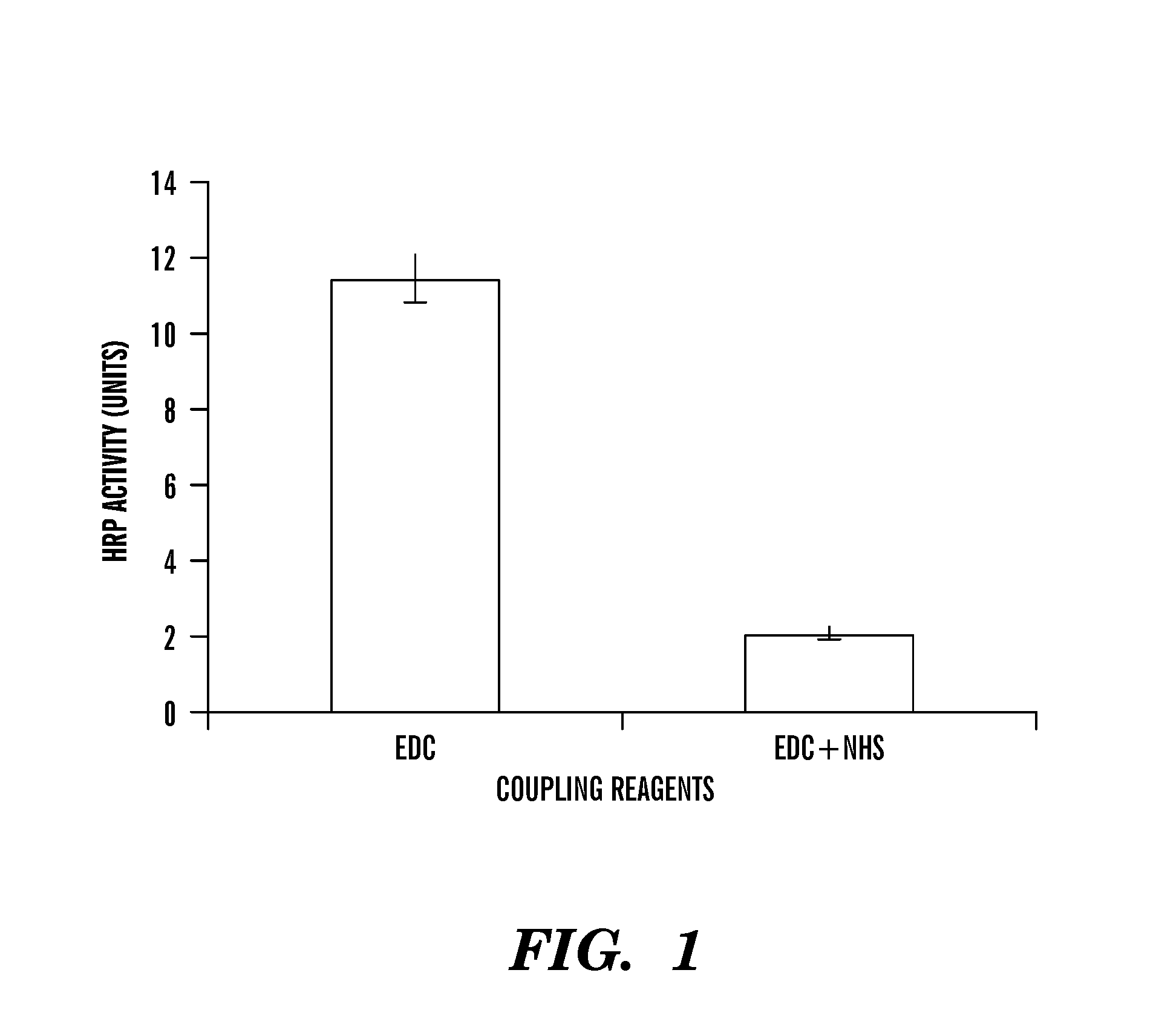
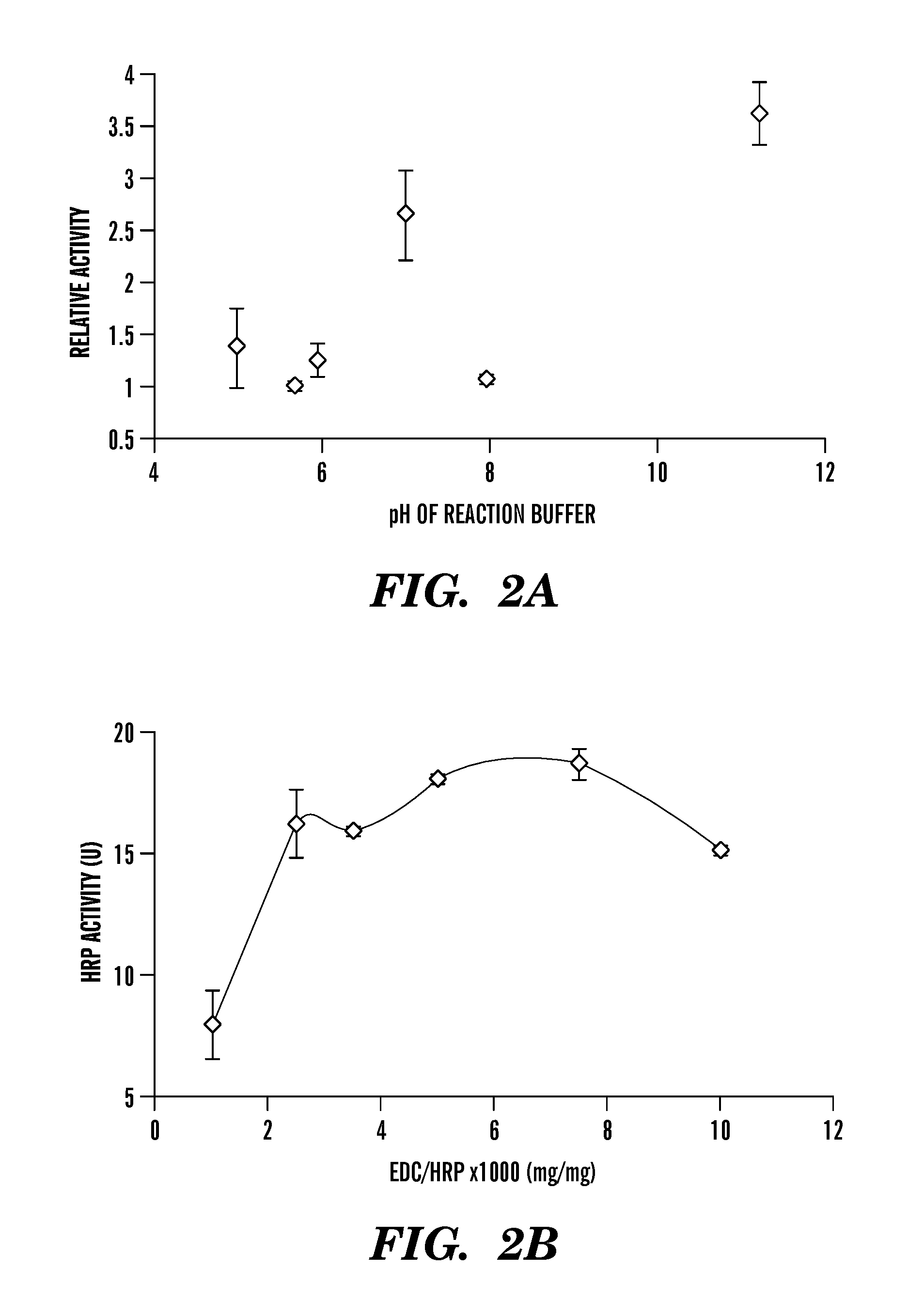
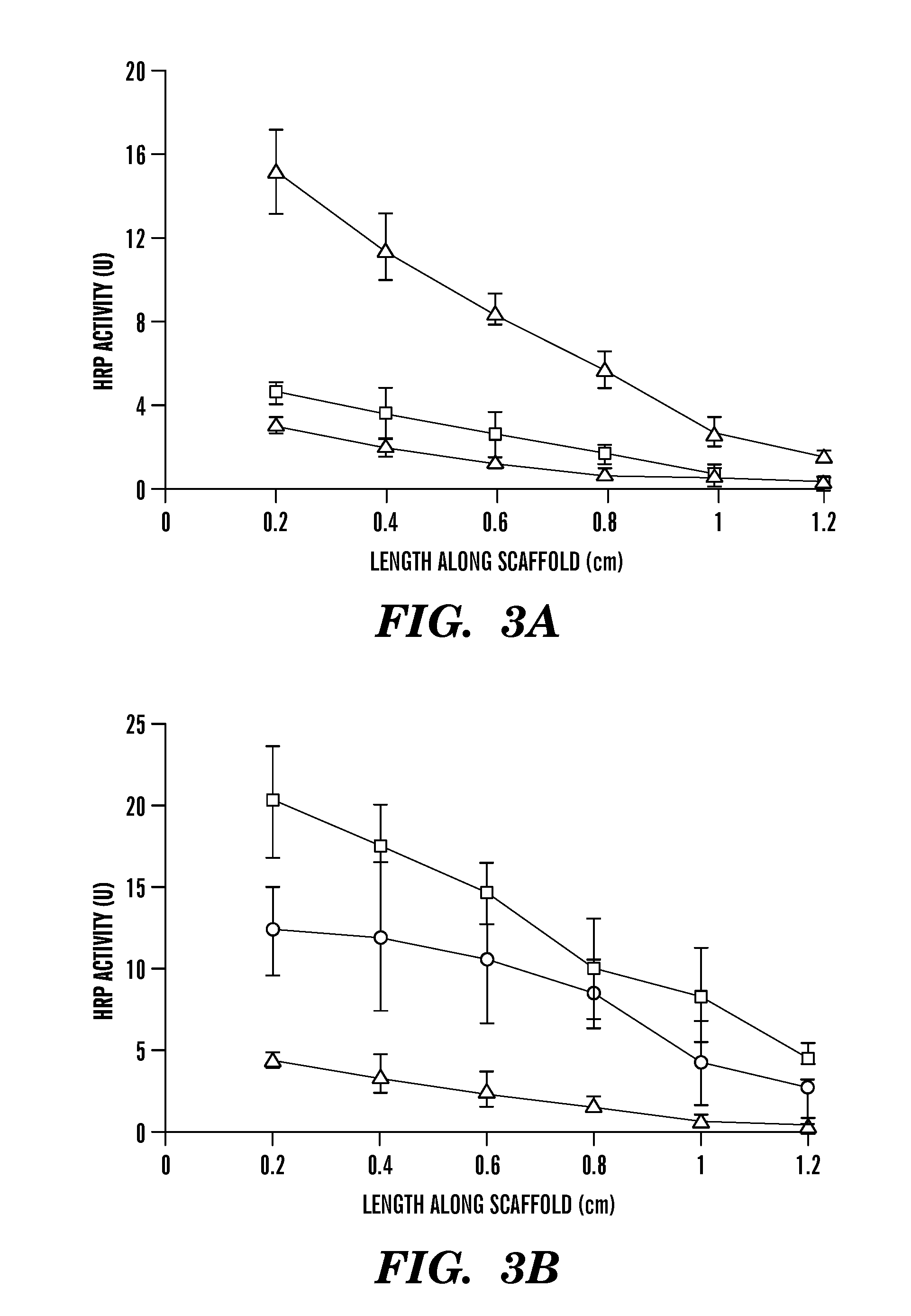
Covalently immobilized protein gradients in three-dimensional porous scaffolds
a technology of porous scaffolds and protein gradients, which is applied in the direction of peptides, biocides, peptide/protein ingredients, etc., can solve the problems of restricting the process to simple patterns, and limiting the scale and speed of such processes
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Materials and Methods
Preparation of Silk Fibroin Scaffold
[0037]Cocoons from Bombyx mori (supplied by M. Tsukada, Institute of Sericulture, Tsukuda, Japan) were de-gummed for 1 hr in 0.02 M Na2CO3 (Fisher, Pittsburgh, Pa.) solution to extract sericin from fibroin. Silk fibroin was washed thoroughly to remove sericin, dried and dissolved in 9.3 M lithium bromide