Solid-phase PCR (Polymerase Chain Reaction) kit for gene detection of precision drug utilization of cardiovascular and cerebrovascular diseases
A technology for cardiovascular and cerebrovascular diseases and precise drug use, applied in the field of PCR application, can solve the problems of hindering the expansion of thrombus and no thrombolytic effect, and achieve the effects of low cost of reagent consumables, improved detection sensitivity and specificity, and simple operation.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 11
[0023] Example 1 Composition of solid-phase PCR detection kit for precision medicine of 13 kinds of cardiovascular and cerebrovascular diseases
[0024]The kit consists of a PCR reaction mixture and 12 strip tubes or a 96-well PCR reaction plate, in which the PCR reaction mixture is 5×PCRBuffer (Tris-HCl pH8.5 100mM, KCl 500nM, MgCl 2 15nM, dNTPs10mM), Mg 2+ , 20 pairs of primers and hot start DNA polymerase.
[0025] The kit is to seal a layer of paraffin wax mixture on the surface of the above PCR reaction mixture, and the paraffin wax mixture is formed by mixing solid paraffin and liquid paraffin at a ratio of 1:8 (W / V).
[0026] The reaction system (25ul) of the kit is: PCR reaction solution 23ul, template 2ul.
Embodiment 2
[0027] Operation and result determination of embodiment 2 kit
[0028] 1. Genomic DNA extraction
[0029] The exfoliated cells on the inner surface of the human oral cavity were extracted with oral swabs, and DNA was extracted with an oral swab genomic DNA extraction kit (Tiangen Biochemical Technology, item number: DP322) according to the kit instructions.
[0030] 2. Preparation of reaction system
[0031] Take the 12-tube strip tube or 96-well PCR reaction plate of the Solid-Phase PCR Reaction Kit for Genetic Detection of Precise Medicine for Cardiovascular and Cerebrovascular Diseases, and add 2ul of the extracted DNA.
[0032] 3. PCR amplification
[0033] Put the PCR tube into an ordinary PCR machine and perform PCR amplification according to the instructions recommended.
[0034] 4. Analysis of PCR products by 1.0% agarose gel electrophoresis
[0035] 5. Interpretation of results
[0036] The result interpretation standard of this kit is as follows Table 1
[0037...
Embodiment 3
[0038] Example 3 Interpretation of sequencing results and medication guidance
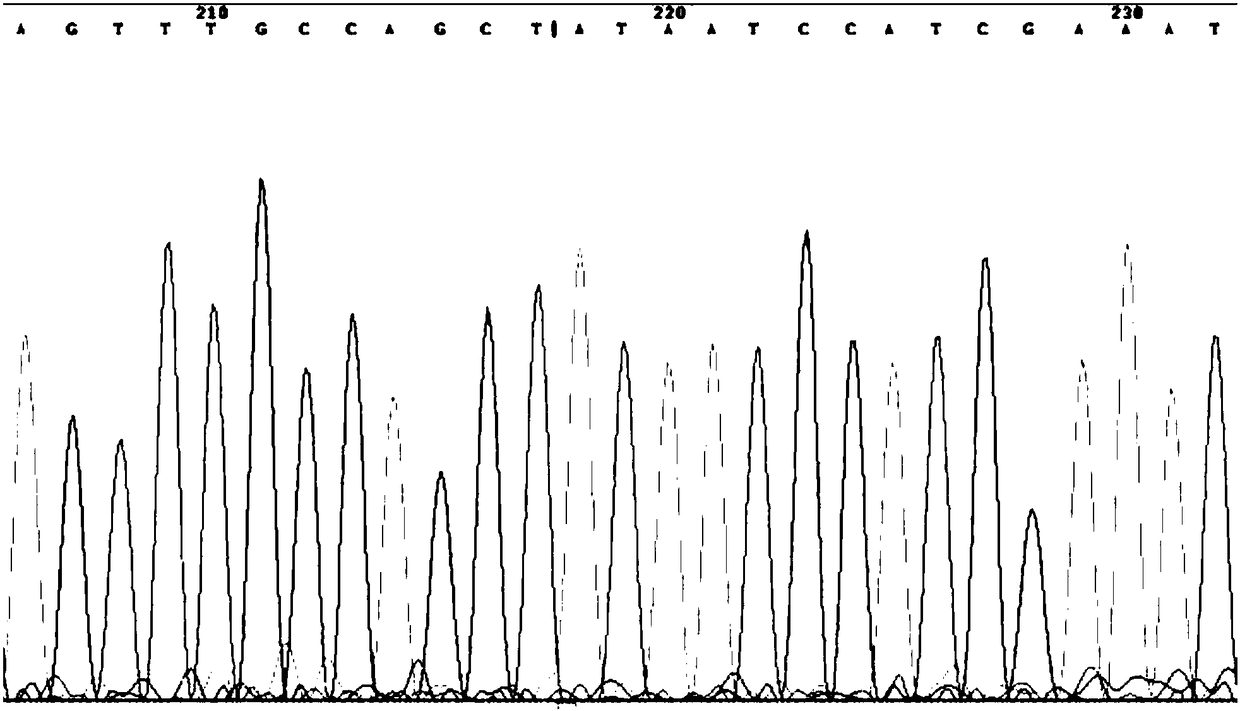
[0039] Using the DNA extracted from oral swabs of patients with cardiovascular and cerebrovascular diseases in Example 1 as a template, after amplification with the above kit, the following sequencing results were obtained, and the corresponding medication instructions were given as follows according to the sequencing results:
[0040]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More