Compositions and Methods of Use for Mgd-Csf in Disease Treatment
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Amino Acid Sequence Alignment of MGD-CSF with MCG34647
[0378] MGD-CSF and NP—689669 were compared by aligning their amino acids using the program clustal format for T-COFFEE Version 1.37, cpu=0.00 sec., score=72, Nseq=3, len=242. As shown in FIG. 1, the amino acid sequence of MGD-CSF differs from the amino acid sequence of NP—689669 (MCG34647). The latter sequence has a glutamine (Q) residue at amino acid 81. The five flanking amino acid residues adjacent to and on either side of amino acid 81 in the NCBI sequence of MCG34647 are NVTRLQRAQVS (SEQ ID NO.:279). In contrast to this published sequence of MCG34647, MGD-CSF contains the amino acid sequence NVTRLRAQVS (SEQ ID NO.:280). The difference between these sequences results from alternative splicing of the MCG34647 gene between exons 3 and 4. The genome sequences at the exon 3-4 boundary are the codons aac gtc acc agg ctg gtg (SEQ ID NO.:281) and cag cag agg gcc cag gtg agc (SEQ ID NO.:282), wherein the gtg codon (shown in italics)...
example 2
Plasmid Vectors for MGD-CSF Expression
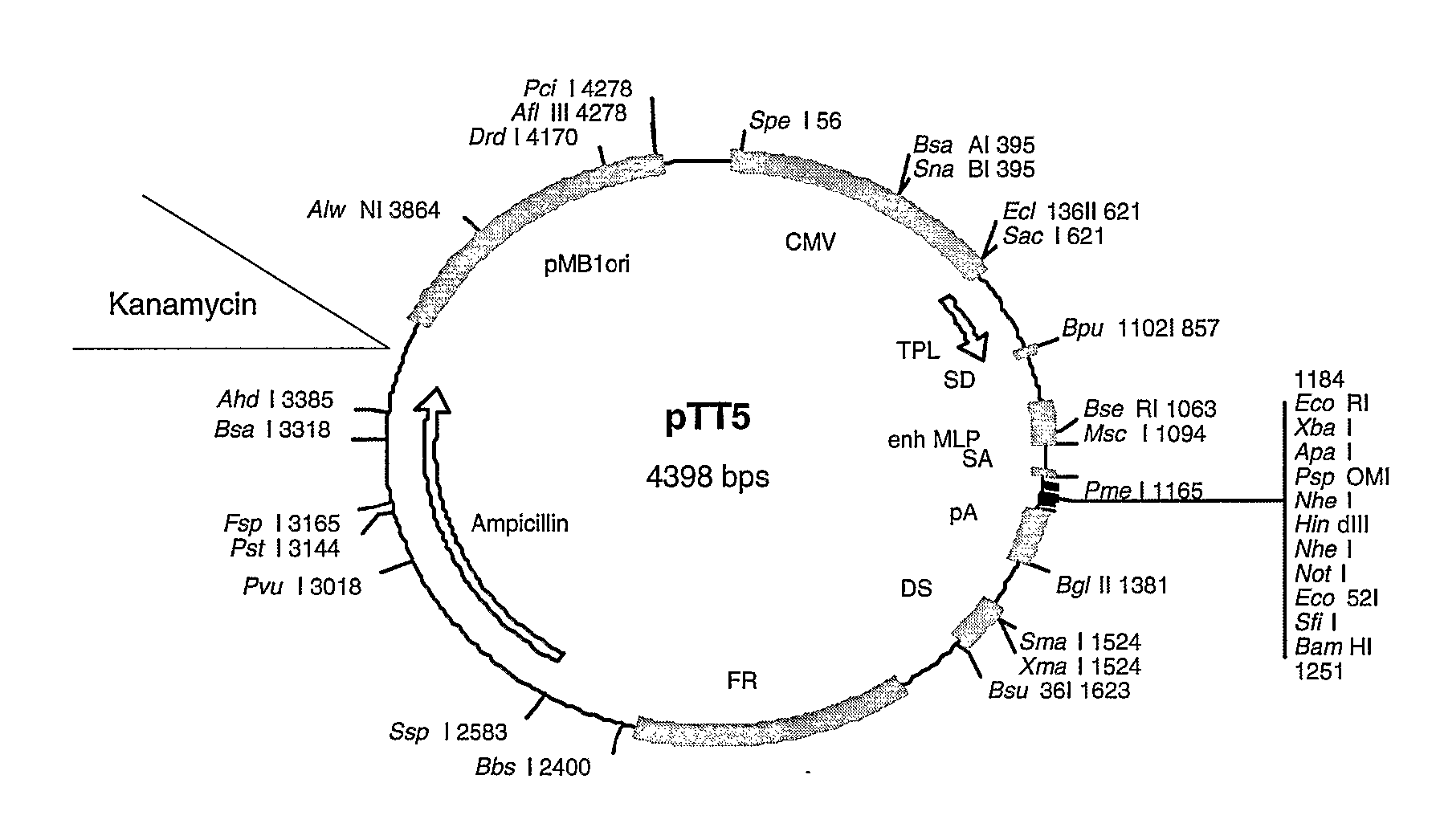
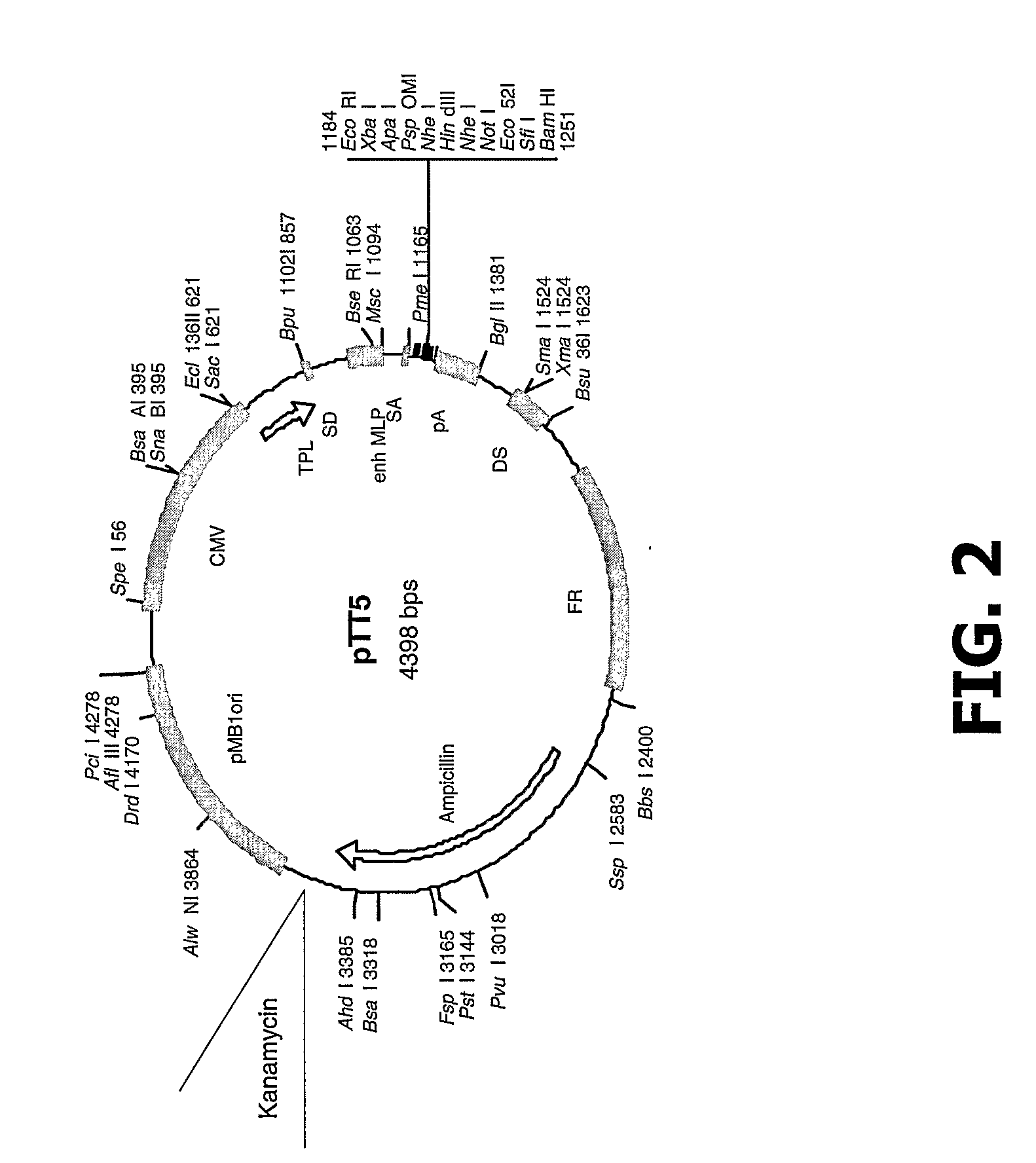
[0380] The MGD-CSF gene was cloned into pTT-5 and pTT-2 mammalian expression vectors modified as shown in FIG. 2 and FIG. 3 using standard cloning procedures. The MGD-CSF gene was also cloned into the pIB / V5His-DEST insect cell expression vector (Invitrogen, Carlsbad Calif.) using standard cloning procedures. The resulting constructs are described in Table 1 and Table 5. They include human MGD-CSF untagged in vector pTT5 (MGD-CSF), human MGD-CSF