A kind of synthetic method for preparing 5-bromo-2-chloro-4'-ethoxydiphenylmethane
A technology for the synthesis of ethoxydiphenylmethane, which is applied in the direction of condensation preparation of carbonyl compounds, ether preparation, halogenated hydrocarbon preparation, etc., can solve the problems of high cost of raw materials, many auxiliary materials and solvents, which are not conducive to production, and achieve Easy to obtain, low equipment requirements, and low cost effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
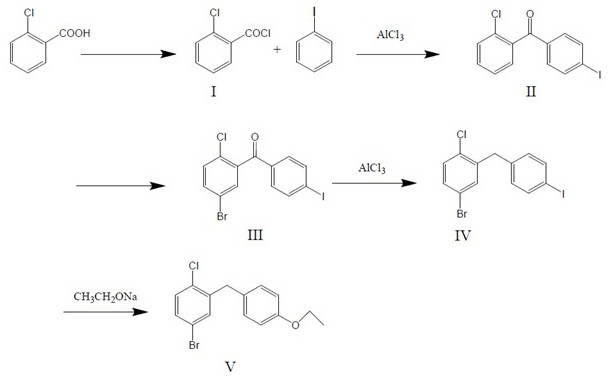
[0032] Such as figure 1 Shown, during concrete implementation, the synthetic method that the present invention prepares 5-bromo-2-chloro-4'-ethoxydiphenylmethane comprises:
[0033] The first step, get o-chlorobenzoic acid, first add solvent and catalyzer, then drip acid chloride reagent, obtain o-chlorobenzoyl chloride ( figure 1 Compound I); Wherein, the weight ratio of o-chlorobenzoic acid and solvent is 1:4.0-6.0, the weight ratio of o-chlorobenzoic acid and catalyst is 1:0.003-0.006, the molar ratio of o-chlorobenzoic acid and acyl chloride reagent It is 1:1.05-1.15.
[0034] Specifically, after adding solvent and catalyst, stir evenly, add acid chloride reagent dropwise at 0°C-10°C; react at 20°C-30°C for 2-4 hours; depressurize at 40°C-70°C after the reaction The solvent is recovered by distillation; the solvent is dichloroethane, toluene or dichloromethane (preferably dichloromethane); the catalyst is pyridine or DMF (preferably pyridine); the acyl chloride reagent i...
Embodiment 1
[0045] This embodiment is for the preparation of o-chlorobenzoyl chloride ( figure 1 Compound I).
[0046] The basic process of this embodiment is the first step of the technical solution of the present invention during specific implementation.
[0047] Some specific parameters of this embodiment are as follows:
[0048] Add 156.0g (1.0mol) of o-chlorobenzoic acid, 800g of dichloromethane, and 0.8g of DMF into a 2000ml four-necked reaction flask, start stirring, cool down to 10°C, add 139.7g (1.1mol) of oxalyl chloride dropwise, and drop to room temperature (20°C-30°C) react for 3 hours (up to 0.2% of raw materials detected by HPLC), evaporate the solvent to obtain an oily substance, mainly o-chlorobenzoyl chloride, and a small amount of oxalyl chloride; the oily substance is detected by HPLC The purity is 99.0%. The purity is calculated according to the HPLC area normalization method, and the solvent peak is deducted during the calculation. Simultaneously, because oxalyl ch...
Embodiment 2
[0050] This embodiment is for the preparation of 2-chloro-4'-iodobenzophenone ( figure 1 compound II).
[0051] The basic process of this embodiment is the second step of the technical solution of the present invention during specific implementation.
[0052] Some specific parameters of this embodiment are as follows:
[0053] Add the oil obtained in Example 1 (containing 172.4 g (0.985 mol) of o-chlorobenzoyl chloride) and 350.0 g of dichloromethane into a 1000 ml four-necked reaction flask, protect with nitrogen, cool down to 0°C, and add aluminum trichloride in batches 144.6g (1.08mol), add 205.0g (1.00mol) of iodobenzene dropwise, react at 0°C-5°C for 2 hours (until the detection of raw materials by HPLC<0.3%), pour into ice water after the end (control the addition temperature <20°C) , Since aluminum trichloride is poured into water, it will exotherm violently, and if the temperature is too high, the impurity content will be too high, so the temperature should be contro...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More