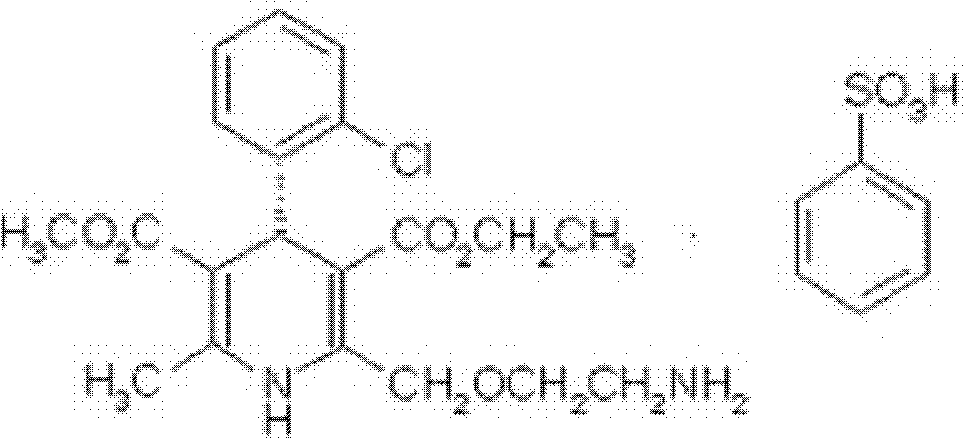A kind of levamlodipine besylate crystal, its preparation method and a new pharmaceutical composition containing the crystal
A technology of levamlodipine besylate and a composition, which is applied in the field of levamlodipine besylate crystals, can solve the problems of slow onset of drug effect, no solubility, low overall level of blood drug concentration, etc., and achieves enhanced curative effect. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0083] [embodiment 1] preparation of levamlodipine besylate crystal
[0084] 1) dissolving levamlodipine besylate in a mixed solvent of dichloromethane and ethanol to obtain a dichloromethane / ethanol solution of levamlodipine besylate;
[0085] 2) Add n-heptane dropwise to the dichloromethane / ethanol solution of levamlodipine besylate obtained in step 1) under an ultrasonic field until crystallization occurs;
[0086] 3) Turn off the ultrasonic field, let stand, filter, wash the filter cake with dichloromethane and ethanol respectively, and dry to obtain the levamlodipine besylate crystal.
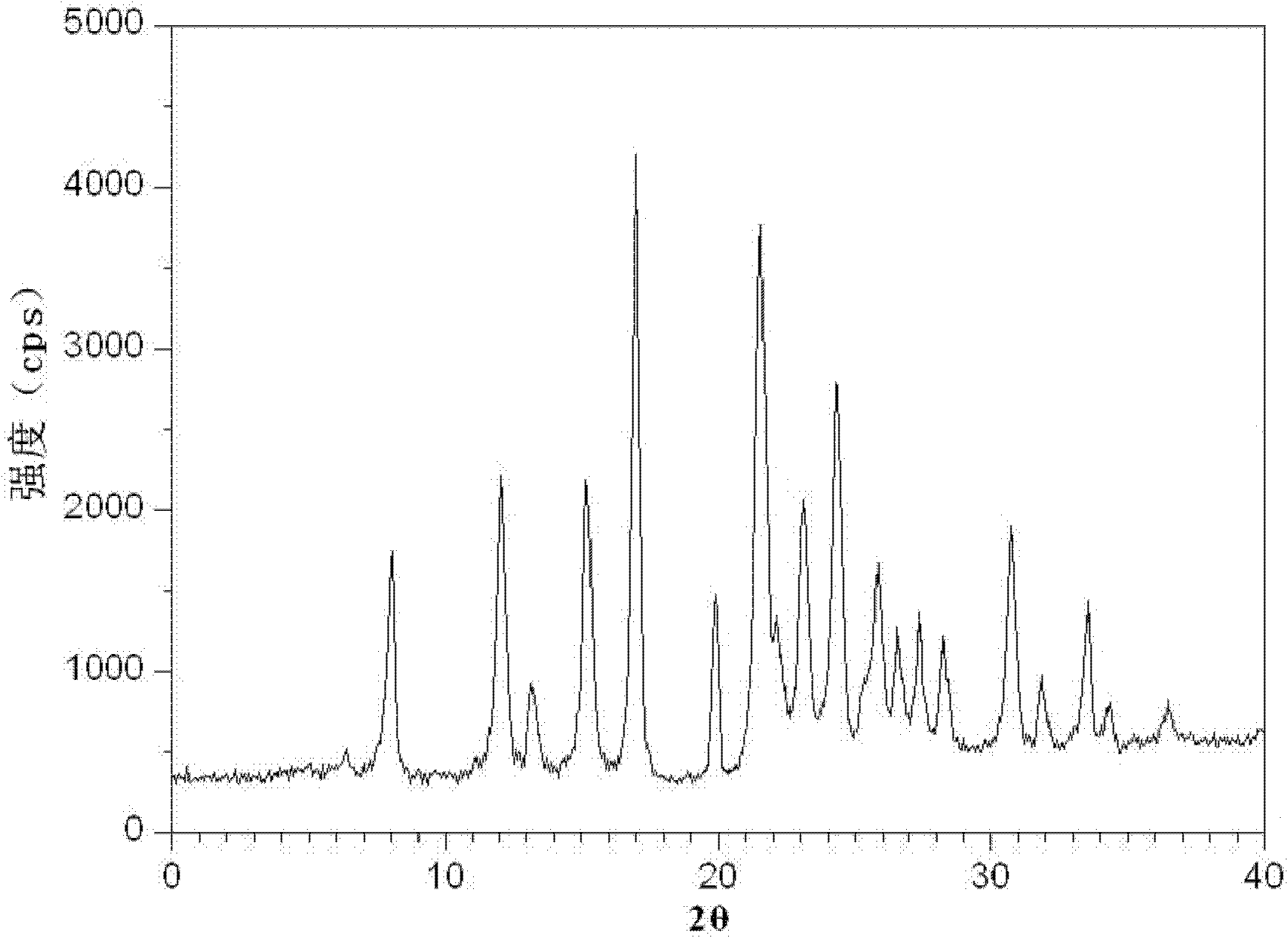
[0087] Gained levamlodipine besylate crystals use Cu-Kα rays to measure characteristic peaks in the X-ray powder diffraction pattern obtained at 2θ of 8.0°, 12.1°, 15.4°, 17.0°, 19.8°, 21.6°, 23.0° , 24.3°, 25.7°, 27.4°, 30.7° and 33.5° display, such as figure 1 shown.
[0088] Below is embodiment 2-9, and preparation method is with embodiment 1, and its concrete process parameter is sh...
