Three-tooth salicylaldehyde imine vanadium olefin polymerizing catalyst, preparation and uses thereof
A technology for tridentate salicylaldimine vanadium olefin and salicylaldimine vanadium olefin, which is applied to the tridentate salicylaldimine vanadium olefin polymerization catalyst and the fields of preparation and application, and can solve the problem of poor high temperature resistance, low catalytic activity, easy deactivation, etc. question
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
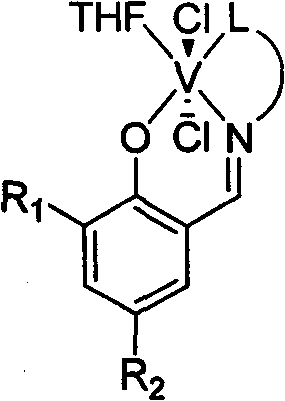
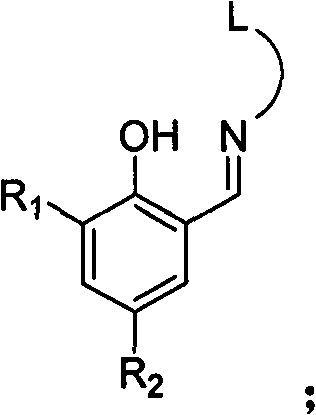
[0028] Add 9.77g of salicylaldehyde equivalent to 80mmol, 6.00g of 2-methoxyethylamine equivalent to 80mmol, 60ml of methanol, and 2ml of formic acid into the dry reactor, and react at 25°C for 12h. The solvent methanol was evaporated with a rotary evaporator, and 1000ml of a solution with a volume ratio of ethyl acetate and petroleum ether of 1:100 was used as an eluent, and the residue was subjected to column chromatography to obtain 16.5 g of yellow solid Schiff's base. 92%. 1 HNMR (300MHz, CDCl3 ): δ3.36(s, 3H, CH 3 ), 3.65(t, 2H, CH 2 N), 3.77(t, 2H, CH 2 O), 6.85-7.31 (m, 4H, Ar-H), 8.37 (s, 1H, CH=N), 13.41 (s, 1H, OH). According to mass spectrometry, the molecular ion peak m / e is 179. Elemental analysis measured value: C, 67.11%; H, 7.33%; N, 7.79%; theoretical value (C 10 h 13 NO 2 ): C, 67.02%; H, 7.31%; N, 7.82%.
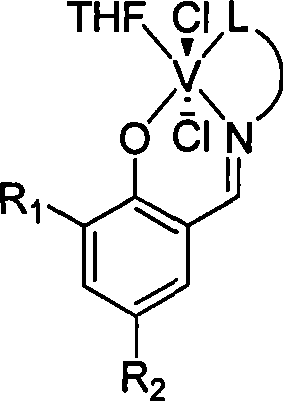
[0029] Under a nitrogen atmosphere, add 0.27 g of the above-obtained Schiffer’s base equivalent to 1.5 mmol and 20 ml of anhydrous tetrahydrofura...
Embodiment 2
[0036] Salicylaldehyde 4.89g is equivalent to 40mmol, and N, N-dimethylethylenediamine 3.53g is equivalent to 40mmol to replace 2-methoxyethylamine in Example 1, methanol 30ml, formic acid 1ml, and react at 25°C for 6h. The experimental operation was the same as in Example 1, and 7.15 g of yellow solid Schiff's base was obtained, with a yield of 93%. 1 H MR (300MHz, CDCl 3 ): δ2.30(s, 6H, CH 3 ), 2.63(t, 2H, CH 2 N), 3.73(t, 2H, CH 2 N), 6.84-7.32 (m, 4H, Ar-H), 8.37 (s, 1H, CH=N), 13.47 (s, 1H, OH). According to mass spectrometry, the molecular ion peak m / e is 192. Elemental analysis measured value: C, 68.70%; H, 8.40%; N, 14.58%; Theoretical value (C 11 h 16 N 2 O): C, 68.72%; H, 8.39%; N, 14.57%.
[0037] Under a nitrogen atmosphere, add 0.19 g of the above-obtained Schiffer’s base equivalent to 1.0 mmol and 10 ml of anhydrous tetrahydrofuran to a dry reactor, stir at room temperature for 10 min to dissolve the solid, add 26.4 mg of sodium hydride equivalent to 1.1 ...
Embodiment 3
[0044] 7.33g of salicylaldehyde is equivalent to 60mmol, and 6.49g of 2-aminomethylpyridine is equivalent to 60mmol instead of 2-methoxyethylamine, 30ml of methanol, 1ml of formic acid in Example 1, and reacted at 25°C for 48h. The experimental operation is the same as the implementation Example 1, 10.19 g of yellow liquid Schiffer's base was obtained, and the yield was 80%. 1 H NMR (300MHz, CDCl 3 ): δ4.95(s, 2H, CH 2), 6.77-7.72 (m, 7H, Ar-H and pyridine-H), 8.55 (s, 1H, CH=N), 8.48 (d, 1H, pyridine-H), 13.28 (s, 1H, OH). According to mass spectrometry, the molecular ion peak m / e is 212. Elemental analysis measured value: C, 73.69%; H, 5.72%; N, 13.24%; Theoretical value (C 13 h 12 N 2 O): C, 73.56%; H, 5.70%; N, 13.20%.
[0045] Under a nitrogen atmosphere, add 0.21 g of the above-obtained Schiffer’s base equivalent to 1.0 mmol and 10 ml of anhydrous tetrahydrofuran to a dry reactor, stir at room temperature for 10 minutes to dissolve the solid, add 36 mg of sodium hy...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight distribution | aaaaa | aaaaa |
| molecular weight distribution | aaaaa | aaaaa |
| molecular weight distribution | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com