Trivalent iron porphrin and its derivative-short peptide compound and its synthesis
A technology of derivatives and ferric iron, applied in the direction of peptides, etc., can solve the problems of many purification process steps, difficult artificial synthesis, complex components, etc., and achieve the effect of broad drug development and application prospects, saving raw materials, and high purity.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0018] Example 1. Determination of the optimum relative position of the active group histidine (His) and sub-heme or heme in the peptide chain
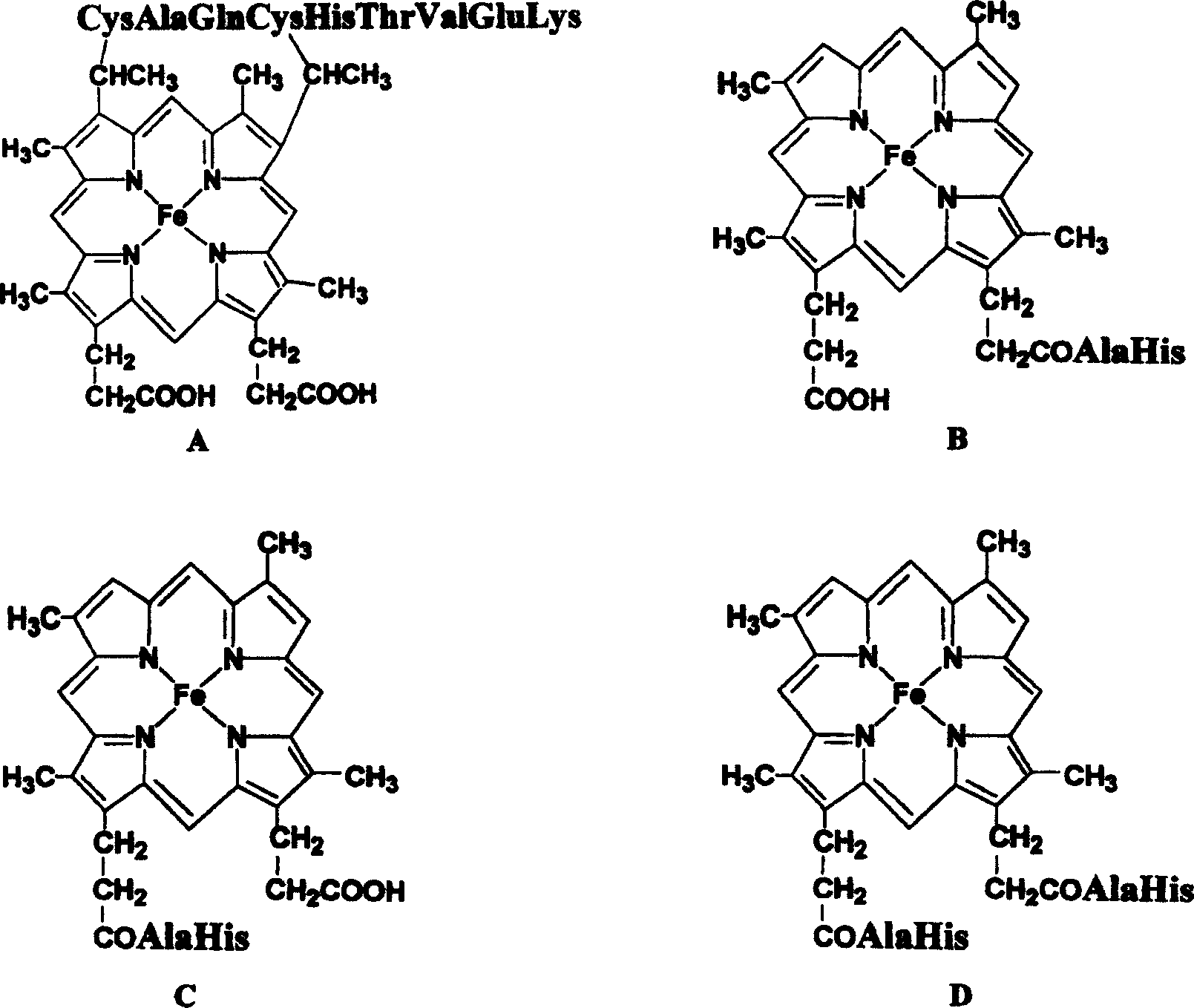
[0019] The sub-heme in the sub-heme-nonapeptide (DhHP-9) of the present invention has a different connection mode with the peptide chain than in MP-9, so the relative positions of His and the sub-heme in the primary structure and spatial structure are also different. Different from MP-9, it is necessary to determine the optimal relative position of His and hypoheme (Dh) in DhHP-9. For this reason, a series of hypoheme-short peptides were synthesized with SP-3 automatic peptide synthesizer: Dh-H, Dh-A-H, Dh-A2-H, Dh-A3-H, Dh-A4-H, Dh-A5 -H, Dh-A6-H.
[0020] The in vitro activity test showed that the specific activity of Dh-A-H enzyme with one Ala between the hypoheme and His was the highest, which was close to the level of natural MP-9 in value, and then the enzyme activity gradually decreased with the increase of the number of Ala. ...
Embodiment 2
[0022] Embodiment 2, the design of optimum short peptide
[0023]The most suitable short peptides were designed according to the experimental results such as assay activity: monomer subhemin-hexapeptide (DhHP-6: Deuterohemin-His-Peptide, Dh-AHTVEK) and heme-hexapeptide. The synthesis is carried out according to the technological process of the present invention by using an automatic polypeptide synthesizer, and the "deprotection coupling cycle" is carried out five times. Vitality assays in vitro showed that the enzyme specific activity (U / μmol) of DhHP-6 had reached the level of natural MP-9. At the same time, heme is replaced by hypoheme in the DhHP-6 molecule, and the peptide chain has three amino acids less than MP-9, so the molecular weight is smaller than natural MP-9, and the specific activity per μg of DhHP-6 is higher than that of natural MP-9, which is An ideal peroxidase mimic.
Embodiment 3
[0024] Embodiment 3, the synthesis of monomer hypoheme-A(D)-HTVEK and monomer heme-β-AHTVEK
[0025] (D)-H and β-A were introduced, and monomeric hypoheme-A(D)-HTVEK and monomeric heme-β-AHTVEK were synthesized with SP-3 automatic peptide synthesizer, both of which were active in vitro.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More