Anti-human serum albumin single-chain antibody and method for connecting polypeptide medicine with nitrogen terminal of antibody
A single-chain antibody and drug technology, applied in the direction of anti-animal/human immunoglobulins, pharmaceutical formulations, peptides, etc., can solve the problem of affecting the biological activity of polypeptide biological drugs, increase the molecular weight of polypeptide biological drugs, and change the pharmacokinetics of drugs. Chemical characteristics and other problems, to achieve the effect of strong product uniformity, easy large-scale production, and small molecular weight
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
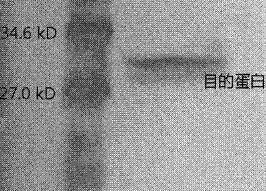
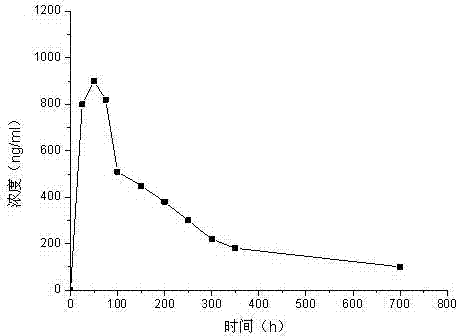
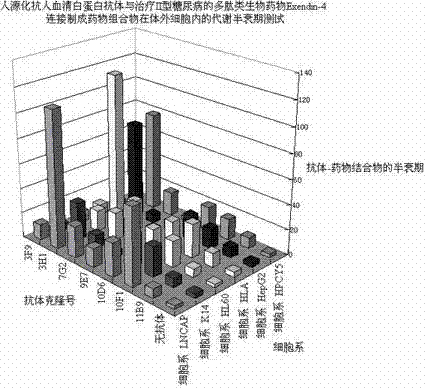
Image
Examples
Embodiment 1
[0026] Example 1, obtaining the single-chain antibody in the present invention:
[0027] 1. Construction of human antibody library
[0028] Screen the variable region sequences of antibodies with specific recognition ability for human serum albumin from the humanized single chain antibody library. The gene fragment is amplified by polymerase chain reaction and spliced into the phage vector. The primers are as follows. An example of primers used to find the light chain CDR: Forward primer [5'-GATATNNAANTTANNNAATNN-3'] Reverse primer [5'-NNNTTTATTTNTTANNTTGG-3'], an example of primers for searching heavy chain CDR: forward primer [5'-GAAGTNNAGNTGNTNG-3'], reverse primer [5'-NGAAGAGANTGTGANTAGNGT-3'].
[0029] Antibody molecular fragments are expressed by phage and displayed on the surface of phage, and human serum albumin (HSA) is used as an antigen to screen the antibodies displayed on the surface of phage to obtain the antibody with the best specificity.
[0030] 2. Antibo...
Embodiment 2
[0036] Example 2, Analysis of the coding sequence of the single-chain antibody, construction of recombinant and expression vectors:
[0037]Propagate the 10D7 strain produced in Example 1 in LB culture medium, then use a DNA extraction kit to purify the plasmid in the cell line, and separate it by restriction endonuclease cutting and 2% agarose gel electrophoresis to obtain The coding sequence of the variable region of the heavy chain, the coding sequence of the variable region of the light chain was obtained by the same method, and the human antibody variable region universal primers were used for PCR amplification, and then the obtained amplification products were sent to Eurofin for sequencing. The results of the obtained DNA sequence and protein sequence are shown in Table 2.1 and Table 2.2, wherein the sequence listed in Table 2.1 is the light chain region (N-SEQ-04) of the humanized anti-human serum albumin single chain antibody, and Its variable region sequence (N-SEQ-0...
Embodiment 3
[0040] Example 3, linking the light and heavy chain sequences of the single-chain antibody and exendin-4, a polypeptide biological drug for the treatment of type 2 diabetes, to form a drug combination:
[0041] The DNA of the light chain region and the heavy chain region of the antibody whose sequence has been determined is linked by direct synthesis or recombinant PNR. The hinge part of the VH chain region of the single-chain antibody has the DNA sequence corresponding to SEQ ID No.2. The recombinant The operation method of PNR is a conventional method commonly used in this field. Sequencing the sequence synthesized by Invitrogen is exactly the same as the sequencing result of the link sequence of antibody light chain and heavy chain completed by our method of recombining PNR. The sequencing result is shown in the table 3.1 shows:
[0042] GAT ATC CAG CTT ACC CAA AGT CCT GCT TTC From [VL] 5 10 ATG GCC GCA AGC ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| absorbance | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More