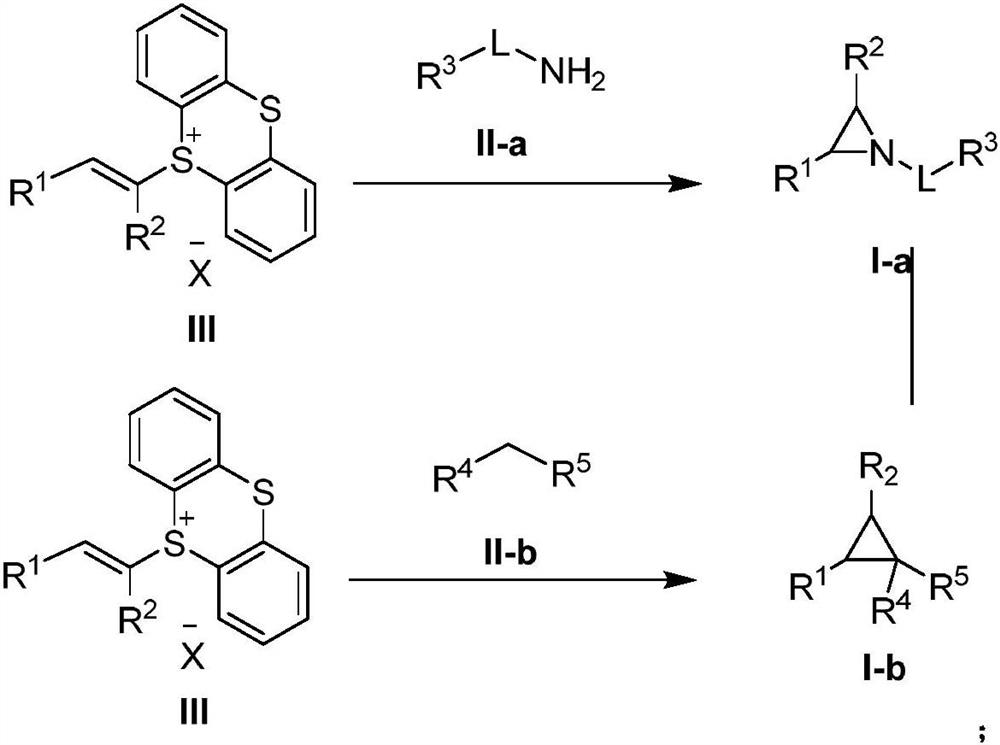
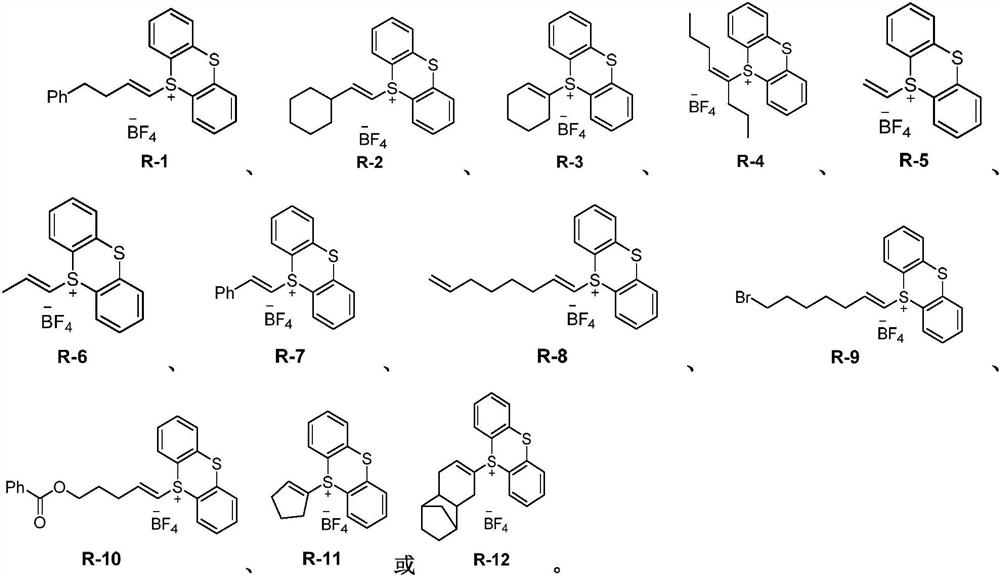
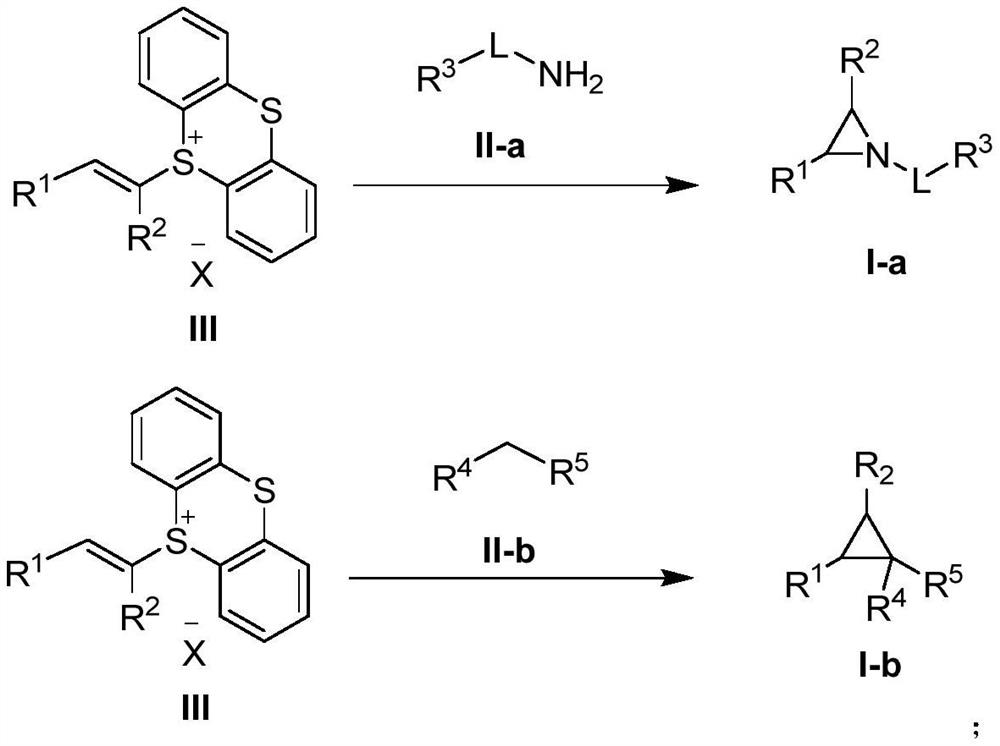
Three-membered ring preparation method without metal catalysis
A three-membered ring and compound technology, which is applied in the field of metal-free catalyzed three-membered ring preparation, can solve problems such as limitations
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0101]
[0102] First p-toluenesulfonamide (41.1mg, 0.24mmol), K 2 CO 3 (27.6 mg, 0.20 mmol) and compound R-1 (86.8 mg, 0.20 mmol) were added to the reaction tube, pumped through a vacuum line for three times, and under a nitrogen atmosphere, DCM (2.0 mL) was added, and the reaction was carried out at room temperature for 24 h. After the reaction was completed, the solvent was removed by rotary evaporation, the sample was loaded by dry method, and purified by column chromatography (petroleum ether:ethyl acetate (v / v)=19:1) to obtain the product P-1 (53.1 mg, yield 88%).
[0103] 1H NMR (400MHz, CDCl 3 )δ7.86–7.81(m,2H),7.34(d,J=8.1Hz,2H),7.32–7.22(m,2H),7.22–7.16(m,1H),7.15–7.09(m,2H) ,2.78(tt,J=7.5,4.7Hz,1H),2.65-2.56(m,3H),2.45(s,3H),2.06(d,J=4.6Hz,1H),1.88(dddd,J=13.9 ,8.8,7.4,4.9Hz,1H),1.67(dtd,J=14.1,8.0,6.4Hz,1H).
[0104] 13 C NMR (101MHz, CDCl 3 )δ144.64,140.80,135.10,129.78,128.57,128.42,128.12,126.23,39.84,34.02,33.25,33.09,21.77;
[0105] HRMS-ESI(m / z)[M+...
Embodiment 2
[0107]
[0108] First p-cyanobenzenesulfonamide (43.7mg, 0.24mmol), K 2 CO 3 (27.6 mg, 0.20 mmol) and compound R-1 (86.8 mg, 0.20 mmol) were added to the reaction tube, pumped through a vacuum line for three times, and under a nitrogen atmosphere, DCM (2.0 mL) was added, and the reaction was carried out at room