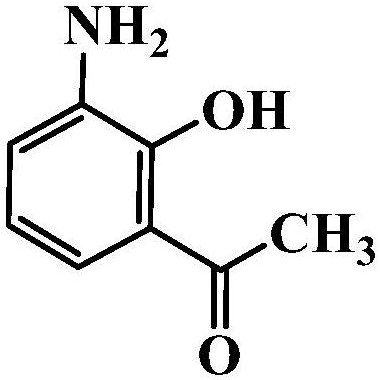
Method for preparing 2-hydroxy-3-aminoacetophenone
A technology of aminoacetophenone and hydroxyacetophenone, applied in chemical instruments and methods, preparation of organic compounds, organic chemistry, etc., can solve the problem of low yield and purity of target products, difficult separation of by-products, poor reaction selectivity, etc. problem, to achieve the effect of high sulfonation conversion rate, high product yield and controllable reaction
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0048] The preparation of embodiment 1 HoNPE
[0049] Preparation of mixed acid: Mix 0.32g (5.25mmol) of nitric acid and 1.03g (10.51mmol) of sulfuric acid in an ice-water bath and set aside.
[0050] Add 0.68g (4.99mmol) of 2-hydroxyacetophenone into a 100mL flask, and add 0.87g (7.47mmol, 1.5eq) of chlorosulfonic acid dropwise under stirring at 0°C to 5°C. After 15 minutes of reaction, almost no bubbles are generated, and it turns into React at room temperature for 30 minutes, then gradually raise the temperature to 80°C for 1 hour. The temperature was lowered to below 20°C, and the prepared mixed acid was added dropwise to the reaction system, and the stirring reaction was continued for 1 h after the dropwise addition was completed. Add 20 g of crushed ice to the system to quench the reaction, add about 0.05 g of urea to remove nitric acid that may remain in the reaction, gradually raise the temperature to reflux to hydrolyze the sulfonic acid group, and monitor the reacti...
Embodiment 2
[0056] The preparation of embodiment 2 HoAPE
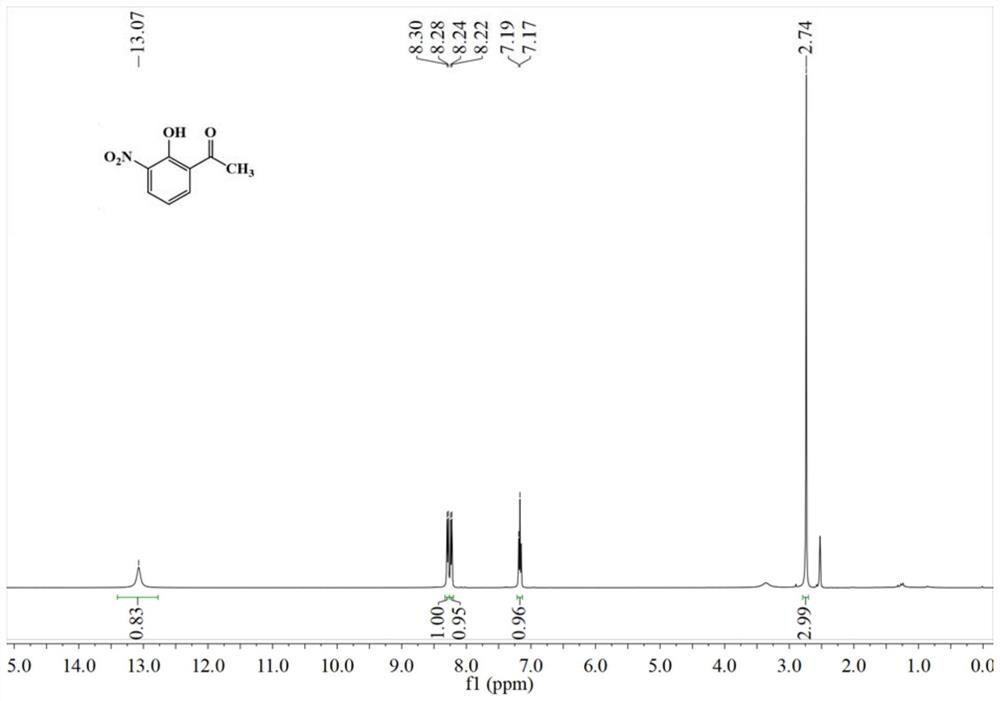
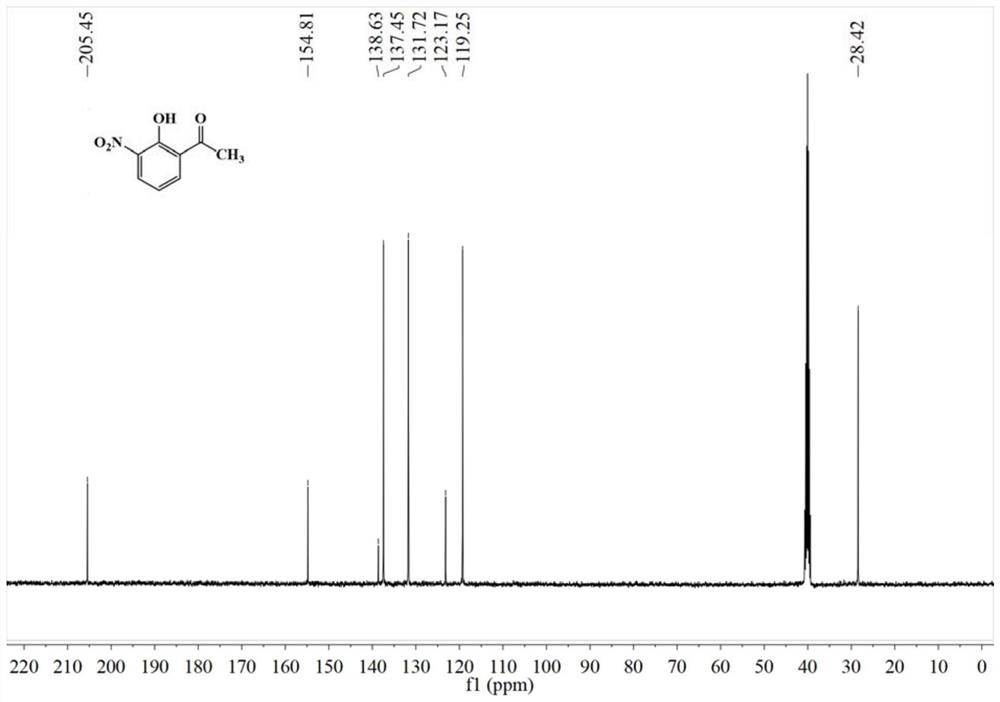