preparation Embodiment 1
[0094] [Preparation Example 1] Levoamlodipine Besylate Tablets
[0095] 1. Prescription
[0096]
[0097] 2. Preparation process:
[0098] (1) Microcrystalline cellulose, sodium starch glycolate and magnesium stearate were baked at 60°C for 2 hours respectively, passed through a 60-mesh sieve, and set aside;
[0099] (2) Take above-mentioned standby microcrystalline cellulose, carboxymethyl starch sodium and magnesium stearate by recipe quantity, adopt equal amount incremental method to mix, obtain mixed powder;
[0100] (3) take by weighing the levamlodipine besylate crystal prepared in Example 1 of the prescription amount, mix with the mixed powder obtained in step 3), obtain the pharmaceutical composition powder, and take a sample for detection;
[0101] (4) The obtained pharmaceutical composition powder is subjected to direct powder compression to obtain the pharmaceutical composition.
[0102] (5) Determine the content of the main drug, calculate the weight of the t...
preparation Embodiment 2
[0103] [Preparation Example 2] Levoamlodipine Besylate Tablets
[0104] 1. Prescription
[0105]
[0106] 2. Preparation process: the same as in the preparation example 1, the difference is that the levamlodipine besylate crystals are the levamlodipine besylate crystals prepared in Example 2, and step 2) is baked at 65°C for 3 hour, through a 80-mesh sieve.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Diameter | aaaaa | aaaaa |
| Weight | aaaaa | aaaaa |
| Hardness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More