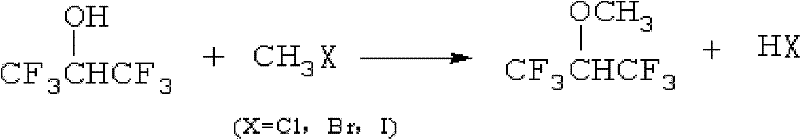Preparation method of hexafluoroisopropyl methyl ether
A technology of hexafluoroisopropyl methyl ether and hexafluoroisopropanol, applied in 1 field, can solve the problems of high operating cost, low product yield and the like
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0022] Add raw material hexafluoroisopropanol 70g, sodium hydroxide 15g, solvent acetonitrile 60g into a 0.25L pressure reactor, heat up to 40°C after mixing, slowly add 50g methyl iodide while stirring, keep constant temperature for 6 hours, and the reaction ends After cooling down, 52.7 g of the product hexafluoroisopropyl methyl ether was obtained by distillation, and the product yield was 82.3%.
Embodiment 2
[0024] Add 70g of raw material hexafluoroisopropanol, 20g of potassium hydroxide, and 50g of solvent dimethyl sulfoxide into a 0.25L pressure reactor, heat up to 50°C after mixing, slowly add 30g of methyl bromide under stirring, and react at constant temperature for 6 hours , Distilled after the reaction to obtain the product hexafluoroisopropyl methyl ether 43g, the product yield was 74.1%.
Embodiment 3
[0026] Add 80g of raw material hexafluoroisopropanol, 20g of sodium hydroxide, 60g of solvent N,N-dimethylformamide into a 0.25L pressure reactor, heat up to 60°C after mixing, and slowly add 40g of methyl chloride under stirring , constant temperature reaction for 6 hours, cooling down after the reaction, distillation to obtain the product hexafluoroisopropyl methyl ether 58.7g, product yield 67.7%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More