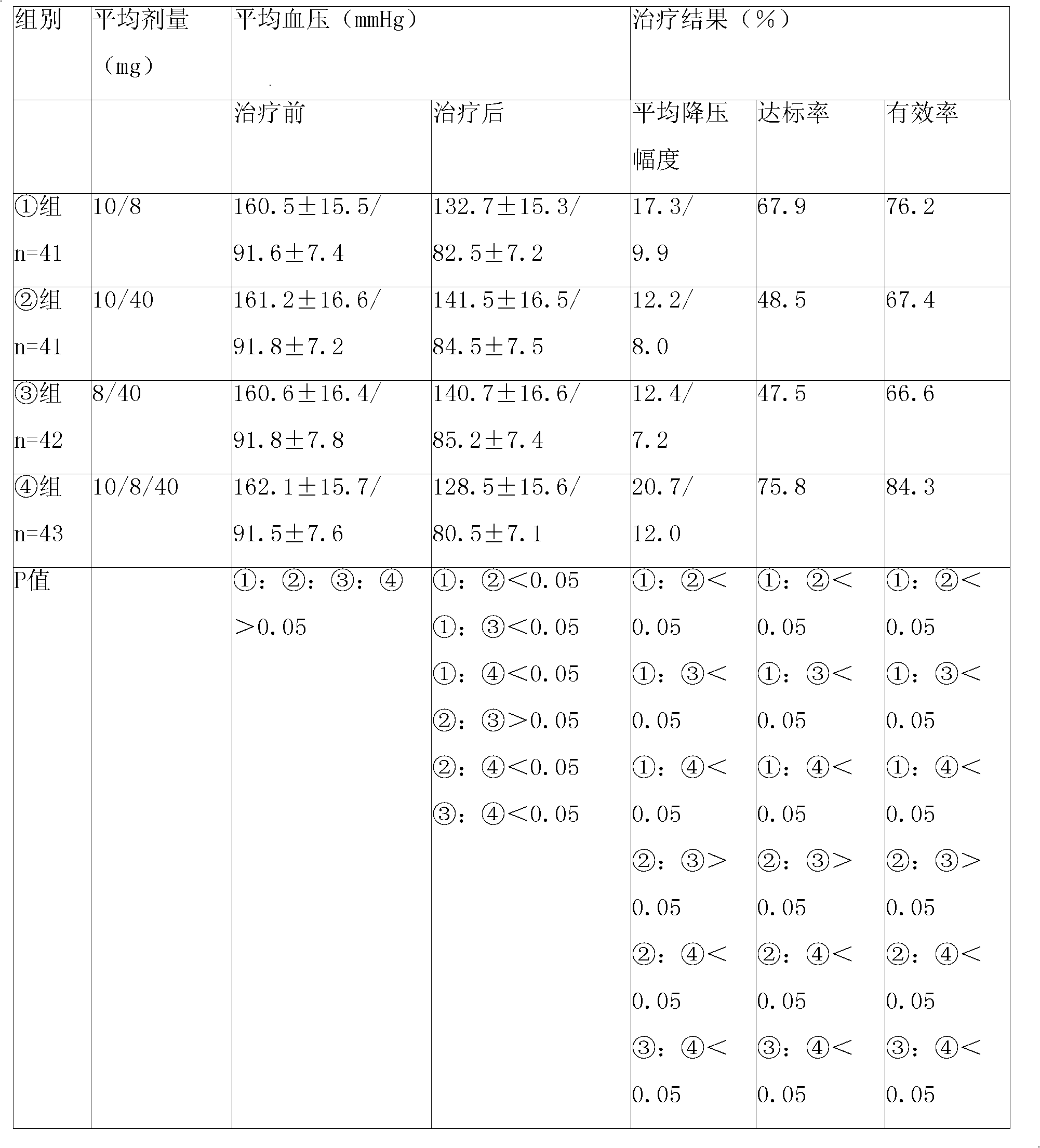Pharmaceutical composition containing calcium blocker, AII receptor blocker and statins
A receptor antagonist, calcium ion antagonist technology, applied in the field of medicine, can solve problems such as unsatisfactory
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
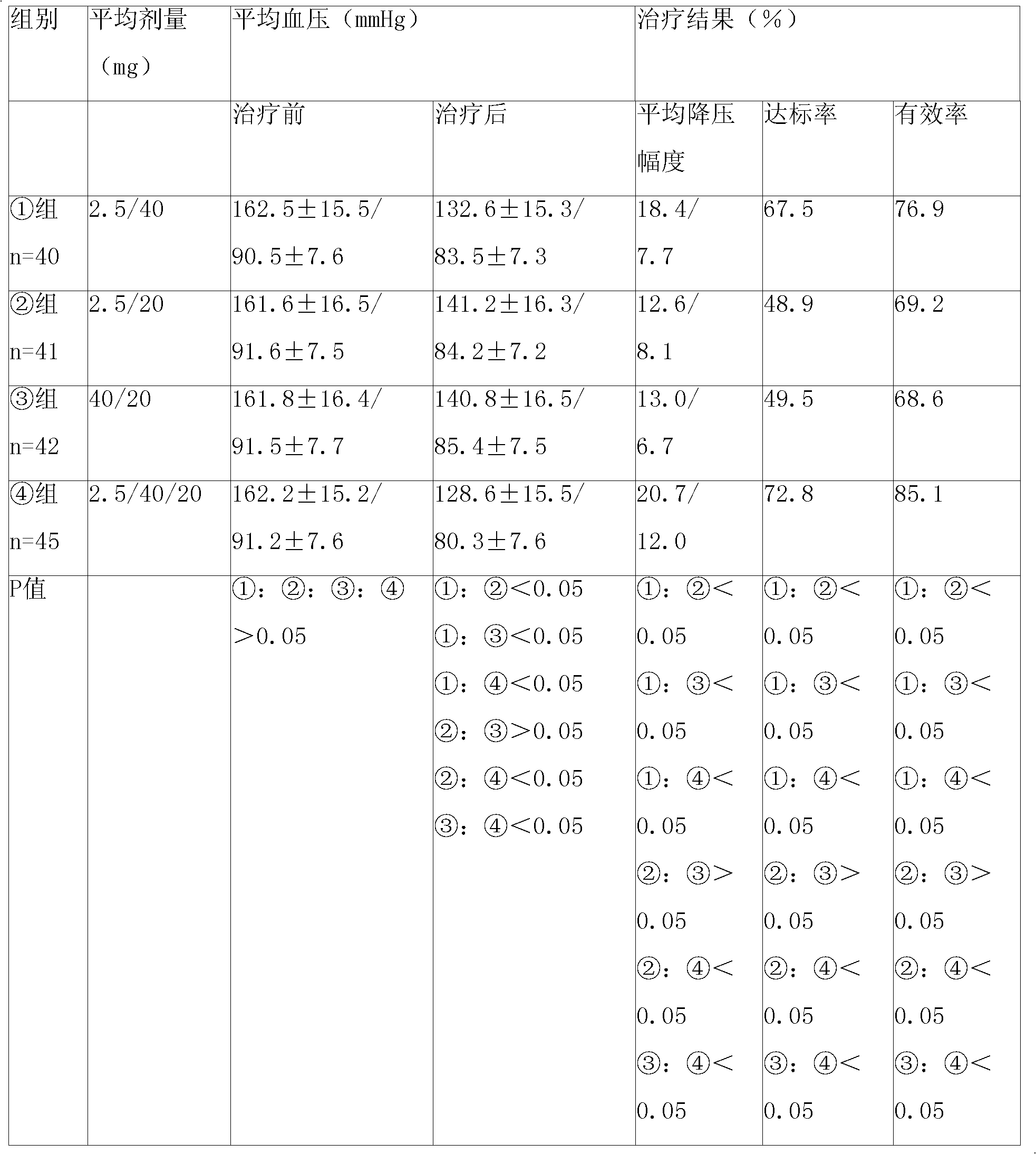
[0092] Embodiment 1: levamlodipine besylate, telmisartan and atorvastatin calcium tablet
[0093] Name of raw material
Specification 1 Dosage
Specification 2 Dosage
Specification 3 Dosage
levamlodipine besylate
(calculated as levamlodipine)
1.25g
2.5g
5.0g
20.0g
40.0g
80.0g
(calculated as atorvastatin)
10.0g
20.0g
80.0g
155g
140g
75g
18g
18g
18g
Polysorbate 80
5g
5g
5g
0.65g
0.65g
0.65g
Micropowder silica gel
1.25g
1.25g
1.25g
37.5g
37.5g
37.5g
8g
8g
8g
Crospovidone
10g
10g
10g
Appropriate am...
Embodiment 2
[0096] Embodiment 2: Nitrendipine, candesartan cilexetil and fluvastatin sodium capsules
[0097] Name of raw material
