Preparation method of flunixin meglumine
A technology of flunixin meglumine and flunixin, which is applied in the field of preparation of flunixin meglumine, can solve the problems of high difficulty in recovery, purification and utilization, and increased production costs, and achieve low cost and short reaction time , the effect of high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
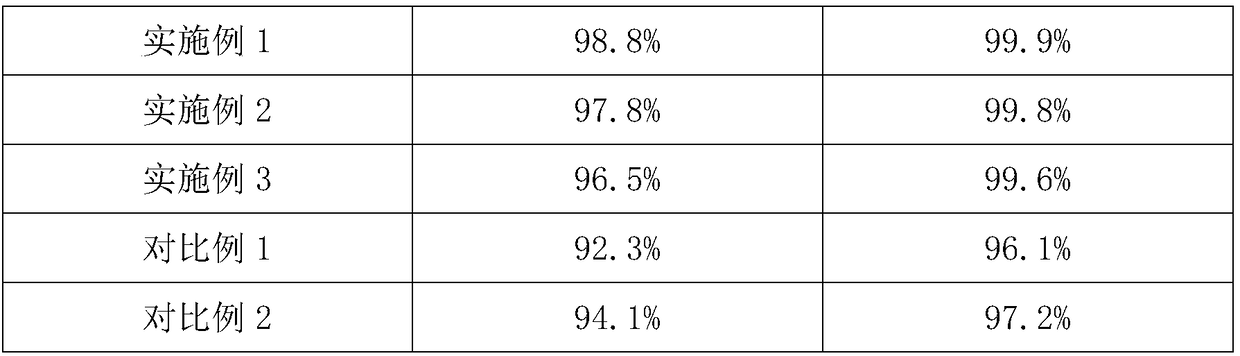
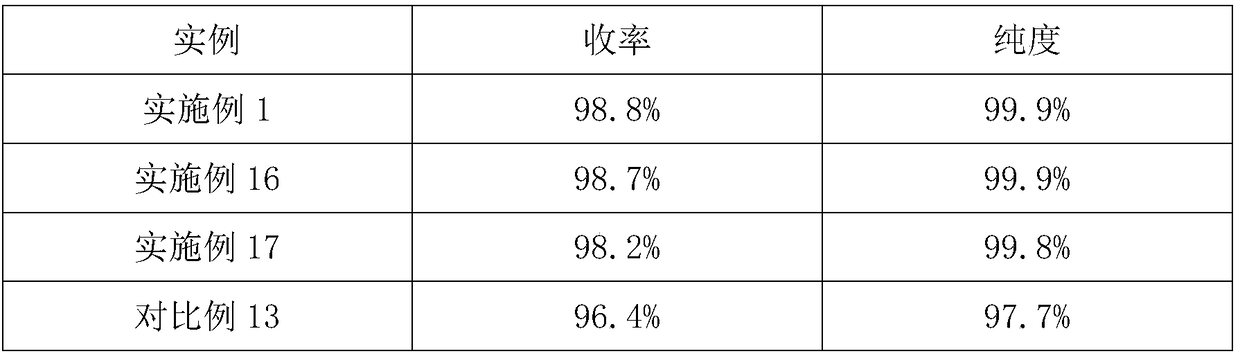
Embodiment 1
[0024] The preparation method of flunixin meglumine in the present embodiment comprises the following steps:
[0025] (1) Add 2-chloronicotinic acid and 2-methyl-3-trifluoromethylaniline into aqueous sodium hydroxide solution and stir, and the 2-chloronicotinic acid and 2-methyl-3-trifluoromethylaniline The molar ratio of aniline is 3:1; the molar ratio of the 2-chloronicotinic acid to the sodium hydroxide used to prepare the aqueous sodium hydroxide solution is 1:2; add ethylene glycol and chain polyethylene glycol dialkyl Ether and catalyst A, the consumption of described chain polyethylene glycol dialkyl ether is 3% of the feeding quality of 2-chloronicotinic acid; Described catalyst A is p-toluenesulfonic acid and copper oxide, and described p-toluenesulfonic acid The molar ratio of toluenesulfonic acid to copper oxide is 3:1; control the temperature at 45°C, adjust the pH of the solution after reacting for 0.6 hours, leave to separate after stirring, filter after stirring...
Embodiment 2
[0028] The difference from Example 1 is that the phase transfer catalyst in the step (1) is benzyltriethylamine chloride.
Embodiment 3
[0030] The difference with embodiment 1 is: the phase transfer catalyst in the described step (1) is tetrabutylammonium bisulfate.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com