[0057] 0.91g (5.02mmol) of HoNPE and 2.81g (50.31mmol, 10eq) of reduced Fe powder were added to a 50mL flask. 15 mL of concentrated HCl was added to the reaction flask with stirring. When the reaction is stable, heat to reflux for 1 h and stop the reaction. After standing at room temperature, a large amount of white solid precipitated out. After freezing, it was suction-filtered, and the solid was washed with saturated sodium chloride solution to obtain HoAPE hydrochloride as a white solid. Add a small amount of water and ethyl acetate to the solid, add 1mol / L NaOH solution dropwise to adjust the pH of the solution to neutral, separate the layers, dry the ethyl acetate layer with anhydrous sodium sulfate, and distill off the ethyl acetate under reduced pressure to obtain HoAPE crude product, HPLC purity 96.33%, the resulting crude product was recrystallized from methanol / water to obtain 0.66 g of a tan solid, with a yield of 86....
Embodiment 3
[0063] Preparation of Example 3 HoNPE
[0064] Add 0.68g (4.99mmol) of 2-hydroxyacetophenone into a 100mL flask, add 4.90g (50.00mmol, 10eq) of concentrated sulfuric acid dropwise under stirring at 0°C, and react at 0-5°C until no bubbles emerge, about 15min Gradually raise the temperature to 110°C for 1 hour, cool down, add a mixed acid consisting of 0.32g (5.25mmol) of concentrated nitric acid (5.25mmol) and 1.47g (15.00mmol) of concentrated sulfuric acid below 20°C, stir at room temperature for 1 hour, add 20g of crushed ice to quench the reaction, add 0.05g urea to remove the remaining nitric acid from the reaction, heat and reflux until the sulfonic acid group is completely hydrolyzed, cool and extract with methyl acetate, dry the ester layer with anhydrous sodium sulfate, spin the solvent to obtain the crude HoNPE, decolorize and remove impurities with activated carbon and diatomaceous earth , ethylene glycol recrystallization to obtain product 0.45g. Yield 49.7%.
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More