Complex containing 2-aminopyridine tetradentate ligand and application thereof
A technology of tetradentate ligands and metal complexes, which is applied in the preparation of hydroxyl compounds, organic compound/hydride/coordination complex catalysts, compounds containing elements of Group 8/9/10/18 of the periodic table, etc., can Solve the problems of low catalytic efficiency and high cost of catalyst use, and achieve the effects of convenient use, strong stability and high activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
specific example
[0082]
[0083]
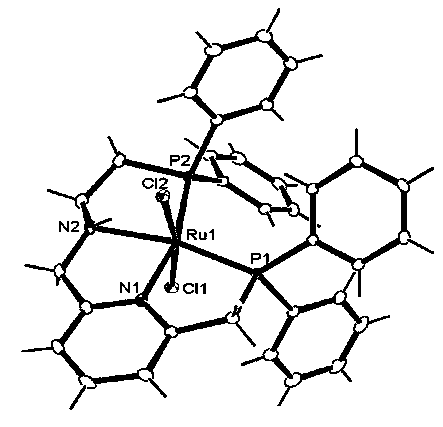
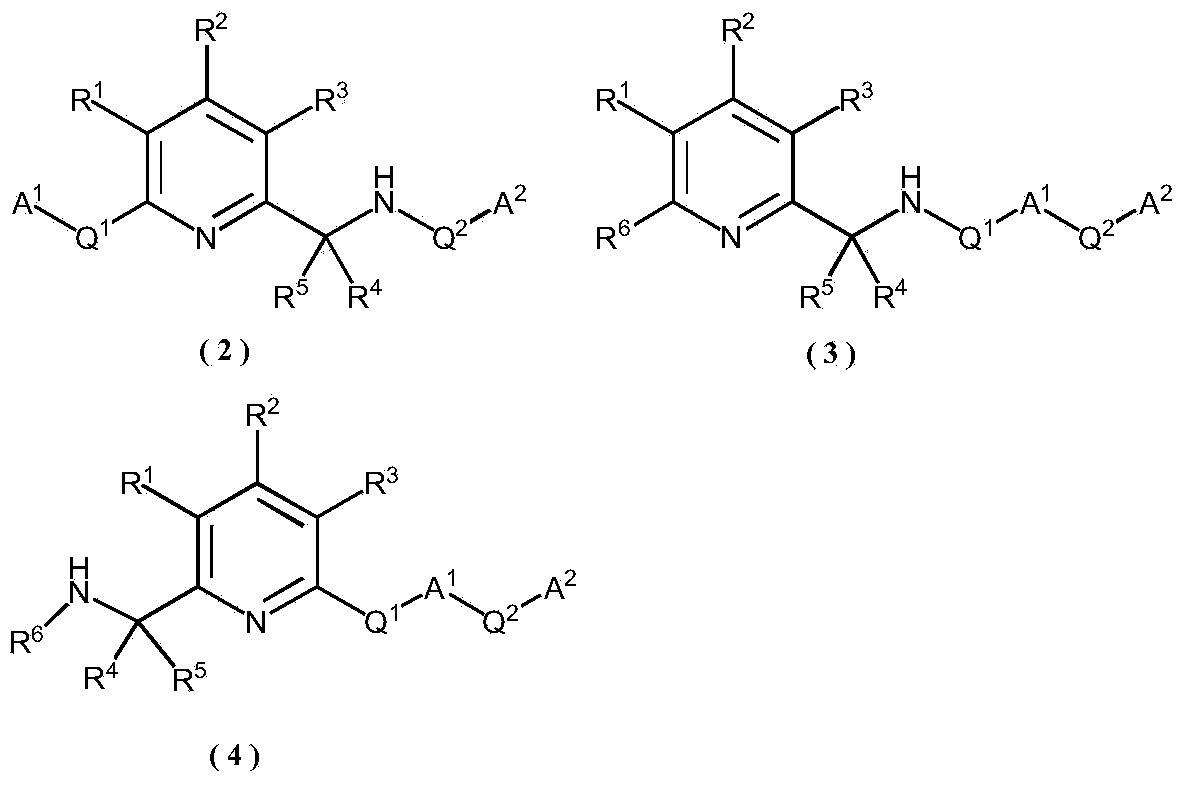
[0084] The metal M in the general formula (1) is described. The metal M can be exemplified as metal elements of the sixth, seventh, and eighth subgroups, preferably the metal elements of the sixth subgroup Fe, Ru, and Os, and more preferably metal Ru.
[0085] The metal compound used as the starting material for the preparation of the metal complex in the present invention is not particularly limited, for example, FeCl 2 , FeBr 2 , FeCl 3 , FeBr 3 、RuCl 3 Hydrate, RuBr 3 Hydrate, RuI 3 Inorganic compounds such as hydrates, RuCl 2 (DMSO) 4 ,, [Ru(cod)Cl 2 ] n , [Ru(nbd)Cl 2 ] n 、(cod)Ru(2-methallyl) 2 , [Ru(benzene)Cl 2 ] 2 , [Ru(p-cymene)Cl 2 ] 2 , [Ru(mesitylene)Cl 2 ] 2 , [Ru(hexamethylbenzene)Cl 2 ] 2 、RuCl 2 (PPh 3 ) 3 , RuHCl (PPh 3 ) 3 、RuH(OAc)(PPh 3 ) 3 、RuH 2 (PPh 3 ) 4 Wait. In the above example, DMSO is dimethyl sulfoxide, cod is 1,5-cyclooctadiene, nbd is norbornadiene, and Ph is phenyl.
[0086] The metal comp...
Embodiment 1
[0104] The preparation of 6-formyl 2-pyridinecarboxylic acid methyl ester, synthetic reaction formula and steps are as follows:
[0105]
[0106] Put 11.5g (50mmol) of ethyl 2,6-pyridinedicarboxylate into a 500ml dry flask, add 400ml of methanol, place the reaction flask in an ice bath at 0°C, and slowly add 1.9g (50mmol) of NaBH 4 , continue to react at this temperature for 1h, slowly add 1g NaBH again 4 , react at 0°C for 1h, add 0.5g NaBH again 4 , react at 0°C for 1h, add 200ml of saturated sodium bicarbonate solution, stir at 0°C for 30min, spin dry the methanol, extract the obtained aqueous phase with chloroform (100ml×5), dry the organic phase with anhydrous sodium sulfate, and spin dry to obtain a white solid . Dissolve the white solid in 300ml of dichloromethane, slowly add 12.9g (60mmol) PCC (pyridinium chlorochromate) at room temperature, stir at room temperature for 4h, filter the obtained black solution with diatomaceous earth, wash with dichloromethane, The...
Embodiment 2
[0108] The preparation of 6-dimethoxymethyl-2-pyridinecarboxylic acid methyl ester, synthetic reaction formula and steps are as follows:
[0109]
[0110] Put 2.4g (14.5mmol) of methyl 6-formylpyridine-2-carboxylate into a 250ml dry flask, add 50ml of anhydrous methanol and 50ml of trimethyl orthoformate successively under a nitrogen atmosphere, heat to 70°C and stir for 4h, After the reaction was detected by TLC, it was cooled to room temperature and spin-dried until the remaining about 5ml of liquid was added, and 50ml of water was added, and the product was extracted three times with ethyl acetate. The obtained organic phase was dried and spin-dried, and the crude product was purified by column chromatography to obtain 2.9 g white solid, yield90%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More