Specification 1 Dosage
Specification 2 Dosage
Specification 3 Dosage
5.0g
10.0g
20.0g
candesartan cilexetil
4.0g
8.0g
32.0g
Fluvastatin sodium (calculated as fluvastatin)
20.0g
40.0g
80.0g
160g
125g
70g
22g
22g
22g
Polysorbate 80
5.5g
5.5g
5.5g
0.65g
0.65g
0.65g
Micropowder silica gel
1.8g
1.8g
1.8g
3.5g
3.5g
3.5g
sodium starch glycolate
8.5g
8.5g
8.5g
Crospovidone
9g
9g
9g
9.5g
9.5g
9.5g
Appropriate amount
Appropr...
Embodiment 3
[0100] Embodiment 3: lacidipine, telmisartan, losartan potassium and rosuvastatin calcium dispersible tablet
[0101] Name of raw material
Specification 1 Dosage
Specification 2 Dosage
Specification 3 Dosage
4.0g
6.0g
8.0g
20.0g
40.0g
80.0g
25.0g
50.0g
100.0g
(Based on rosuvastatin)
5.0g
10.0g
40.0g
165g
150g
145g
18.5g
18.5g
18.5g
Polysorbate 80
4.5g
4.5g
4.5g
0.6g
0.6g
0.6g
Micropowder silica gel
1.5g
1.5g
1.5g
35g
35g
35g
sodium starch glycolate
8.5g
8.5g
8.5g
Crospovidone
8g
8g
8g
95% ethanol...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More