temperature for 24 h. After the reaction was completed, the solvent was removed by rotary evaporation, dry method was applied, and the product was purified by column chromatography (petroleum ether:ethyl acetate (v / v)=19:1) to obtain the product P-2 (40.5 mg, yield 65%).
[0109] 1 H NMR (400MHz, CDCl 3 )δ8.09–8.03(m,2H),7.87–7.81(m,2H),7.28(td,J=7.7,7.1,1.1Hz,2H),7.23–7.17(m,1H),7.13(dd, J=7.0, 1.8Hz, 2H), 2.91 (tt, J=7.3, 4.9Hz, 1H), 2.72–2.59 (m, 3H), 2.12 (d, J=4.7Hz, 1H), 1.91 (dddd, J =13.9,8.7,7.5,5.1Hz,1H),1.73(dtd,J=14.1,7.9,6.4Hz,1H);
[0110] 13 C NMR (101MHz, CDCl 3 )δ142.59,140.38,132.96,128.68,128.68,128.41,126.42,117.38,117.29,40.52,34.84,33.08,...
Embodiment 3
[0113]
[0114] First p-methoxybenzenesulfonamide (44.9mg, 0.24mmol), K 2 CO 3 (27.6 mg, 0.20 mmol) and compound R-1 (86.8 mg, 0.20 mmol) were added to the reaction tube, pumped through a vacuum line for three times, and under a nitrogen atmosphere, DCM (2.0 mL) was added, and the reaction was carried out at room temperature for 24 h. After the reaction was completed, the solvent was removed by rotary evaporation, the sample was loaded by dry method, and purified by column chromatography (petroleum ether:ethyl acetate (v / v)=19:1) to obtain the product P-3 (43.8 mg, yield 69%).
[0115] 1 H NMR (600MHz, CDCl 3 )δ7.88(d,J=8.6Hz,2H),7.27(t,J=7.5Hz,2H),7.19(t,J=7.4Hz,1H),7.12(d,J=7.5Hz,2H) ,7.00(d,J=8.5Hz,2H),3.88(s,3H),2.75(td,J=7.4,3.7Hz,1H),2.66–2.55(m,3H),2.04(d,J=4.5 Hz,1H),1.88(dddd,J=13.8,8.8,7.3,4.9Hz,1H),1.73–1.63(m,1H);
[0116] 13 C NMR (151MHz, CDCl 3 )δ163.76,140.85,130.32,129.65,128.60,128.46,126.26,114.38,55.80,39.82,33.97,33.27,33.14;
[0117] HRMS-ESI(m...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More