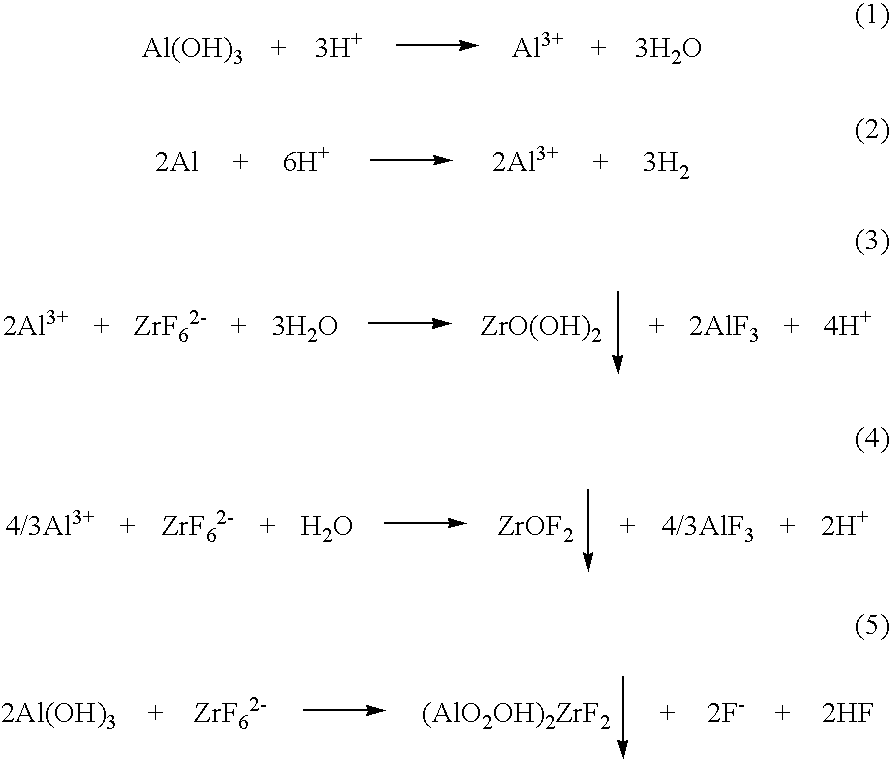In such an electroless reaction using a zirconium-based chemical conversion treatment agent, since it is extremely difficult to cause a homogeneous reaction to occur over the entire said surface, it is difficult to form an adequately dense and uniform coat and the resulting coat becomes one containing a high proportion of oxides and fluorides due to
etching of substrates, and therefore the corrosion resistance is deteriorated.
Therefore, only a rough and thin chemical conversion coat composed of a substrate metal or an
alkali metal can be attained and it is difficult to attain a uniform and dense protection coat.
consequently, it was difficult that a chemical conversion coat obtained by using the electroless treatment with a zirconium-based chemical conversion treatment agent provide a sufficient
rust prevention property particularly for an article to be treated such as an iron-based substrate and a
zinc-based substrate, having low reactivity with the chemical conversion treatment agent.
However, these methods concern a treatment method of
phosphate compounds and
titanium-based compounds, but are not methods for forming a uniform and dense zirconium chemical conversion coat.
Particularly, in a method of chemical conversion treatment in which
phosphate compounds are used, there is a problem of placing a burden on the environment due to issues of the
eutrophication.
Further, in chemical conversion treatment using a
titanium-based compound, a high degree of corrosion resistance cannot be attained.
However, in this method, when the chemical conversion treatment is conducted by the
electrolysis treatment using a composition for surface treatment, which is formed by dissolving compounds containing
fluorine and zirconium, it is difficult to attain an effect of protecting a
cathode in applying an
electrolysis voltage to a substrate to be treated and a chemical conversion coat, containing a relatively large amount of fluorides and
alkali metal compound, is formed since a large amount of and excessive fluorine and alkali metals are present in the solution.
Therefore, the corrosion resistance becomes unsatisfactory.
Further, a corrosion problem of facilities arises due to a large amount of fluorine.
When the concentration is less than 10 ppm, the corrosion resistance may not be achieved since the zirconium compound is not adequately deposited on the metal surface.
When it is more than 10000 ppm, it may be economically disadvantageous since further improvement is not recognized.
Further the corrosion resistance may be deteriorated since a chemical conversion coat having relatively much fluorine content.
When it is more than 1.0,
precipitation of metal salt may be occurred since the amount of the total fluorine becomes insufficient.
When the pH is less than 1, the zirconium compound becomes difficult to deposit, and therefore a sufficient amount of the coat cannot be obtained and the corrosion resistance may be deteriorated.
When the
voltage is less than 0.1 V, the amount of the coat is insufficient; therefore the corrosion resistance may be deteriorated.
When it is more than 40 V, effect from increase in the amount of the coat becomes saturated and energy
disadvantage may occur.
When the
current density is less than 0.1 A / dm.sup.2, the amount of the coat is insufficient; therefore the corrosion resistance may be deteriorated.
When it is more than 30 A / dm.sup.2, effect from increase in the amount of the coat becomes saturated and energy
disadvantage may occur.
When the
treatment time is less than 3 seconds, the amount of the coat is insufficient; therefore the corrosion resistance maybe deteriorated.
When it is more than 180 seconds, effect from increase in the amount of the coat becomes saturated and energy
disadvantage may occur.
When the treatment temperature is less than 10.degree. C., the amount of the coat is insufficient; therefore the corrosion resistance may be deteriorated.
When it is more than 70.degree. C., effect from increase in the amount of the coat becomes saturated and energy disadvantage may occur.
In addition, the
lower limit of the treatment temperature is not particularly controlled and the cathodic electrolysis treatment can be conducted at
room temperature.
When this amount is less than 10 mg / m.sup.2, the corrosion resistance may be insufficient.
When it is more than 300 mg / m.sup.2, it may be economically disadvantageous since further improvement in the corrosion resistance is not recognized.
 Login to View More
Login to View More