Patents
Literature
673 results about "Amino terminal" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Amino-terminal (not comparable) (biochemistry) Describing the amino acid, at one end of a polypeptide or protein, that has a free amino group.
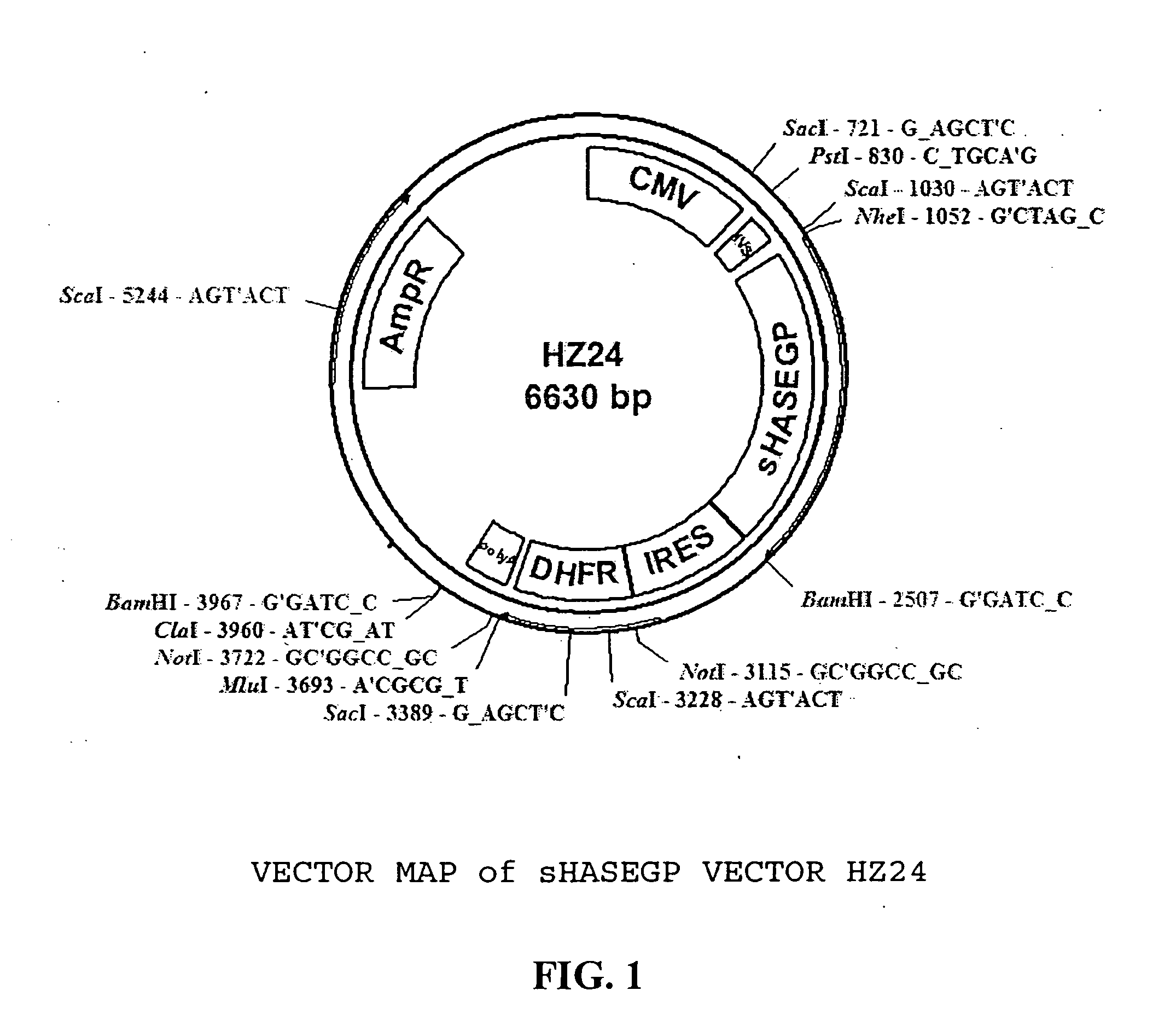
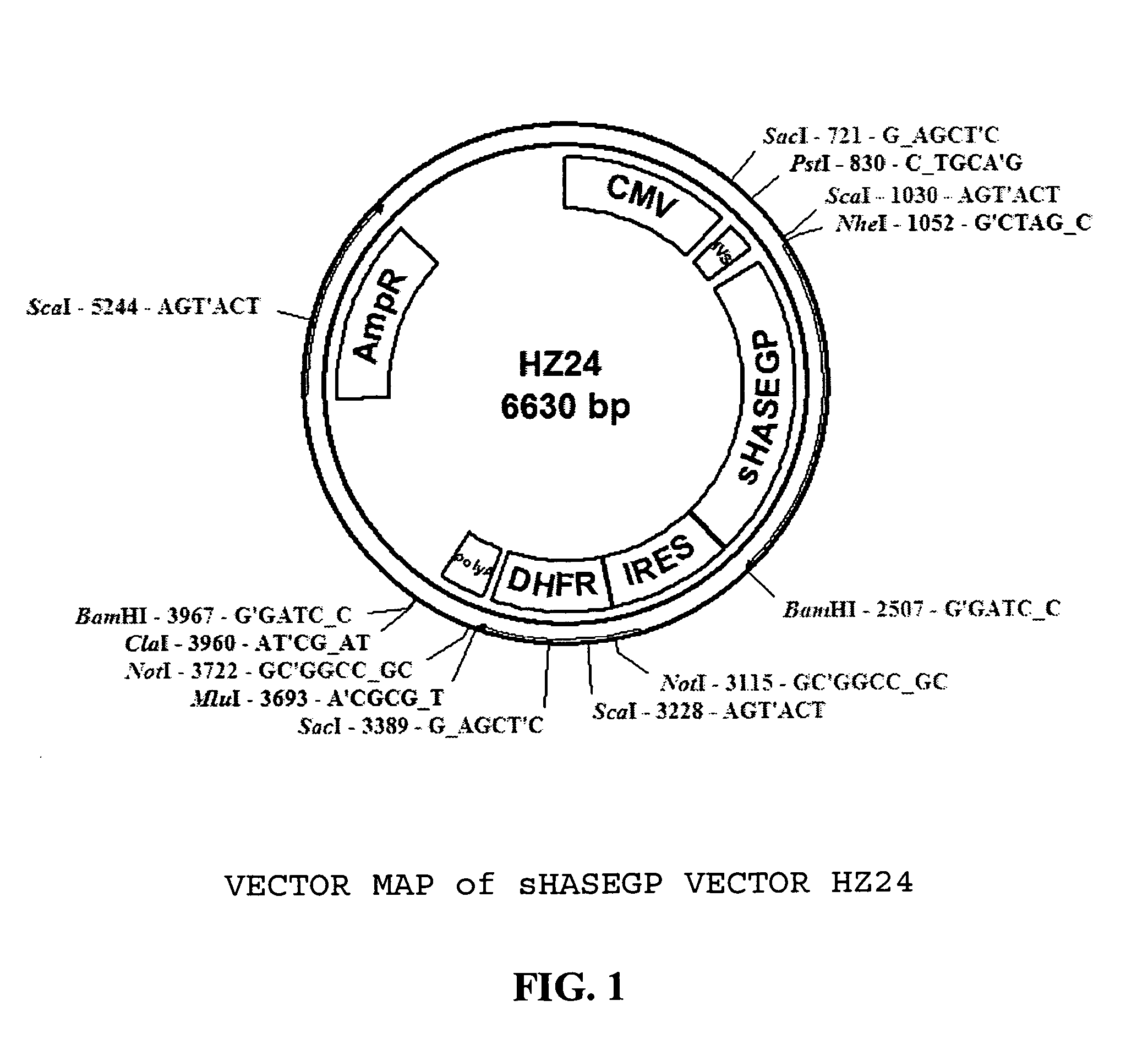
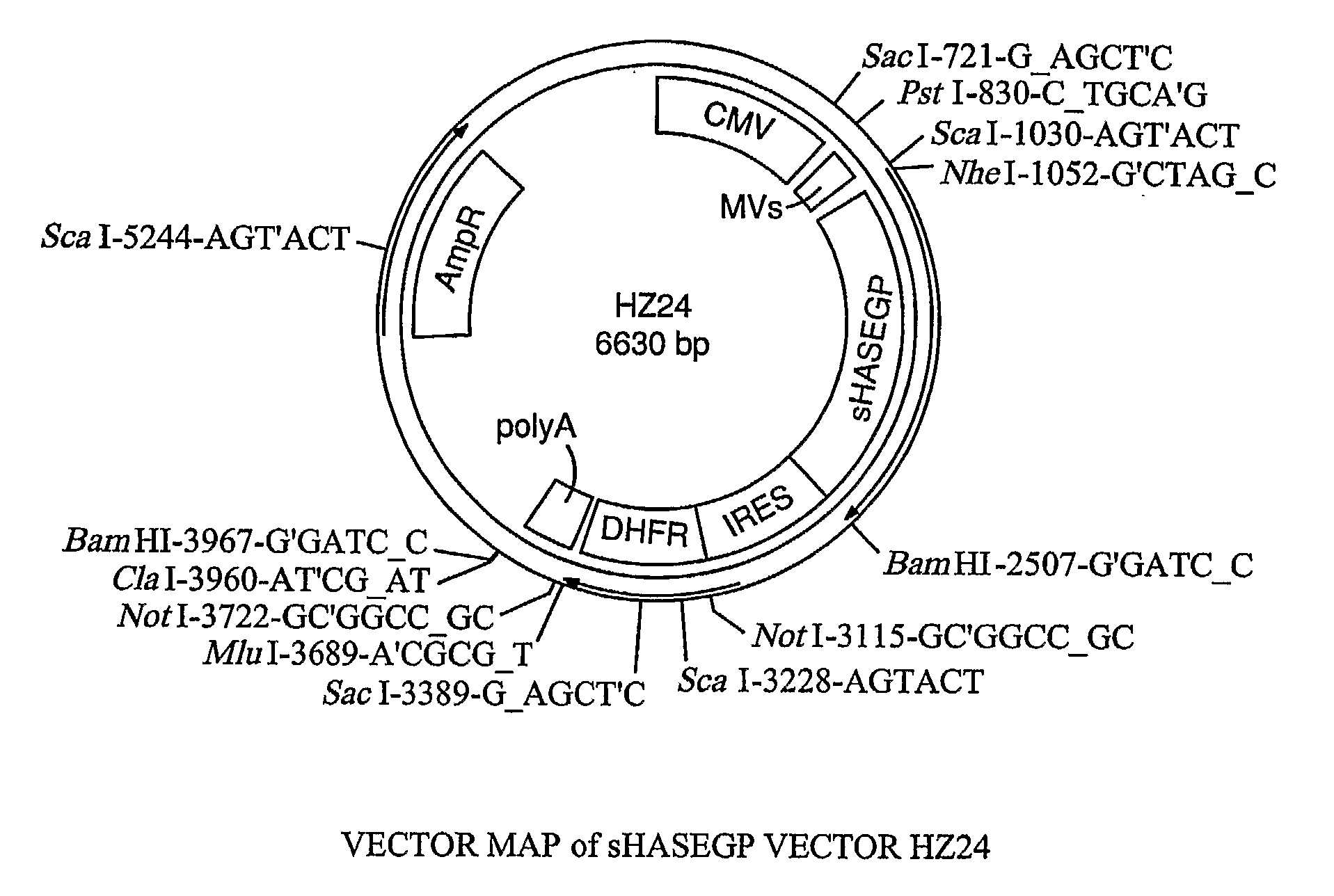
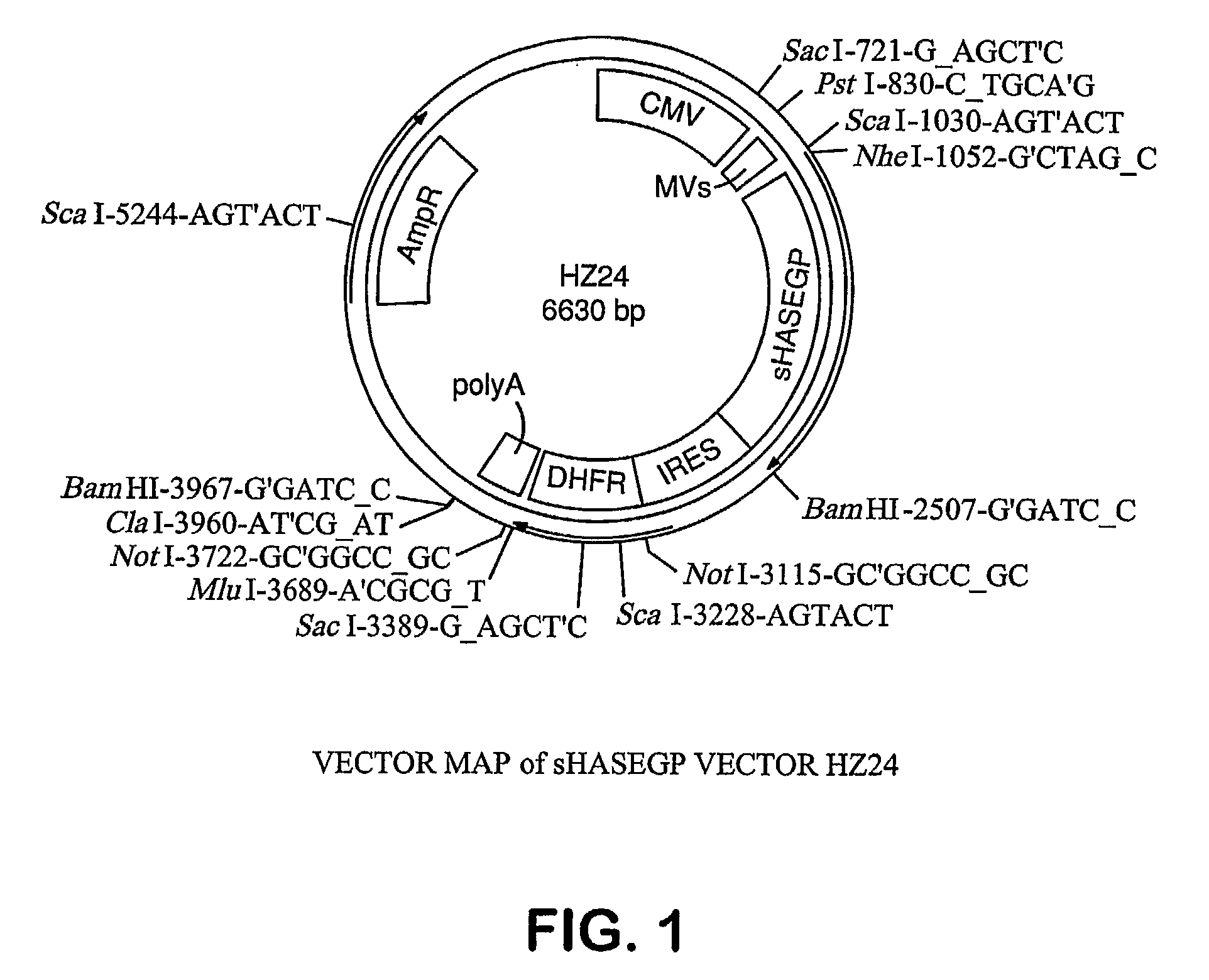
Soluble glycosaminoglycanases and methods of preparing and using soluble glycosaminogly ycanases
PendingUS20060104968A1Improve extentIncrease ratingsSenses disorderNervous disorderHyaluronidaseRecombinant glycoprotein
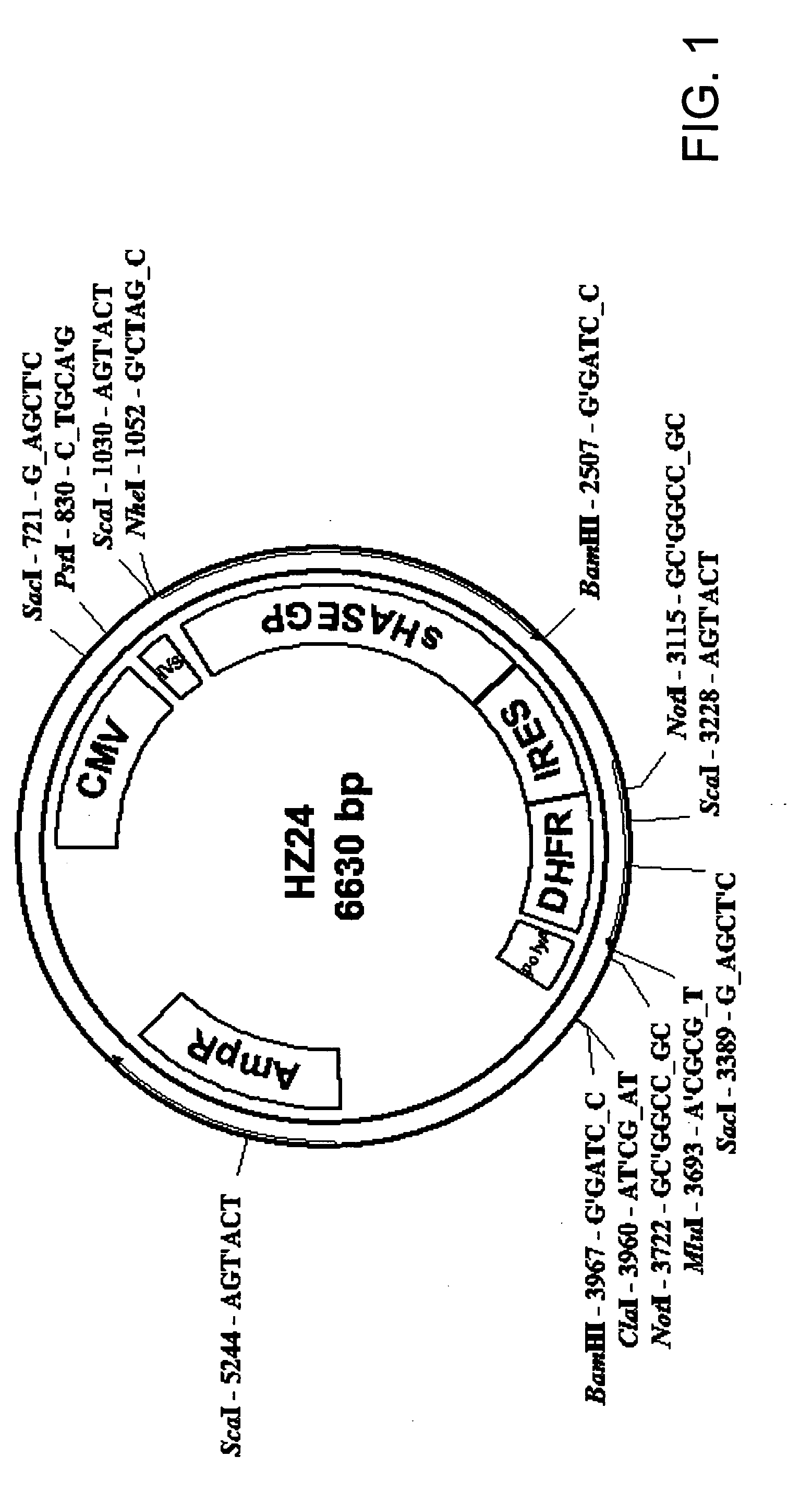
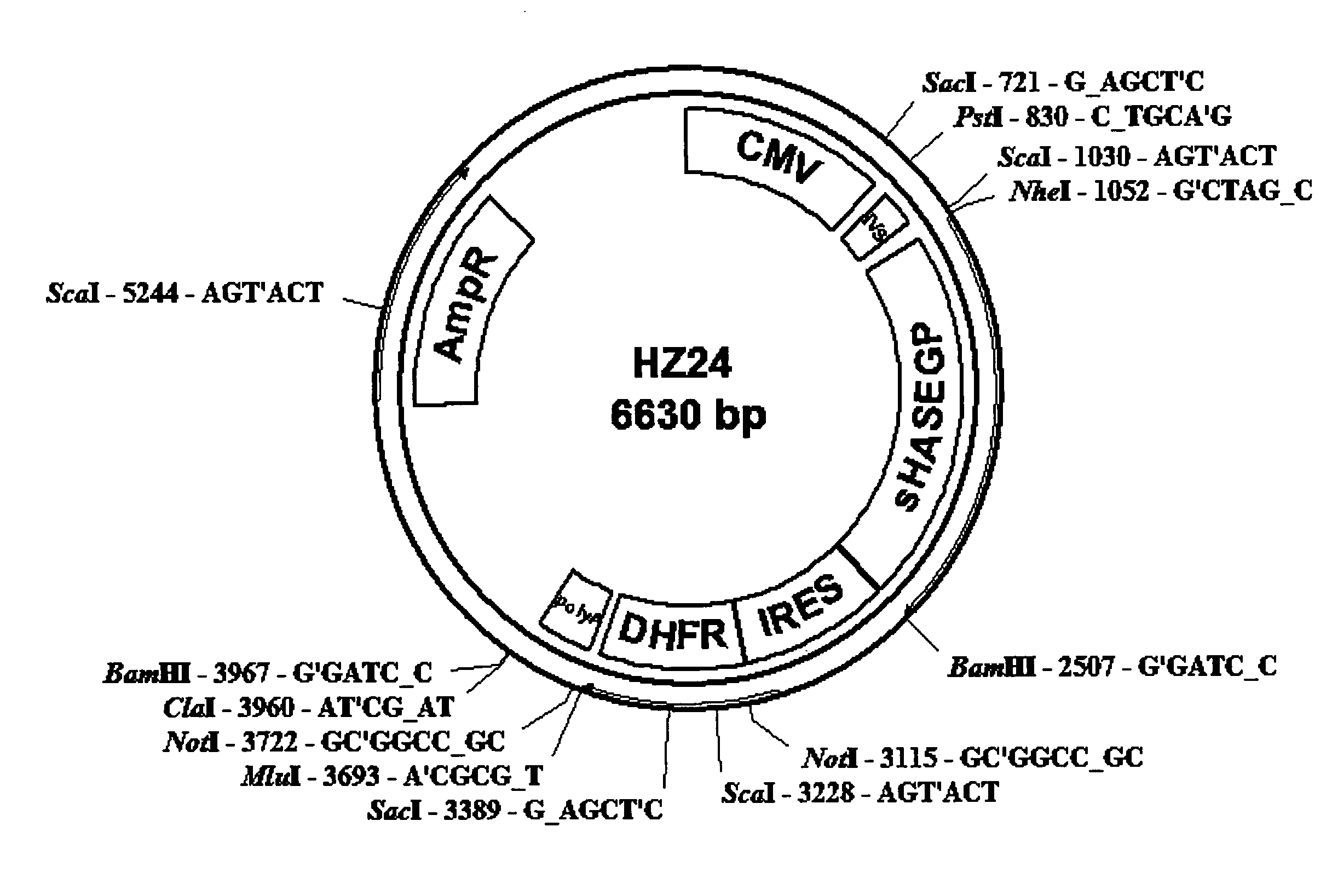
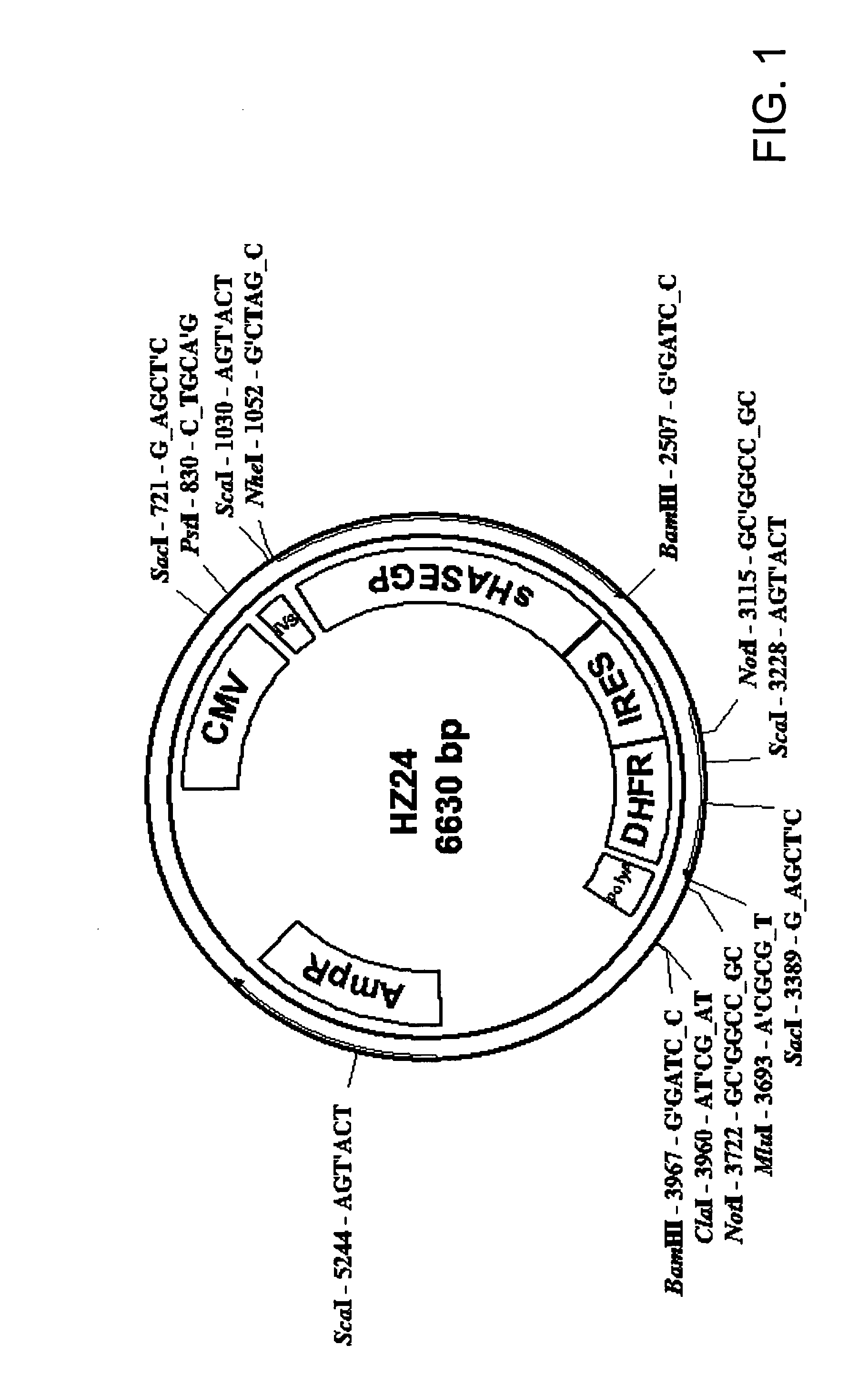
The invention relates to the discovery of novel soluble neutral active Hyaluronidase Glycoproteins (sHASEGPs), methods of manufacture, and their use to facilitate administration of other molecules or to alleviate glycosaminoglycan associated pathologies. Minimally active polypeptide domains of the soluble, neutral active sHASEGP domains are described that include asparagine-linked sugar moieties required for a functional neutral active hyaluronidase domain. Included are modified amino-terminal leader peptides that enhance secretion of sHASEGP. The invention further comprises sialated and pegylated forms of a recombinant sHASEGP to enhance stability and serum pharmacokinetics over naturally occurring slaughterhouse enzymes. Further described are suitable formulations of a substantially purified recombinant sHASEGP glycoprotein derived from a eukaryotic cell that generate the proper glycosylation required for its optimal activity.