at 500 c to generate a 10% (w / v) solution. The solution was dialyzed in a Slide-A-Lyzer (3500MW cassette, Pierce, IL) against water for 3 days to remove lithium bromide. The solution was lyophilized and then dissolved in hexafluoro-2-propanol (Aldrich, Milwaukee, Wis.) to generate a 15% silk solution, a modification of our previously reported procedure [(Nazarov et al. 2004; Sofia et al. 2001).
[0038]Granular sodium chloride (crystal sizes from 150 to 700 μm, Sigma, Mo.) was used as a porogen (Nazarov et al, 2004). The 15% silk solution was poured into a Teflon hexagon dish (medium size, Fisher, Pittsburgh, Pa.) containing leve...
example 2
[0066]Recombinant Human BMP-2 (rhBMP-2) Gradients
[0067]Using methods similar to Example 1 to confirm our findings with HRP, rhBMP-2 gradients were produced. Silk fibroin scaffolds were activated using EDC / NHS for 15 minutes. Different volumes of rhBMP-2 were allowed to diffuse and create different immobilized gradients of rhBMP-2 within the silk fibroin scaffold (FIGS. 9A and 9B).
[0068]Bone marrow stromal cells were seeded into the silk fibroin scaffolds and supplied with osteogenic media. Scaffolds were grown in 6 well plates for 4-5 weeks and supplied with 50 μg / ml ascorbic acid and 3 mM B-glycerophosphate and 10−8 M dexamethasone. Calcium mineral assay showed a gradient response along the length of the scaffold.
[0069]Increase in the amount of BMP-2 (from 4 μL to 9 μL), results in an increase in the calcium mineral content showing a dose dependent increase in mineral deposition with increase in BMP-2. Immobilized BMP-2 shows control in the response of human bone marrow stromal cel...
PUM
| Property | Measurement | Unit |
|---|---|---|
| porosity | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More