Applications of stilbene compounds
A compound and drug technology, applied in the field of medicine, can solve problems such as poor curative effect and aggravated disease
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
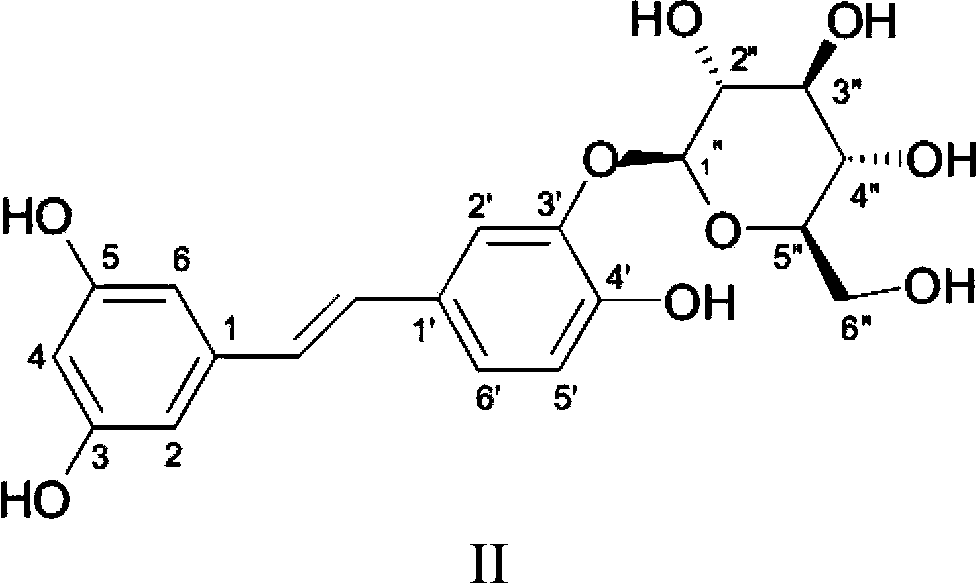
[0022] Embodiment 1: the preparation of trizastilbene glycoside
[0023] Materials: Lhasa rhubarb medicinal material (purchased in Lhasa, Tibet, batch number 20110308, content 4.8%), remove impurities, clean, crushed into coarse powder; D101 macroporous adsorption resin (Cangzhou Baoen Adsorption Material Co., Ltd.) pretreated according to the instructions .
[0024]Preparation: Take 1Kg of Lhasa rhubarb crude powder, calculated as Kg / L, add 8 times the volume (W / V) of ethanol to extract 3 times, 2 hours / time, filter, combine the filtrates, concentrate until there is no alcohol smell, add 1L of water, mix Evenly, leave for 24 hours. Take about 900mL of the supernatant, put it on the D101 macroporous adsorption resin (the resin column is about 2000mL), elute with 3 times the column volume of water (V / V), and then use 6 times the column volume of 25% ethanol (V / V V) Elution, collect 0.5-6 times the ethanol eluent, concentrate to dryness, add 100mL water, mix well, and crystall...
Embodiment 2
[0026] Embodiment 2: the inspection of tristilbene raw material
[0027] Take an appropriate amount of stilbeneside (No. 20110401) prepared in Example 1, and conduct an inspection according to the regulations under the item of U injection in Appendix I of "Chinese Pharmacopoeia".
[0028] Properties: Off-white powder with bitter taste.
[0029] Loss on drying: According to the provisions of Appendix IX G of the 2010 edition of "Chinese Pharmacopoeia", trizastilbene glycosides were taken, and phosphorus pentoxide was used as a desiccant, and dried under reduced pressure at room temperature to constant weight, and the weight loss was 1.2%.
[0030] Residue on ignition: According to the provisions of Appendix IX J of the 2010 edition of "Chinese Pharmacopoeia", take an appropriate amount of trizastilbeside for inspection, and the residue on ignition is 0.1%.
[0031] Related substances: take an appropriate amount of tristilbene, according to the provisions of Appendix IX S of th...
Embodiment 3
[0042] Example 3: Acute Toxicity Test of Trizastilbene
[0043] 1. Experimental animals
[0044] 50 SPF grade ICR mice (provided by the Experimental Animal Center of Kunming Pharmaceutical Group), weighing 18-22 g, half male and half male. The pre-experiment animals were acclimatized indoors for one week at a room temperature of 18-25°C. They were housed in rack cages and fed with standard feed.
[0045] 2. Preparation of test drug
[0046] The tristilbene (No. 20110401) prepared in Example 1 was prepared into a 40 mg / mL tristilbene test drug solution with relevant excipients. 0.45um microporous membrane filtration for standby.
[0047] 3. Experimental methods and results
[0048] 50 mice were weighed and marked, and randomly divided into 5 different dosage groups of trizaperoside (10 mice in each group). According to the results obtained in the pilot experiment, the 0% (Dn) and 100% (Dm) lethal doses of the tested drug trizaperone were 0.51 g and 1.13 g, respectively. ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More