Owner:HALOZYME
Soluble glycosaminoglycanases and methods of preparing and using soluble glycosaminoglycanases
ActiveUS20050260186A1Improve extentIncrease ratingsAntibacterial agentsSenses disorderHyaluronidasePathology diagnosis
The invention relates to the discovery of novel soluble neutral active Hyaluronidase Glycoproteins (sHASEGPs), methods of manufacture, and their use to facilitate administration of other molecules or to alleviate glycosaminoglycan associated pathologies. Minimally active polypeptide domains of the soluble, neutral active sHASEGP domains are described that include asparagine-linked sugar moieties required for a functional neutral active hyaluronidase domain. Included are modified amino-terminal leader peptides that enhance secretion of sHASEGP. The invention further comprises sialated and pegylated forms of a recombinant sHASEGP to enhance stability and serum pharmacokinetics over naturally occurring slaughterhouse enzymes. Further described are suitable formulations of a substantially purified recombinant sHASEGP glycoprotein derived from a eukaryotic cell that generate the proper glycosylation required for its optimal activity.
Owner:HALOZYME
Nucleic acid encoding poly-zinc finger proteins with improved linkers
InactiveUS7153949B2Enhanced affinity and specificityImprove the level ofPeptide/protein ingredientsAntibody mimetics/scaffoldsDNA-binding domainNucleotide
