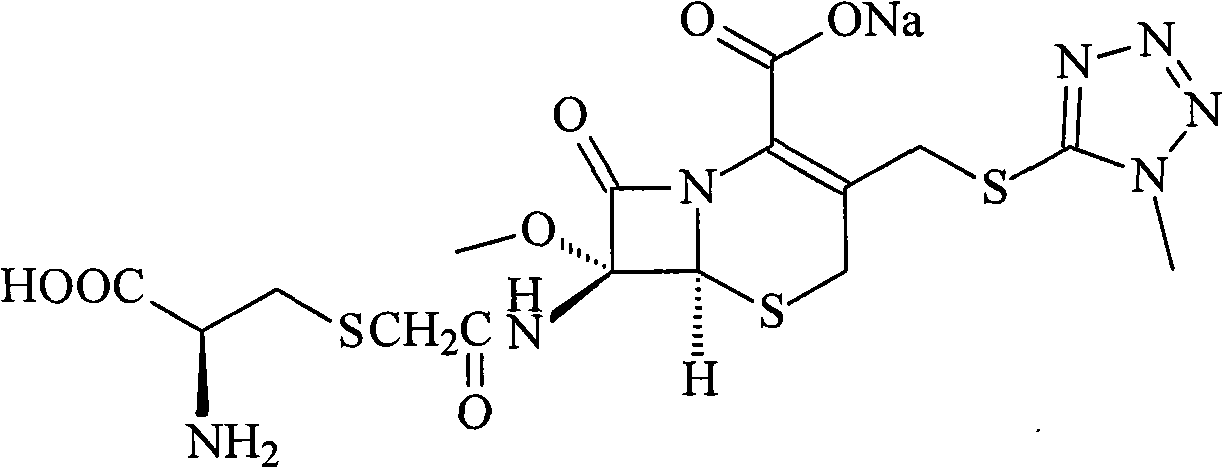
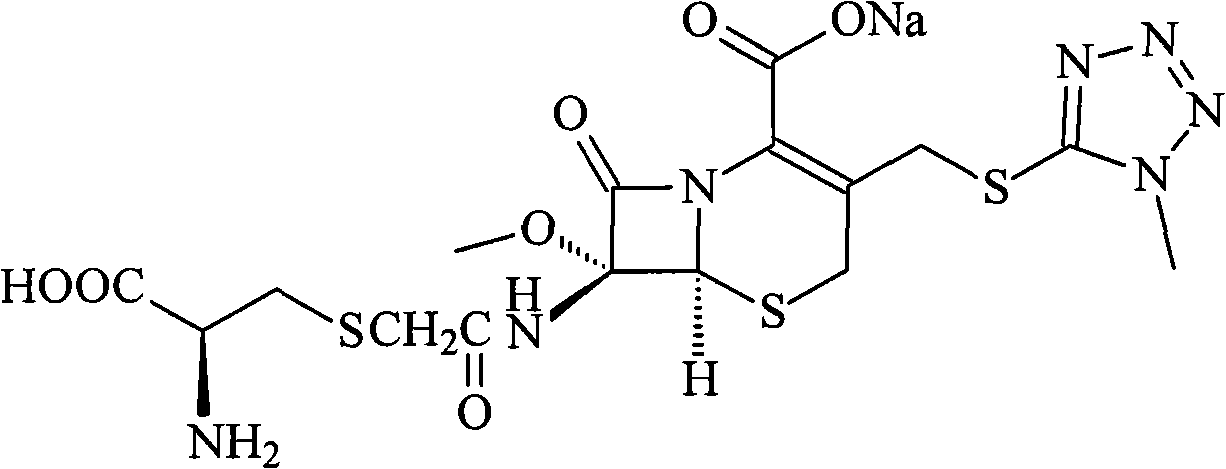
Cefminox sodium compound and new preparation method thereof
A technology of cefminox sodium and its compound, which is applied in the field of medicine, can solve the problems of large related impurities, large toxic and side effects of preparations, and low purity, and achieve the effects of low cost, reduced toxic and side effects, and high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0026] The refining of embodiment 1 cefminox sodium
[0027] (1) First dissolve 100g of cefminox sodium in 600ml of water, slowly add 200ml of 1mol / L hydrochloric acid solution, stir and react for 30min, gradually precipitate a large amount of solid, filter, wash the filter cake with 100ml of purified water, and vacuum dry at 40°C for 4 hours ;
[0028] (2) Dissolve the dry solid obtained in step (1) in 800ml of ethanol, add 0.8g of activated carbon, stir and adsorb at 65°C for 15min, filter for decarburization, and obtain the filtrate;
[0029] (3) The filtrate is separated and purified by a chromatographic column, using isopropanol and acetonitrile as a mixed solvent mobile phase with a volume ratio of 2:3, the stationary phase filler is silica gel, the flow rate is 2.8ml / min, and the column temperature is 35° C. to collect the filtrate;
[0030] (4) Add 10% sodium carbonate solution to the filtrate obtained in step (3), stir and react for 40min, separate out insoluble soli...
Embodiment 2
[0036] The refining of embodiment 2 cefminox sodium
[0037] (1) Dissolve 100g of cefminox sodium in 1000ml of water earlier, slowly add 150ml of 10% citric acid solution, stir and react for 60min, a large amount of solids are gradually precipitated, filter, the filter cake is washed with 100ml of purified water, and vacuum-dried at 50°C for 8 Hour;
[0038] (2) Dissolve the dry solid obtained in step (1) in 1000ml of isopropanol, add 5g of activated carbon, stir and adsorb at 60°C for 30min, filter and decarburize to obtain the filtrate;
[0039] (3) The filtrate is separated and purified by a chromatographic column, with a volume ratio of 2:3 isopropanol and acetonitrile as a mixed solvent mobile phase, the stationary phase packing is alumina, the flow rate is 3.2ml / min, and the column temperature is 35°C, collect the filtrate ;
[0040] (4) Add 10% sodium citrate solution to the filtrate obtained in step (3), stir and react for 20min, precipitate insoluble solids, and fil...
Embodiment 3
[0046] The refining of embodiment 3 cefminox sodium
[0047] (1) First dissolve 100g of cefminox sodium in 800ml of water, slowly add 100ml of 1mol / L phosphoric acid solution, stir for 50min, gradually precipitate a large amount of solid, filter, wash the filter cake with 100ml of purified water, and vacuum dry at 45°C for 6 hours ;
[0048] (2) Dissolve the dried solid obtained in step (1) in 800ml of dichloromethane, add 2.4g of activated carbon, stir and adsorb for 20min, filter for decarburization, and obtain the filtrate;
[0049] (3) The filtrate is separated and purified by a chromatographic column, using isopropanol and acetonitrile as a mixed solvent mobile phase with a volume ratio of 2:3, the stationary phase filler is silica gel, the flow rate is 4.2ml / min, and the column temperature is 34° C. to collect the filtrate;
[0050] (4) Add 10% sodium acetate solution to the filtrate obtained in step (3), stir and react for 30min, separate out insoluble solid, filter; ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More