Mammallian cell culture process for protein production
a cell culture and protein technology, applied in the field of mammalian cell culture methods and processes, can solve the problems of affecting the immunogenicity and clearance rate of products, affecting the run time of cell culture processes, and reducing the viability of cells, so as to achieve the effect of increasing cell viability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0170]This Example provides materials and reagents employed in the processes of the present invention for the culturing of recombinant cells that produce the exemplified CTLA4Ig fusion proteins as described herein in Examples 2-4.
1. Cell Culture Medium
[0171]The basal cell culture medium used for all phases of cell inoculum generation and for growth of cultures in bioreactors, including 5 liter (5 L) and 50 liter (50 L) production reactors, was modified CD-CHO medium containing glutamine, sodium bicarbonate, insulin and methotrexate (Invitrogen, Carlsbad, Calif.), as exemplified in Table 1. The pH of the medium was adjusted to 7.0 with 1 N HCl.
TABLE 1Modified CD-CHOMedium ComponentConcentrationCD-CHO 25x Acids I40ml / L(Invitrogen, Carlsbad, CA)CD-CHO 25x Acids II40ml / L(Invitrogen, Carlsbad, CA)CD-CHO 25x Salts I40ml / L(Invitrogen, Carlsbad, CA)CD-CHO 25x Salts II40ml / L(Invitrogen, Carlsbad, CA)L-glutamine0.585g / L(Invitrogen)r-human insulin0.1ml / L(10 mg / mL)(Invitrogen)Methotrexate5μl / L(...
example 2
[0177]This Example describes the production of CTLA4Ig, shown as −1 to 357 or +1 to 357 in FIG. 3, (encoding DNA deposited as ATCC 68629), from cultured CHO cells.
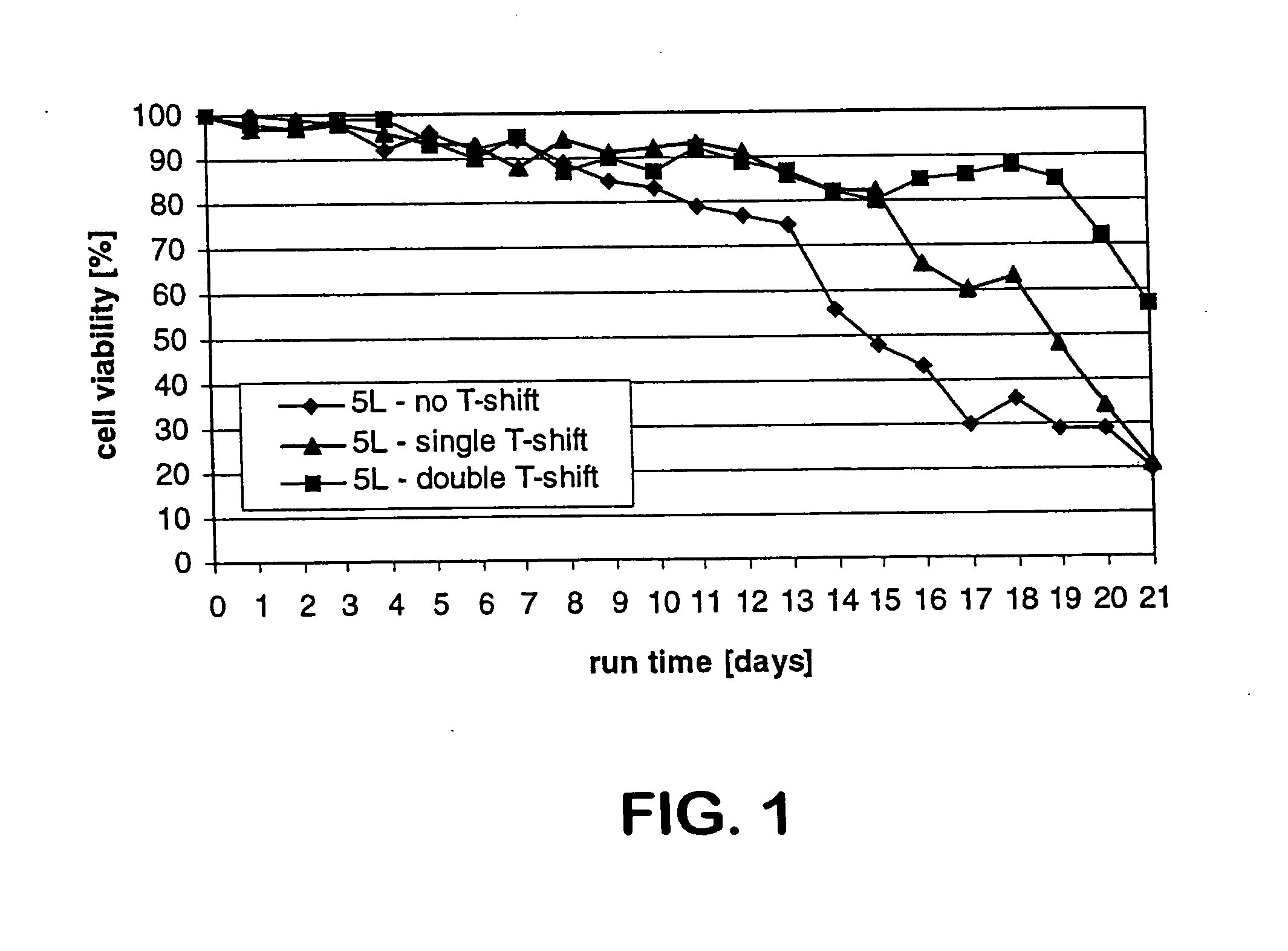
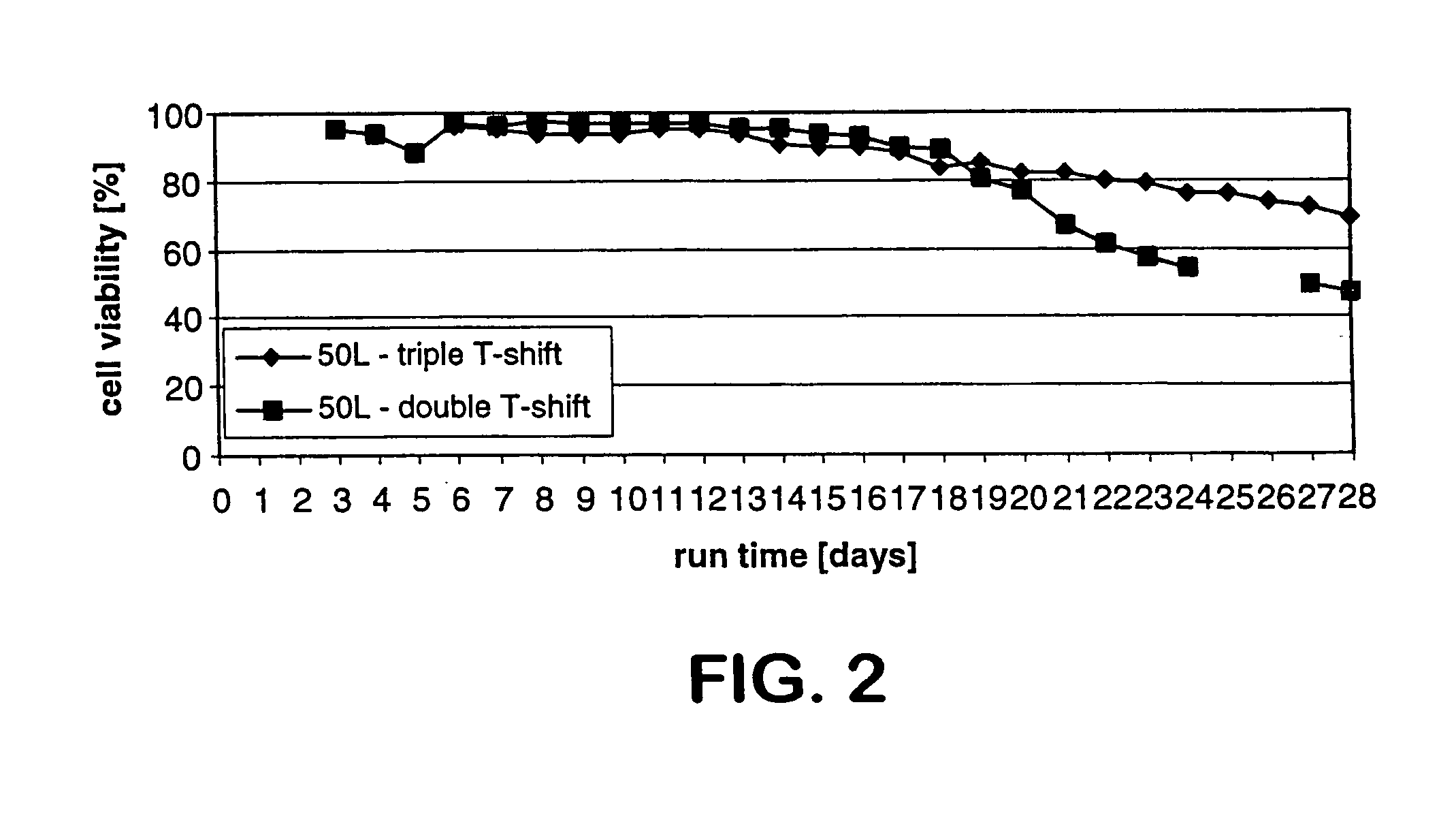