Polynucleotides encoding chimeric proteins, and methods for their production and use are disclosed. The chimeric proteins comprise a flexible linker between two zinc finger DNA-binding domains, wherein the linker contains eight or more amino acids between the second conserved histidine residue of the carboxy-terminal zinc finger of the first domain and the first conserved cysteine residue of the amino-terminal zinc finger of the second domain.
Owner:MASSACHUSETTS INST OF TECH
RNA interference mediated inhibition of MAP kinase gene expression using short interfering nucleic acid (siNA)
InactiveUS20050239731A1Improve bioavailabilityMinimize the possibilityBiocideSugar derivativesMAP Kinase GeneMitogen-activated protein
This invention relates to compounds, compositions, and methods useful for modulating mitogen activated protein kinase (MAP kinase) gene expression using short interfering nucleic acid (siNA) molecules. This invention also relates to compounds, compositions, and methods useful for modulating the expression and activity of other genes involved in pathways of MAP kinase gene expression and / or activity by RNA interference (RNAi) using small nucleic acid molecules. In particular, the instant invention features small nucleic acid molecules, such as short interfering nucleic acid (siNA), short interfering RNA (siRNA), double-stranded RNA (dsRNA), micro-RNA (miRNA), and short hairpin RNA (shRNA) molecules and methods used to modulate the expression of MAP kinase genes, such as Jun amino-terminal kinase (e.g., JNK-1, JNK-2), p38 (MAPK 14), ERK (e.g., ERK-1, ERK-2) and / or c-Jun.
