Radio-pharmaceutical complexes
a radiopharmaceutical and complex technology, applied in the field of complexes of thorium isotopes, can solve the problems of unsatisfactory liposome administration, undesired systemic toxicity, and difficulty in commercial production and distribution of radiopharmaceuticals based on these radionuclides
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Isolation of Pure Thorium-227
[0134]Thorium-227 is isolated from an actinium-227 cow. Actinium-227 was produced through thermal neutron irradiation of Radium-226 followed by the decay of Radium-227 (t1 / 2=42.2 m) to Actinium-227. Thorium-227 was selectively retained from an Actinium-227 decay mixture in 8M HNO3 solution by anion exchange chromatography. A column of 2 mm internal diameter, length 30 mm, containing 70 mg of AG®1-X8 resin (200-400 mesh, nitrate form) was used. After Actinium-227, Radium-223 and daughters had eluted from the column, Thorium-227 was extracted from the column with 12M HCl. The eluate containing Thorium-227 was evaporated to dryness and the residue resuspended in 0.01M HCl.
example 2
Synthesis of Compound 12
Step 1
[0135]
[0136]2-benzyloxyethylamine (31 g, 207 mmol) and glycolonitrile (16 mL, 70% solution in water, 207 mmol) was dissolved in 300 mL EtOH (abs) and refluxed for 4 h. The volatiles were removed under reduced pressure. The crude product (24.7 g, 130 mmol) was carried on to the next step without further purification.
[0137]1H-NMR (CDCl3, 400 MHz): 2.92 (m, 2H), 3.58-3.62 (m, 4H), 4.51 (s, 2H), 7.25-7.37 (m, 5H)
Step 2
[0138]
[0139]1 (24.7 g, 130 mmol) was dissolved in dry ether. HCl (g) was bubbled through the solution for 30 minutes. The precipitate was filtered off and dried under reduced pressure, giving the desired product (27.8 g, 122.6 mmol. The product was carried on to the next step without further purification or analysis.
Step 3
[0140]
[0141]2 (27.8 g, 122.6 mmol) was dissolved in 230 mL chlorobenzene at room temperature. Oxallyl chloride (45 mL, 530 mmol) dissolved in 100 mL chlorobenzene was added drop wise over 30 minutes at room temperature. The r...
example 3
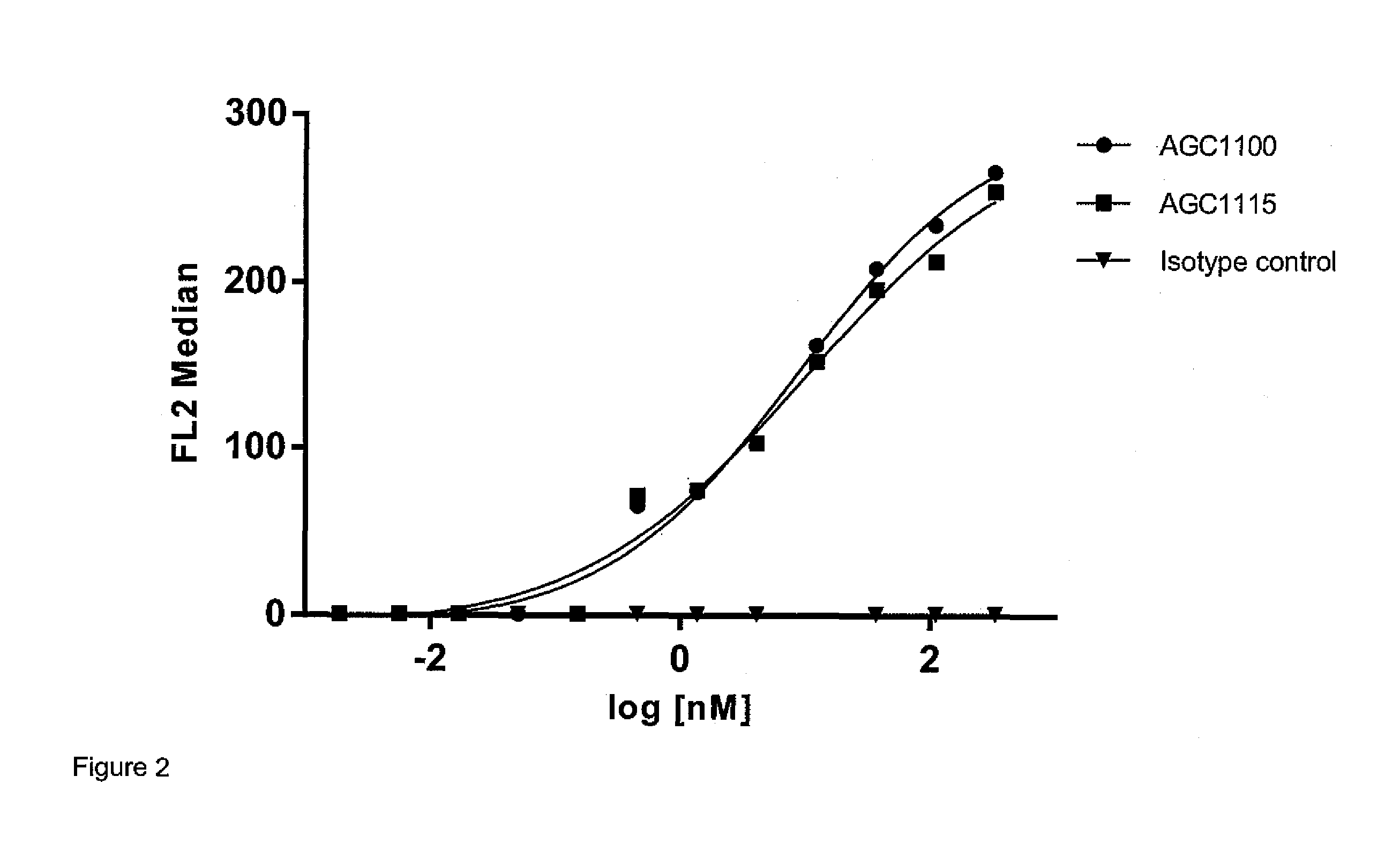
Generation of the Anti-CD22 Monoclonal Antibody (AGC1100)
[0169]The sequence of the monoclonal antibody (mAb) hLL2, also called epratuzumab, here denoted AGC1100, was constructed as described in (1). The mAb used in the current examples was produced by Immunomedics Inc, New Jersey, USA. Production of this mAb could for example be done in Chinese hamster ovarian suspension (CHO-S) cells, transfected with a plasmid encoding the genes encoding the light and the heavy chain. First stable clones would be selected for using standard procedures. Following approximately 14 days in a single-use bioreactor, the monoclonal antibody may be harvested after filtration of the supernatant. AGC1100 would be further purified by protein A affinity chromatography (MabSelect SuRe, Atoll, Weingarten / Germany), followed by an ion exchange step. A third purification step based on electrostatic and hydrophobicity could be used to remove aggregates and potentially remaining impurities. The identity of AGC1100 ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameters | aaaaa | aaaaa |
| energy | aaaaa | aaaaa |
| physiological temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More