[0178]This Example also describes a process of this invention for producing both high quantity and high quality CTLA4Ig protein, involving culture runs having two- or three-step temperature shifts and total run times of 14, 21, or 28-30 days. A temperature shift (T-shift) from 37° C. to 34° C. occurred on day 6 (end of logarithmic growth phase) and a second T-shift from 34° C. to 32° C. occurred on day 10. The run was ended on day 14, day 21, or day 28, and for the two-step shift, the temperature was controlled at 32° C. from the shift on day 10 until the end of the run. For the three-step shift, the temperature was controlled at 30° C. from the day of the shift until the end of the run The processes described resulted in increased end titer of protein product, increased end cell viability, and volumetric productivity, com...
example 3
[0183]This Example describes and presents the results of comparative evaluations to assess various culturing procedures, including the multi-step culturing methods performed in accordance with the present invention. The end titer (in g / L) of glycoprotein product was determined, as were the end titer sialic acid content of the protein, the cell viability at the end of the runs (end cell viability) and the cell density at the end of the runs (viable end cell density).
[0184]Experiments I-A, I-B and I-C; II-A, II-B and II-C; and III-A, III-B and III-C refer to the same cell culture run with the same temperature shift profile assessed at different times, i.e., for I-A, II-A, and III-A, the product and cell parameters were assessed after 14 days, for I-B, II-B and III-B after 21 days, and for I-C, II-C and III-C after 28 days. These experiments were performed in a 5 L bioreactor in which the culture conditions were controlled as follows: pH at 7.0; dissolved oxygen at 40%; agitation at 60...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More