Owner:SIRNA THERAPEUTICS INC
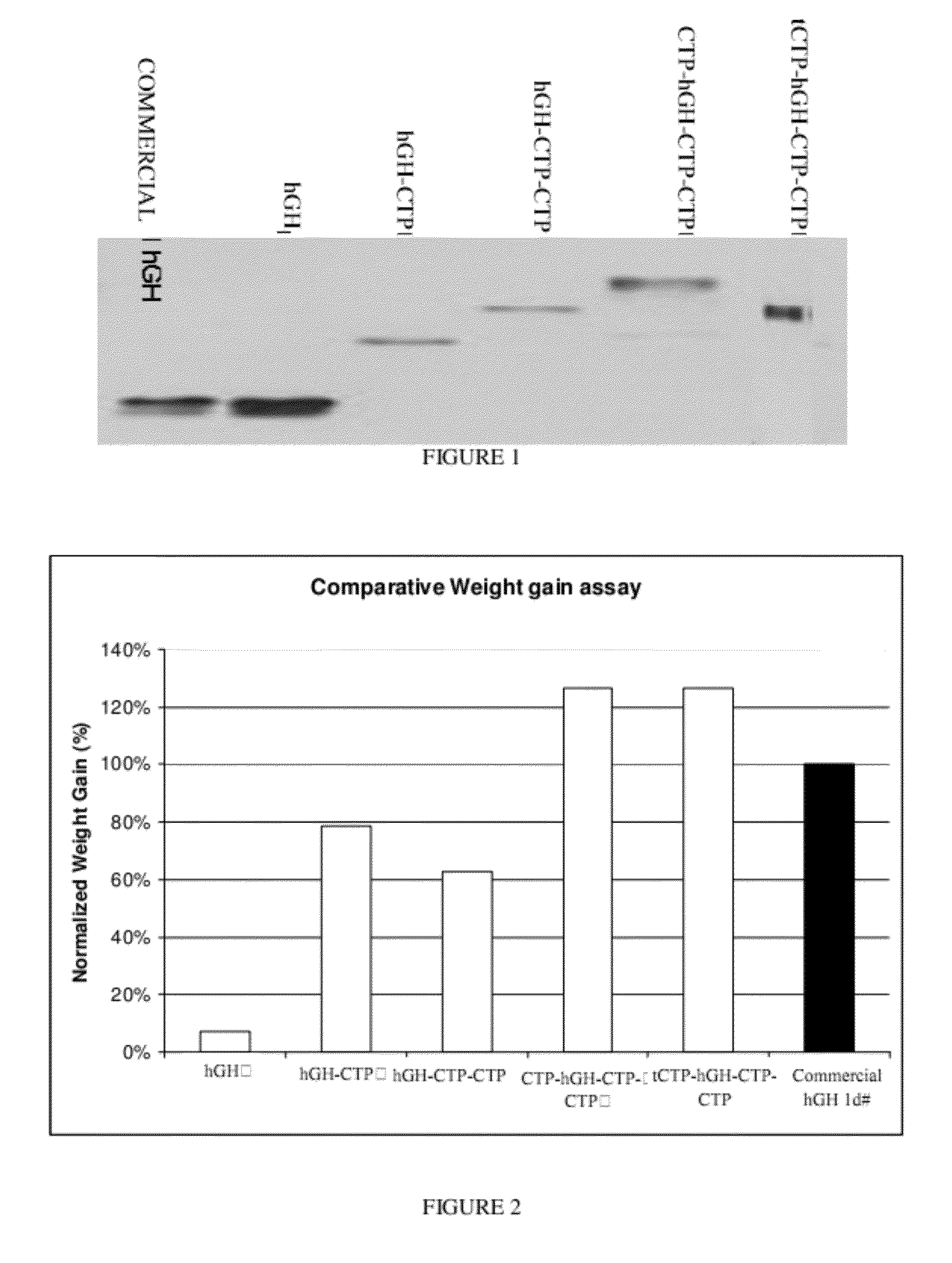
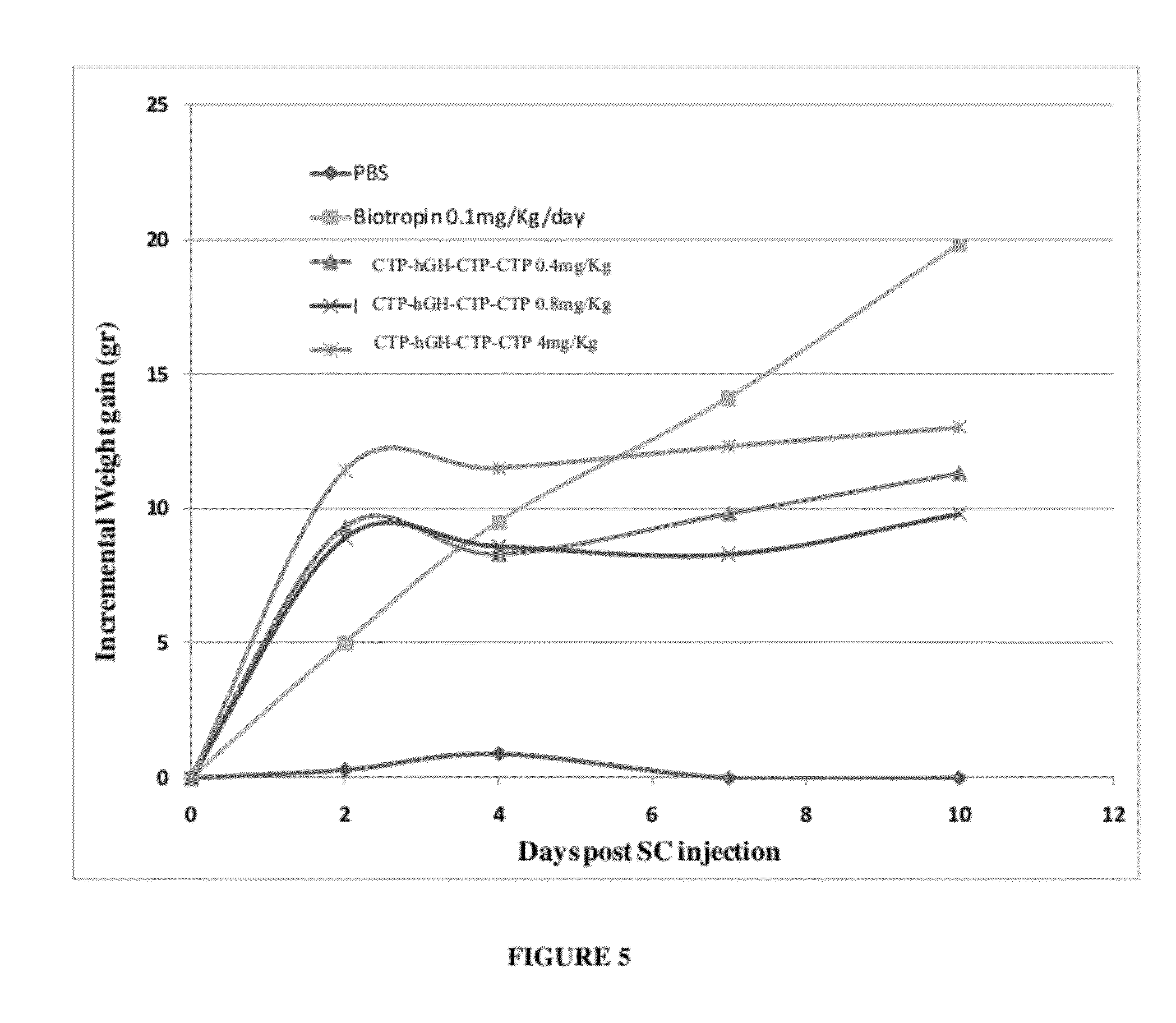
Long-acting growth hormone and methods of producing same
ActiveUS20120035101A1Decreasing body fatReduce weight lossPeptide/protein ingredientsMetabolism disorderSomatotropic hormoneNucleotide
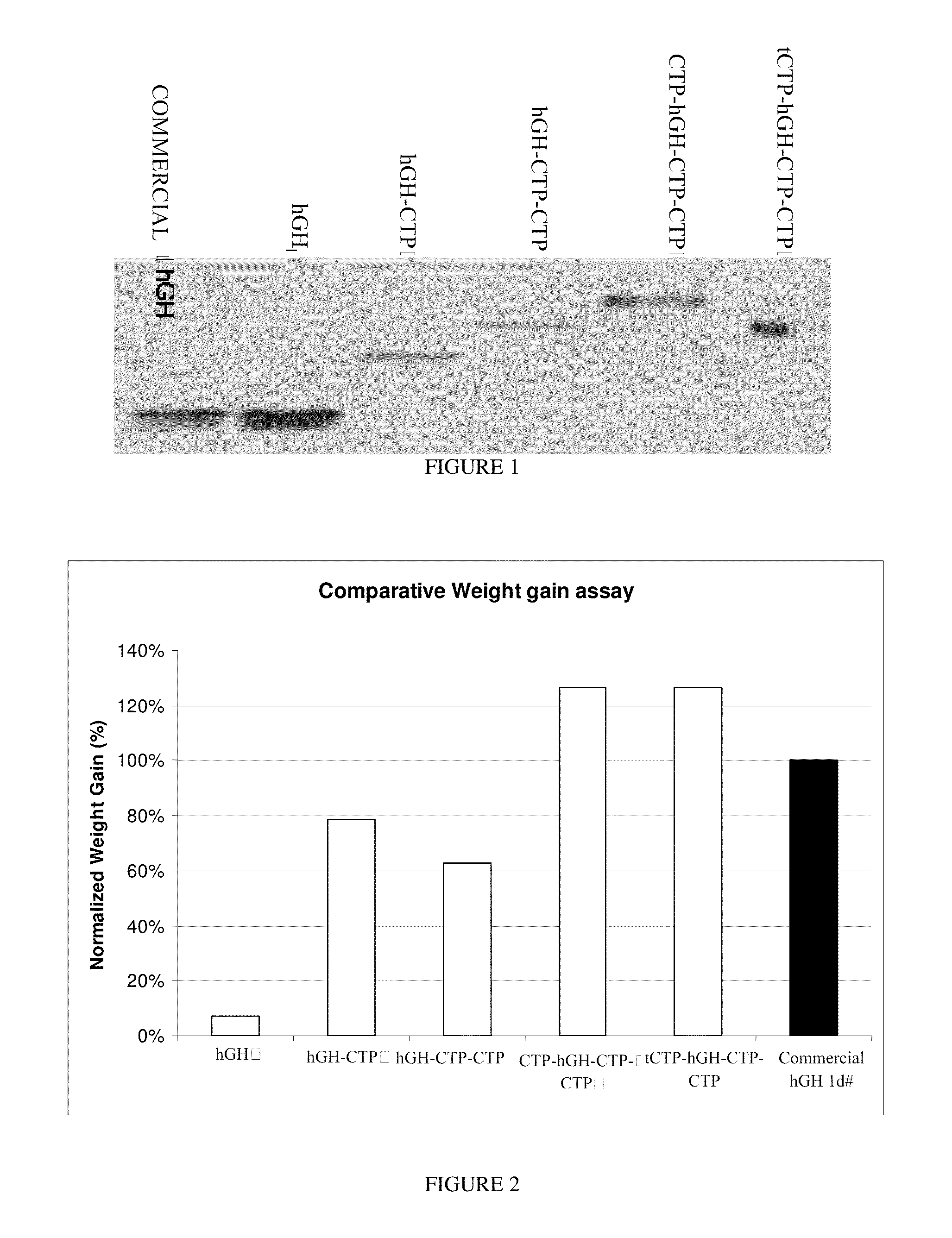
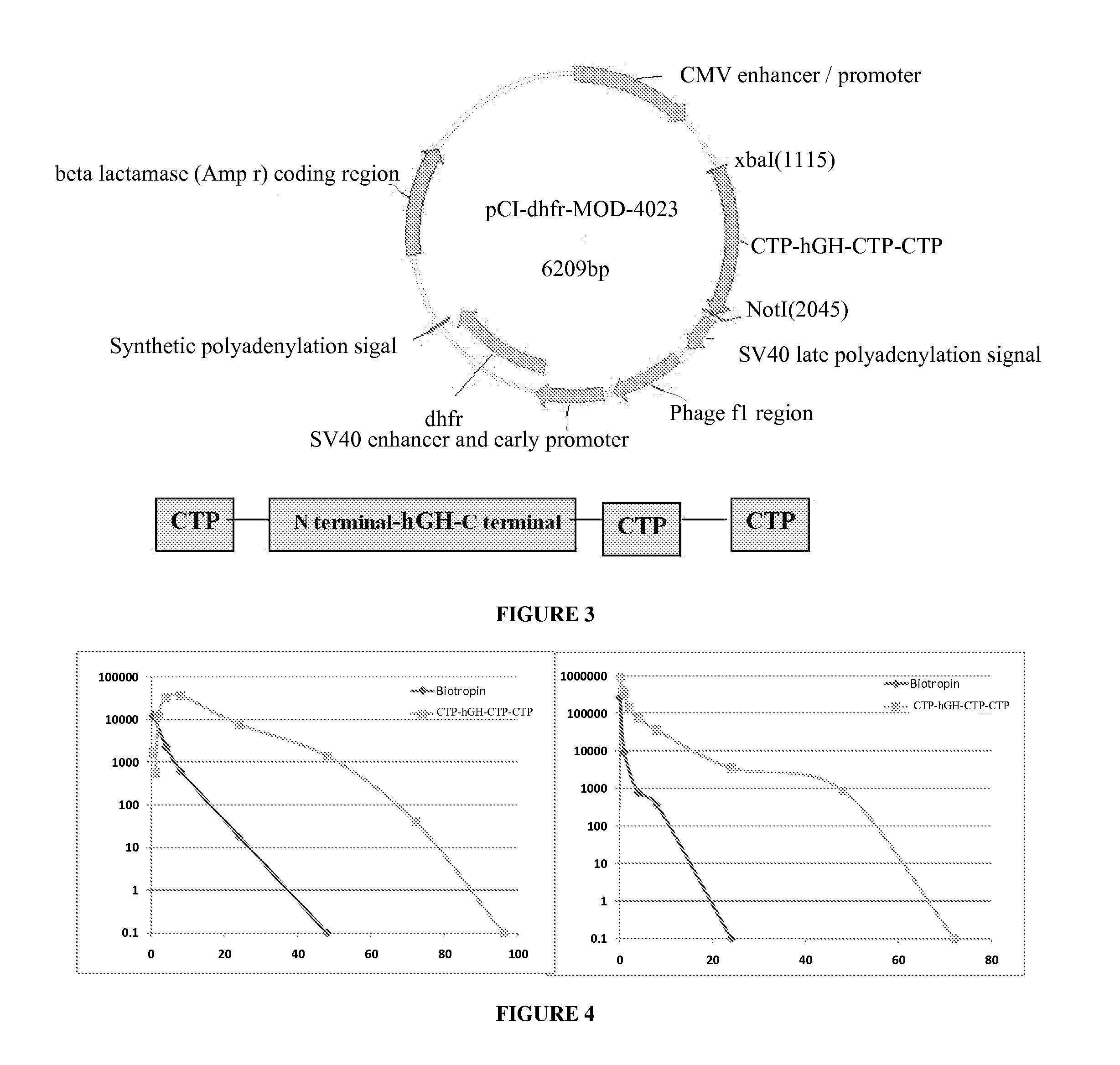
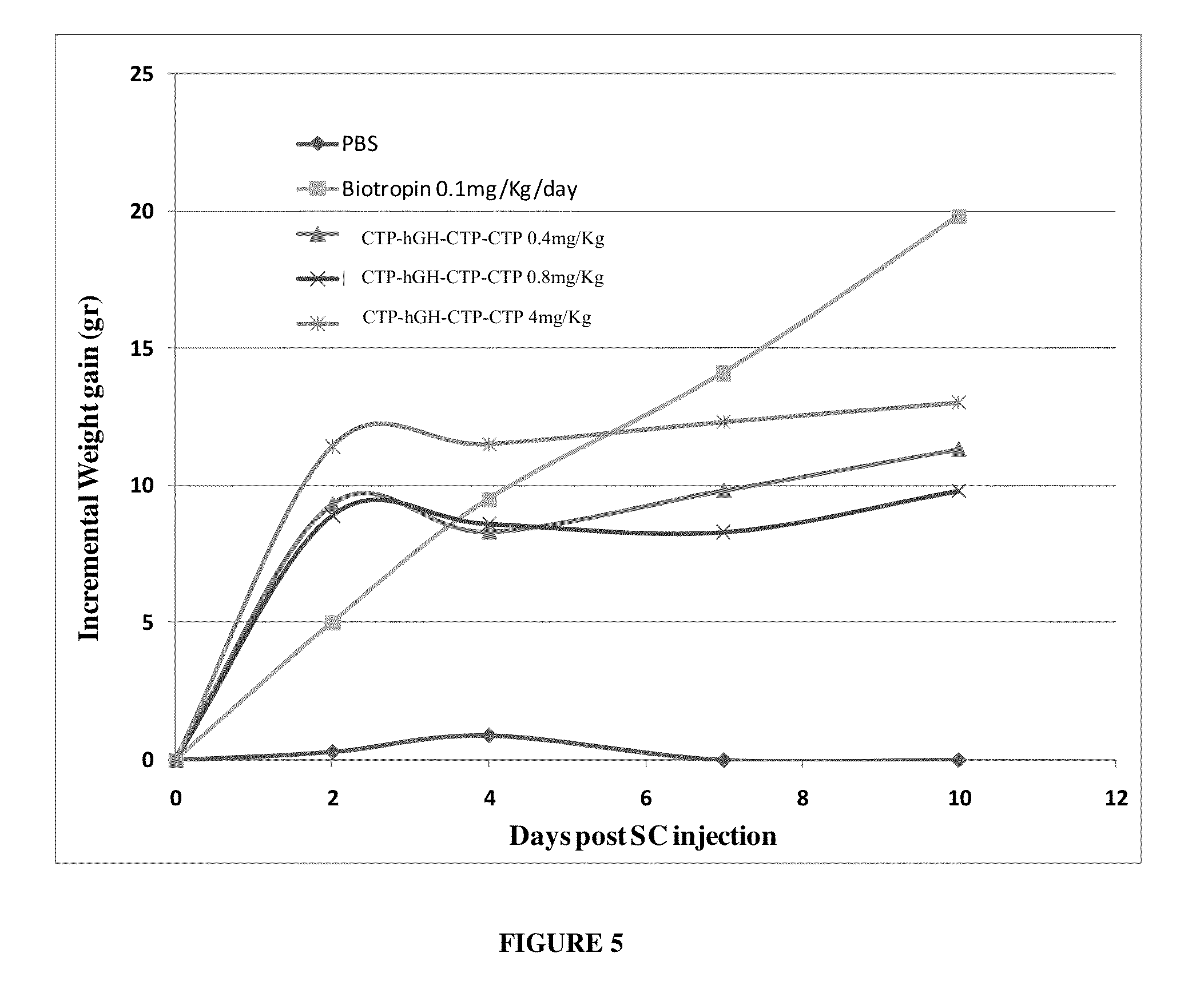
Use of a growth hormone protein and polynucleotides encoding same comprising an amino-terminal carboxy-terminal peptide (CTP) of chorionic gonadotrophin and two carboxy-terminal chorionic gonadotrophin CTPs attached to the growth hormone in methods of inducing growth or weight gain, method of increasing insulin-like growth factor (IGF-1) levels, and methods of reducing the dosing frequency of a growth hormone in a human subject are disclosed. Pharmaceutical compositions comprising the growth hormone and polynucleotides encoding the growth hormone of the invention and methods of using same are also disclosed.
Owner:OPKO BIOLOGICS
Fully active alternansucrases partially deleted in its carboxy-terminal and amino-terminal domains and mutants thereof
Nucleic acid sequences of truncated or mutated alternansucrases, vectors containing these nucleic acids sequences, host cells transformed with the nucleic acid sequences encoding truncated or mutated alternansucrases are provided. Furthermore, a process to recombinantly alternansucrase with a high level of expression, while retaining the enzymatic activity is described.
Owner:CENT NAT DE LA RECHERCHE SCI