untagged in vector pTT2 (CLN00839395), human MGD-CSF with a C-terminal V5H8 tag in vector pTT5 (CLN00732663), human MGD-CSF tagged with V5H8 (CLN00732663), human MGD-CSF with a C-terminal V5H8 tag in vector pTT2 (CLN00840351), human MGD-CSF with a C-terminal V5H8 tag in vector pIB / V5His-DEST (CLN00758593), and human MGD-CSF with a collagen secretory leader and a C-terminal Streptag in vector pTT5-G (CLN00816424).
[0381] To monitor the expression and secretion of MGD-CSF and to aid in its purification, t...
example 3
Transient Expression in Mammalian Cells
[0387] Complementary DNA encoding the MGD-CSF polypeptide was cloned into the expression vectors pTT5 and pCDNA-pDEST40 and expressed as both a tagged and untagged protein. Protein levels were quantified by measuring the levels of the tag, for example, a V5His tag, by quantitative Western blot analysis. The expression vectors were transfected into adherent 293T cells using the transfection agent Fugene 6 (Roche, Nutley N.J.) in DMEM with 10% fetal bovine serum (FBS) and penicillin / streptomycin (100 μg / ml, 100 U / ml), and incubated at 37° C. in 5% CO2 for 40 hours, after which the cells were washed with PBS and incubated for an additional 48 hours in complete DMEM. Cell supernatant was harvested, cleared of cell debris by centrifugation, and tested for biological activity (untagged cDNA) and protein expression (V5 tagged cDNA) by Western blot assay using an anti-V5 antibody.
[0388] Expression studies were also performed with 293-6E cells transie...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Volume | aaaaa | aaaaa |
| Volume | aaaaa | aaaaa |
| Volume | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More