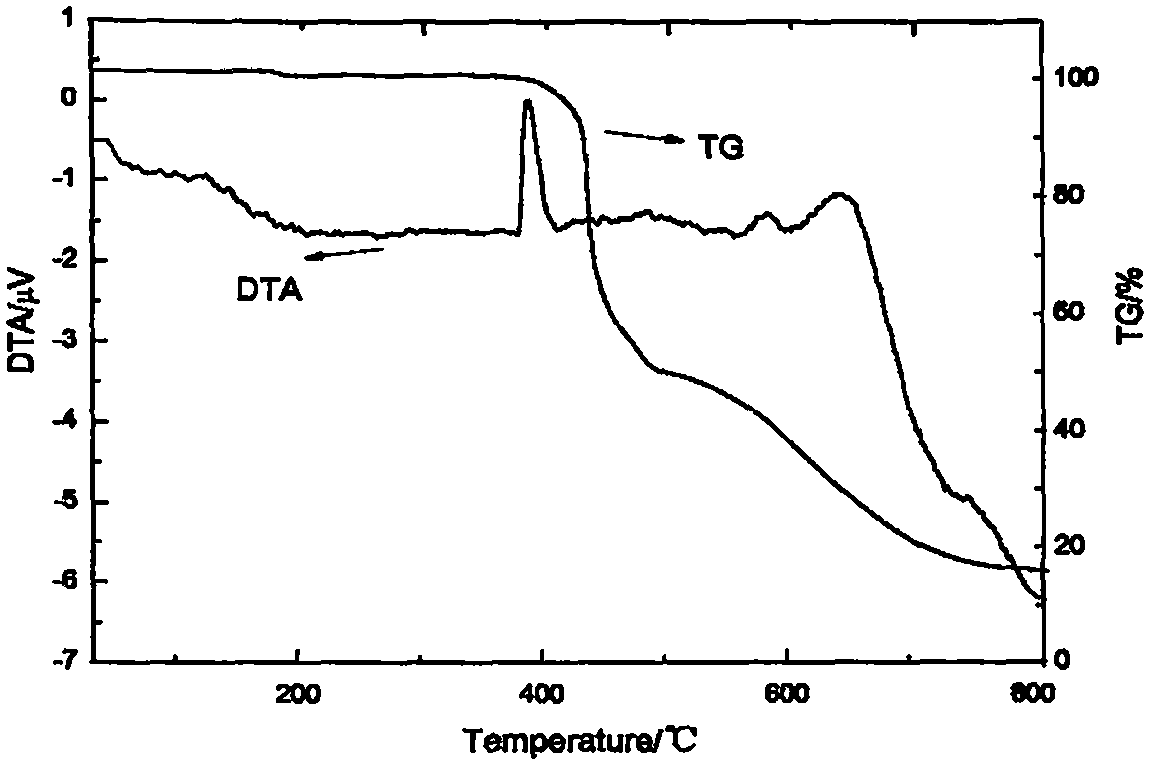
Colored flame retardant tetra(bromophenoxy)p-benzoquinone compound and preparation method thereof
A technology of bromophenoxy and tetrachloro-p-benzoquinone, which is applied in the field of tetra-p-benzoquinone compounds and its preparation, to achieve the effects of cheap raw materials, good compatibility and high decomposition temperature
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] Example 1 In a reactor equipped with a stirrer, a thermometer, and a high-efficiency reflux device, at 20°C, first add 80ml of benzene, then add 10.1g (0.04mol) of 2,4-dibromophenate sodium, and then add 2.46g (0.01mol) tetrachloro-p-benzoquinone, heat preservation reaction at 80°C for 5 hours, change the reflux device to a vacuum distillation device, evaporate benzene to dryness under reduced pressure (recycling), then add 200ml methanol to dissolve the product, filter to remove chlorine NaCl and trace insolubles, the filtrate was concentrated to 100ml, cooled and crystallized, filtered, and dried to obtain a brown solid product tetrakis(2,4-dibromophenoxy)-p-benzoquinone with a yield of 80.0%.
Embodiment 2
[0030] Example 2 In a reactor equipped with a stirrer, a thermometer, and a high-efficiency reflux device, at 20°C, first add 90ml of toluene, then add 10.1g (0.04mol) of 2,4-dibromophenol sodium, and then add 2.46g (0.01mol) tetrachloro-p-benzoquinone, heat preservation reaction at 100°C for 6h, change the reflux device to a vacuum distillation device, evaporate the toluene to dryness under reduced pressure (recycling), then add 200ml methanol to dissolve the product, filter to remove chlorine Sodium chloride and trace insolubles, the filtrate was concentrated to 100ml, cooled and crystallized, filtered, and dried to obtain a brown solid product tetrakis (2,4-dibromophenoxy)-p-benzoquinone with a yield of 82.3%.
Embodiment 3
[0031] Example 3 In a reactor equipped with a stirrer, a thermometer, and a high-efficiency reflux device, at 20°C, first add 100ml of chlorobenzene, then add 11.3g (0.045mol) of 2,4-dibromophenate sodium, and then add 2.46 g (0.01mol) tetrachloro-p-benzoquinone, heat preservation reaction at 110°C for 7 hours, change the reflux device to a vacuum distillation device, evaporate chlorobenzene to dryness under reduced pressure (recycling), then add 200ml of methanol to dissolve the product, filter Sodium chloride and trace insolubles were removed, the filtrate was concentrated to 100ml, cooled, crystallized, filtered, and dried to obtain a brown solid product tetrakis(2,4-dibromophenoxy)-p-benzoquinone with a yield of 86.5%.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Length | aaaaa | aaaaa |
| Diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More