Soluble Glycosaminoglycanases and Methods of Preparing and Using Soluble Glycosaminoglycanases
InactiveUS20090123367A1Facilitated DiffusionEnhance convective transportBacterial antigen ingredientsPeptide/protein ingredientsHyaluronidaseNuclear chemistry
The invention relates to the discovery of novel soluble neutral active Hyaluronidase Glycoproteins (sHASEGPs), methods of manufacture, and their use to facilitate administration of other molecules or to alleviate glycosaminoglycan associated pathologies. Minimally active polypeptide domains of the soluble, neutral active sHASEGP domains are described that include asparagine-linked sugar moieties required for a functional neutral active hyaluronidase domain. Included are modified amino-terminal leader peptides that enhance secretion of sHASEGP. The invention further comprises sialated and pegylated forms of a recombinant sHASEGP to enhance stability and serum pharmacokinetics over naturally occurring slaughterhouse enzymes. Further described are suitable formulations of a substantially purified recombinant sHASEGP glycoprotein derived from a eukaryotic cell that generate the proper glycosylation required for its optimal activity.
Owner:HALOZYME +6
Rationally designed antibodies
Antibodies or fragments thereof having CDR regions replaced or fused with biologically active peptides are described. Flanking sequences may optionally be attached at one or both the carboxy-terminal and amino-terminal ends of the peptide in covalent association with adjacent framework regions. Compositions containing such antibodies or fragments thereof are useful in therapeutic and diagnostic modalities.
Owner:ALEXION PHARMA INC
Soluble hyaluronidase glycoprotein (sHASEGP), process for preparing the same, uses and pharmaceutical compositions comprising thereof
ActiveUS20090214505A1Extended half-lifeOptimize allocationAntibacterial agentsOrganic active ingredientsHyaluronidasePathology diagnosis
Provided are soluble neutral active Hyaluronidase Glycoproteins (sHASEGP's), methods of manufacture, and their use to facilitate administration of other molecules or to alleviate glycosaminoglycan associated pathologies. Minimally active polypeptide domains of the soluble, neutral active sHASEGP domains are described that include asparagine-linked sugar moieties required for a functional neutral active hyaluronidase domain. Included are modified amino-terminal leader peptides that enhance secretion of sHASEGP. Sialated and pegylated forms of the sHASEGPs also are provided. Methods of treatment by administering sHASEGPs and modified forms thereof also are provided.
Owner:HALOZYME
Dipeptidyl-peptidase protected protein
InactiveUS20070060512A1Improve biostabilityLower blood sugar levelsPeptide/protein ingredientsMetabolism disorderDiabetes mellitusDipeptidyl peptidase
The present invention provides modified therapeutic polypeptides or peptides partially or completely protected from DPP activity. The modified polypeptides or peptides comprise at least one additional amino acid at the amino terminus. The modified therapeutic polypeptides or peptides are useful in the treatment of diseases such as diabetes.
Owner:BIOREXIS PHARMA CORP
Diagnostic or therapeutic somatostatin or bombesin analog conjugates and uses thereof
InactiveUS20050070470A1Quick eliminationReduce drug toxicityOrganic active ingredientsSenses disorderDiseaseCytotoxicity
Disclosed are peptide agents and uses thereof that are analogs of biologically active peptides such as somatostatin and bombesin. The compounds of the invention have the general formula X-Y-Z-Q, where X is a cytotoxic agent, therapeutic agent, detectable label or chelating group, and Q is a biologically active peptide. In peptide agents of the invention Y is optionally a hydrophilic polymer or peptide, and Z is a linking peptide bonded to Q at the amino terminus of Q, having two, three, four, or five, amino acid residues selected to link X to Q, while retaining the biological activity of Q. Methods of using these peptide agents in the diagnosis and treatment of diseases are also disclosed.
Owner:TULANE EDUCATIONAL FUND
Soluble hyaluronidase glycoprotein (sHASEGP), process for preparing the same, uses and pharmaceutical compositions comprising thereof
ActiveUS20090181032A1Extended half-lifeImprove distributionAntibacterial agentsOrganic active ingredientsHyaluronidasePathology diagnosis
The invention relates to the discovery of novel soluble neutral active Hyaluronidase Glycoproteins (sHASEGP's), methods of manufacture, and their use to facilitate administration of other molecules or to alleviate glycosaminoglycan associated pathologies. Minimally active polypeptide domains of the soluble, neutral active sHASEGP domains are described that include asparagine-linked sugar moieties required for a functional neutral active hyaluronidase domain. Included are modified amino-terminal leader peptides that enhance secretion of sHASEGP. The invention further comprises sialated and pegylated forms of a recombinant sHASEGP to enhance stability and serum pharmacokinetics over naturally occurring slaughterhouse enzymes. Further described are suitable formulations of a substantially purified recombinant sHASEGP glycoprotein derived from a eukaryotic cell that generate the proper glycosylation required for its optimal activity.
Owner:HALOZYME
Soluble hyaluronidase glycoprotein (sHASEGP), process for preparing the same, uses and pharmaceutical compositions comprising thereof
ActiveUS20090181013A1Reduce deliveryImprove usabilityAntibacterial agentsOrganic active ingredientsHyaluronidasePathology diagnosis
The invention relates to the discovery of novel soluble neutral active Hyaluronidase Glycoproteins (sHASEGP's), methods of manufacture, and their use to facilitate administration of other molecules or to alleviate glycosaminoglycan associated pathologies. Minimally active polypeptide domains of the soluble, neutral active sHASEGP domains are described that include asparagine-linked sugar moieties required for a functional neutral active hyaluronidase domain. Included are modified amino-terminal leader peptides that enhance secretion of sHASEGP. The invention further comprises sialated and pegylated forms of a recombinant sHASEGP to enhance stability and serum pharmacokinetics over naturally occurring slaughterhouse enzymes. Further described are suitable formulations of a substantially purified recombinant sHASEGP glycoprotein derived from a eukaryotic cell that generate the proper glycosylation required for its optimal activity.
Owner:HALOZYME
Fusion proteins, uses thereof and processes for producing same
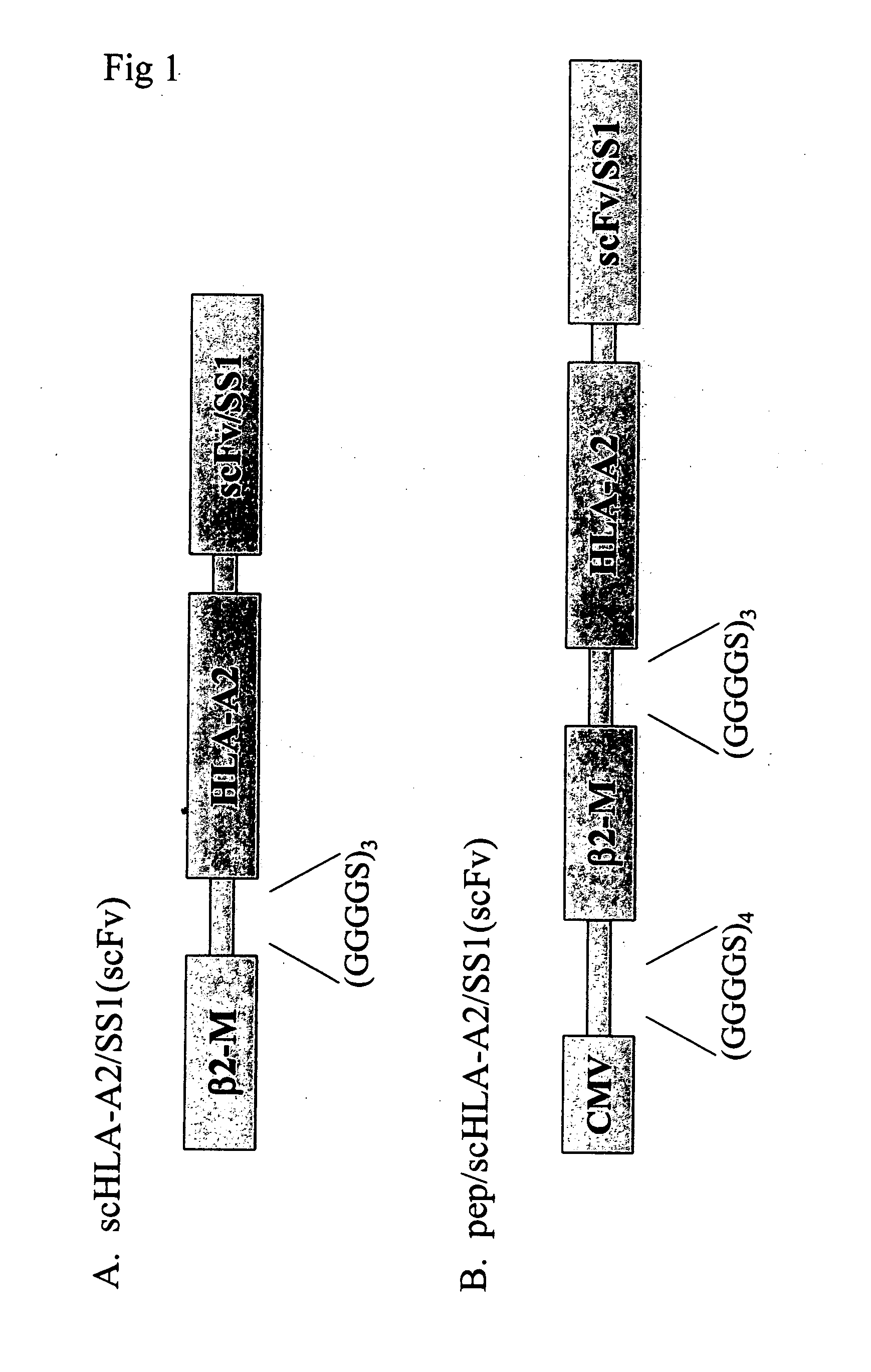
InactiveUS20080014208A1Efficient expressionAntibody mimetics/scaffoldsImmunoglobulins against cell receptors/antigens/surface-determinantsMHC class IAmino acid
This invention provides fusion proteins comprising consecutive amino acids which beginning at the amino terminus of the protein correspond to consecutive amino acids present in (i) a cytomegalovirus human MHC-restricted peptide, (ii) a first peptide linker, (iii) a human β-2 microglobulin, (iv) a second peptide linker, (v) a HLA-A2 chain of a human MHC class I molecule, (vi) a third peptide linker, (vii) a variable region from a heavy chain of a scFv fragment of an antibody, and (viii) a variable region from a light chain of such scFv fragment, wherein the consecutive amino acids which correspond to (vii) and (viii) are bound together directly by a peptide bond or by consecutive amino acids which correspond to a fourth peptide linker, wherein the antibody from which the scFv fragment is derived specifically binds to mesothelin. This invention provides nucleic acid constructs encoding same, processes for producing same, compositions, and uses thereof.
Owner:TECHNION RES & DEV FOUND LTD
Modified beta-lactamase and method for its preparation
The invention relates to targeted post translational modifi-cation of metallo-beta-lactamase by truncation and inser-tion of a dipeptide at the amino terminal end to reduce amino terminal heterogeneity in a recombinant DNA pro-duction system. A protein K-T-E-ΔBL is expressed, and modified by host proteases to E-ΔBL. Appropriate nucleotide molecules, vectors and hosts are also de-scribed. E-ΔBL is useful in a pharmaceutical composition for treating antibiotic induced adverse effects in the intes-tine of patients treated with beta-lactam antibiotics.
Owner:SYNTHETIC BIOLOGICS INC
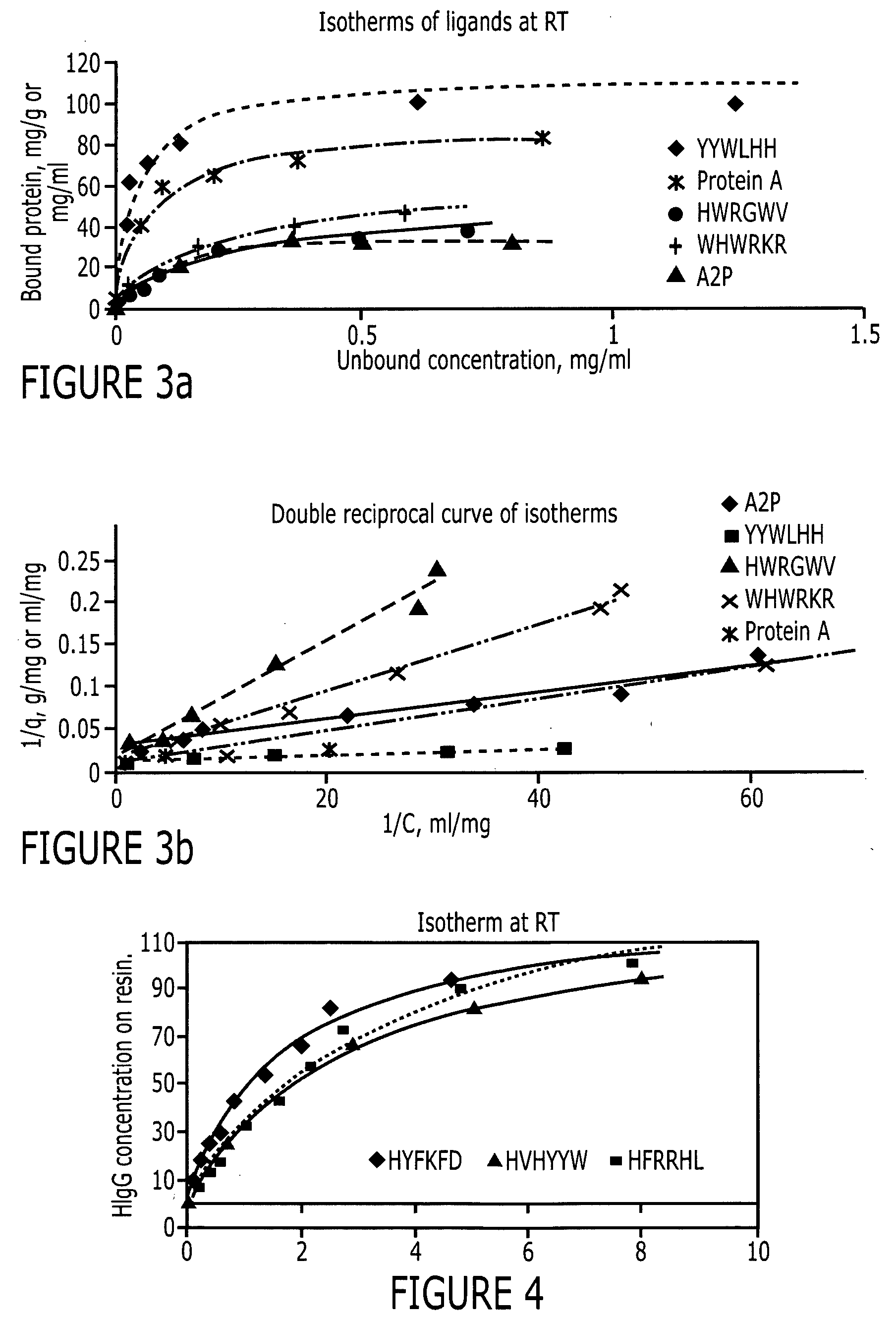
Purification of immunoglobulins using affinity chromatography and peptide ligands
ActiveUS20060153834A1High affinityPeptide librariesPeptide/protein ingredientsCarboxyl radicalPeptide ligand
An immunoglobulin binding peptide having the general formula, from amino terminus to carboxy terminus, of Z-R1—R2—R3—R4—R5—R6—X, is described, wherein: R1 is H or Y; R2 is a hydrophobic, preferentially aromatic, amino acid (for example W, F, Y, V); R3 is a positively charged or aromatic amino acid (for example R, H, F, W); R4 is a hydrophobic or positively charged amino acid (for example G, Y, R, K, L); R5 is a positively charged or aromatic amino acid (for example W, F, R, H, Y); R6 a random amino acid but preferably hydrophobic or negatively charged (for example V, W, L, D, H); X is present or absent and when present is a linking group; and Z is present or absent and when present is a capping group bonded to the N terminus of R1; and wherein the amino acids of said peptide are in D form, L form, or a combination thereof. Methods of using such peptides for the purification of Immunoglobulins are also described
Owner:NORTH CAROLINA STATE UNIV
Artificial combined antibacterial engineering polypeptide and its preparation method
Owner:四川新泰克控股有限责任公司
Modified beta-lactamase and method for its preparation
The invention relates to targeted post translational modification of metallo-beta-lactamase by truncation and insertion of a dipeptide at the amino terminal end to reduce amino terminal heterogeneity in a recombinant DNA production system. A protein K-T-E-ΔBL is expressed, and modified by host proteases to E-ΔBL. Appropriate nucleotide molecules, vectors and hosts are also described. E-ΔBL is useful in a pharmaceutical composition for treating antibiotic induced adverse effects in the intestine of patients treated with beta-lactam antibiotics.
Owner:SYNTHETIC BIOLOGICS INC
Rationally designed antibodies
Antibodies or fragments thereof having CDR regions replaced or fused with biologically active peptides are described. Flanking sequences may optionally be attached at one or both the carboxy-terminal and amino-terminal ends of the peptide in covalent association with adjacent framework regions. Compositions containing such antibodies or fragments thereof are useful in therapeutic and diagnostic modalities.
Owner:ALEXION PHARMA INC
Nanostructures, methods of synthesizing thereof, and methods of use thereof
ActiveUS20090196831A1Powder deliveryImmunoglobulins against animals/humansCrystallographySynthesis methods
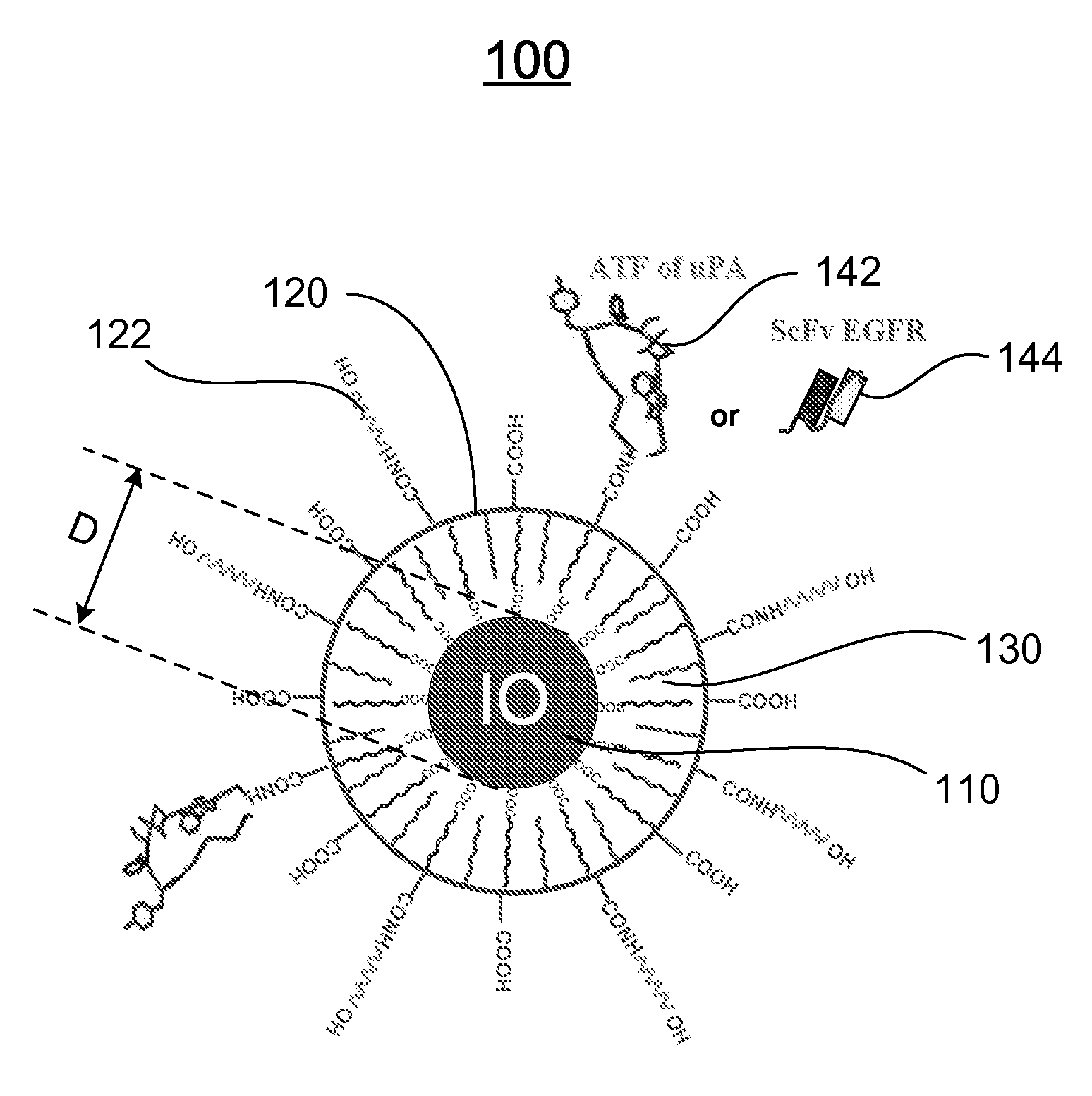
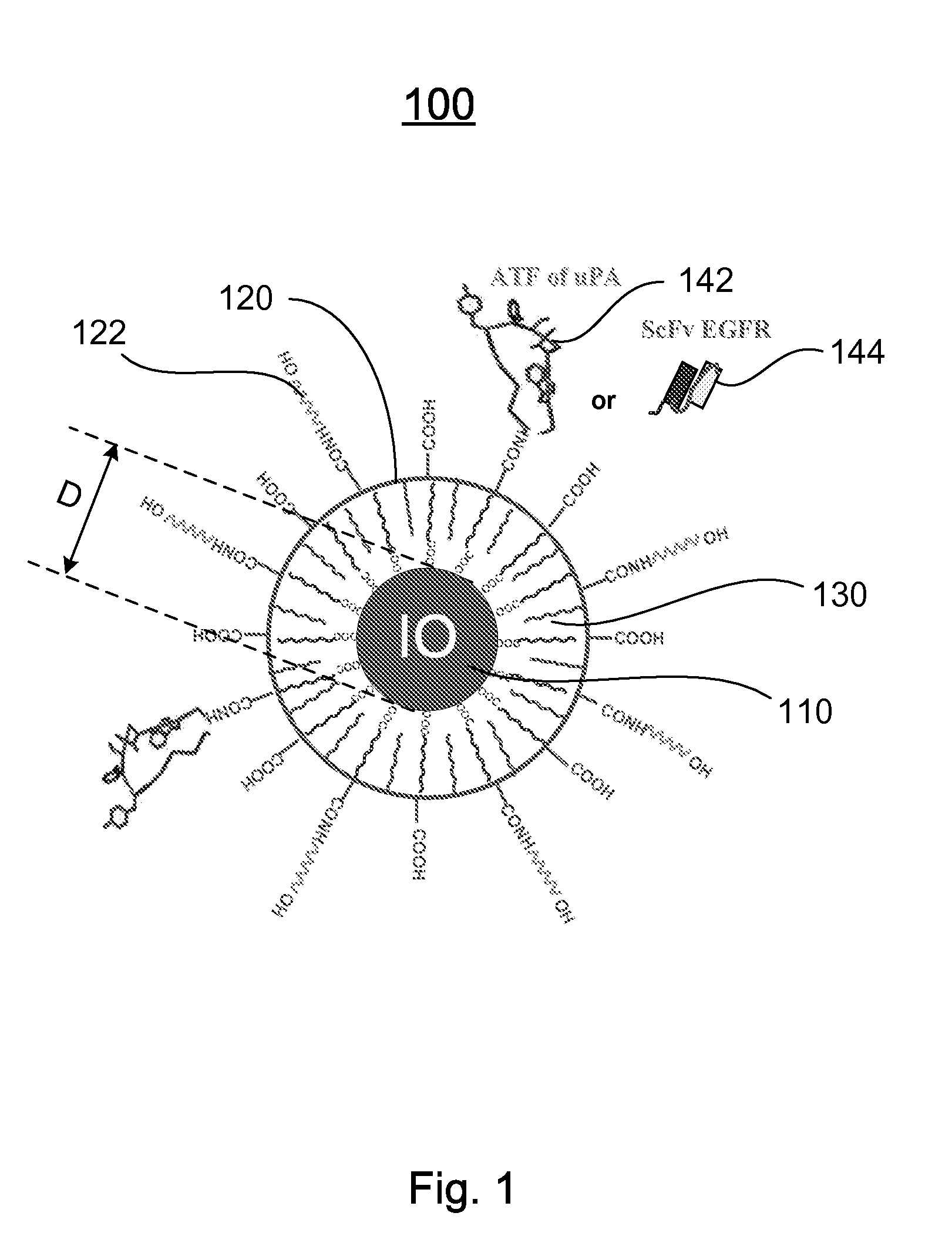
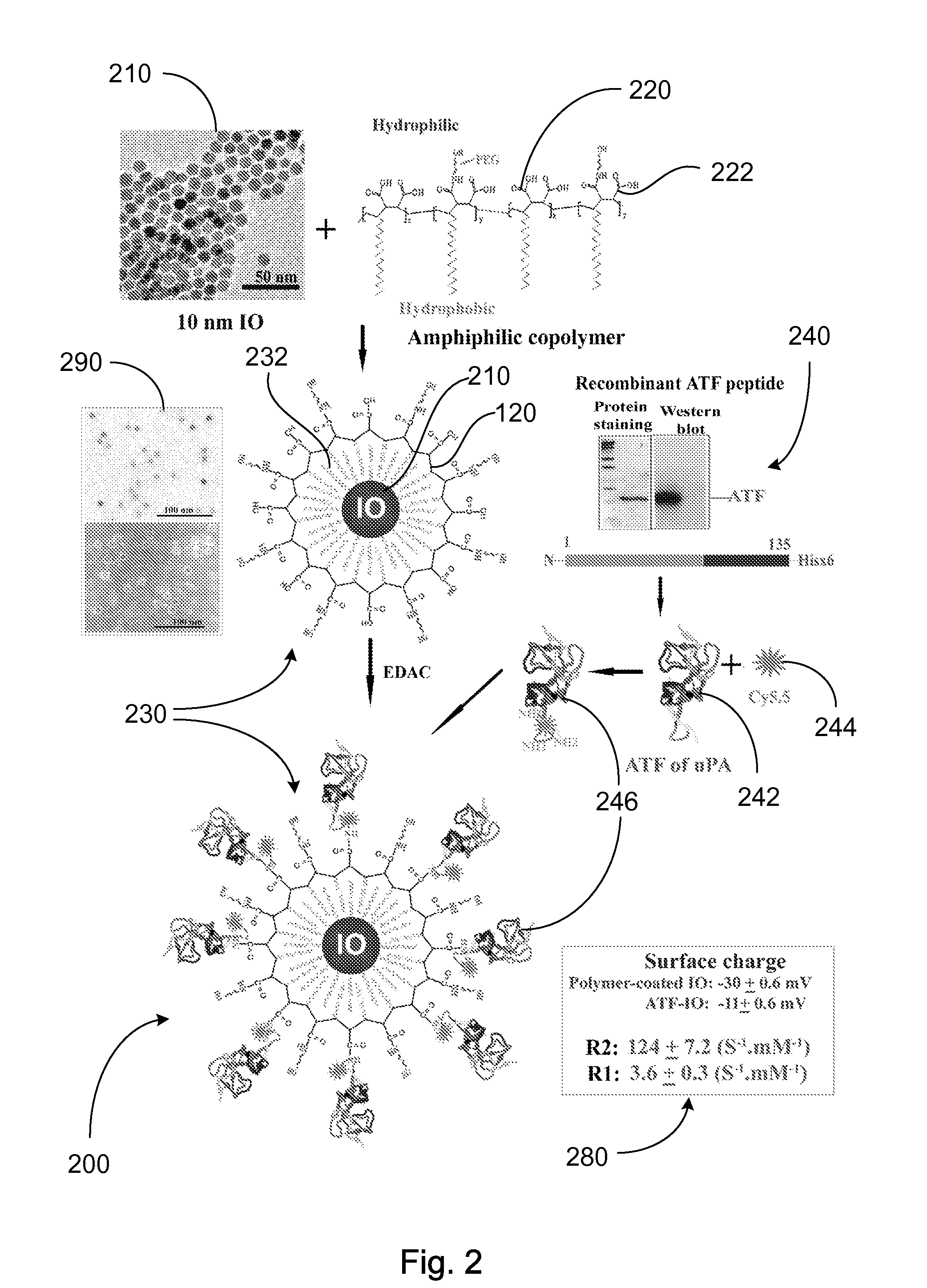
A nanostructure and methods of synthesizing same. In one embodiment, the nanostructure includes a magnetic iron oxide nanoparticle, a hydrophobic protection structure including at least an amphiphilic copolymer, wherein the hydrophobic protection structure encapsulates the magnetic iron oxide nanoparticle, and at least one amino-terminal fragment (ATF) peptide or epidermal growth factor receptor (EGFR) antibody conjugated to the amphiphilic copolymer.
Owner:EMORY UNIVERSITY
Soluble glycosaminoglycanases and methods of preparing and using soluble glycosaminoglycanases
ActiveUS20100196423A1Reduce sensitivityGreater serum half-livesAntibacterial agentsSenses disorderHyaluronidaseNuclear chemistry
The invention relates to the discovery of novel soluble neutral active Hyaluronidase Glycoproteins (sHASEGPs), methods of manufacture, and their use to facilitate administration of other molecules or to alleviate glycosaminoglycan associated pathologies. Minimally active polypeptide domains of the soluble, neutral active sHASEGP domains are described that include asparagine-linked sugar moieties required for a functional neutral active hyaluronidase domain. Included are modified amino-terminal leader peptides that enhance secretion of sHASEGP. The invention further comprises sialated and pegylated form of a recombinant sHASEGP to enhance stability and serum pharmacokinetics over naturally occurring slaughterhouse enzymes. Further described are suitable formulations of a substantially purified recombinant sHASEGP glycoprotein derived from a eukaryotic cell that generate the proper glycosylation required for its optimal activity.
Owner:HALOZYME
Prion-specific peptoid reagents
The invention relates to peptoid reagents that interact preferentially with a pathogenic form of a conformational disease protein as compared to a nonpathogenic form of the conformational disease protein where the peptoid reagent comprises an amino-terminal region, a carboxy-terminal region, and at least one peptoid region between the amino-terminal region and the carboxy-terminal region where the peptoid region comprises 3 to about 30 N-substituted glycines, and optionally one or more amino acids. The invention also relates to methods of using the peptoids in detecting and isolating prions, and in the treatment and prevention of prion-related diseases.
Owner:NOVARTIS AG
Protein appropriate for orientation-controlled immobilization and immobilization carrier on which the proteins are immobilized
InactiveUS20090299035A1Prepared efficiently and rapidlyFunction increasePeptide/protein ingredientsDepsipeptidesAcidic amino acidsIsoelectric point
An object of the present invention is to provide a novel protein having the following amino acid sequence altered for specifically and efficiently binding a protein to an immobilization carrier via the carboxy terminus. The protein is used for immobilizing a portion represented by R1-R2 on an immobilization carrier, comprising the amino acid sequence represented by the general formula R1-R2-R3-R4-R5 [wherein:the sequences are oriented from the amino terminal side to the carboxy terminal side;the sequence of the R1 portion is the sequence of a subject protein to be immobilized and contains neither a lysine residue nor a cysteine residue;the sequence of the R2 portion may be absent, but when the sequence of the R2 portion is present, the sequence of the R2 portion is a spacer sequence composed of amino acid residues other than lysine and cysteine residues;the sequence of the R3 portion is composed of two residues of amino acid represented by cysteine-X (where X denotes an amino acid residue other than lysine or cysteine);the sequence of the R4 portion may be absent, but when the sequence of the R4 portion is present, the sequence of the R4 portion contains neither a lysine residue nor a cysteine residue, but contains an acidic amino acid residue capable of acidifying the isoelectric point of the entire protein comprising the amino acid sequence represented by the general formula R1-R2-R3-R4-R5; andthe sequence of an R5 portion is an affinity tag sequence for protein purification.
Owner:NAT INST OF ADVANCED IND SCI & TECH
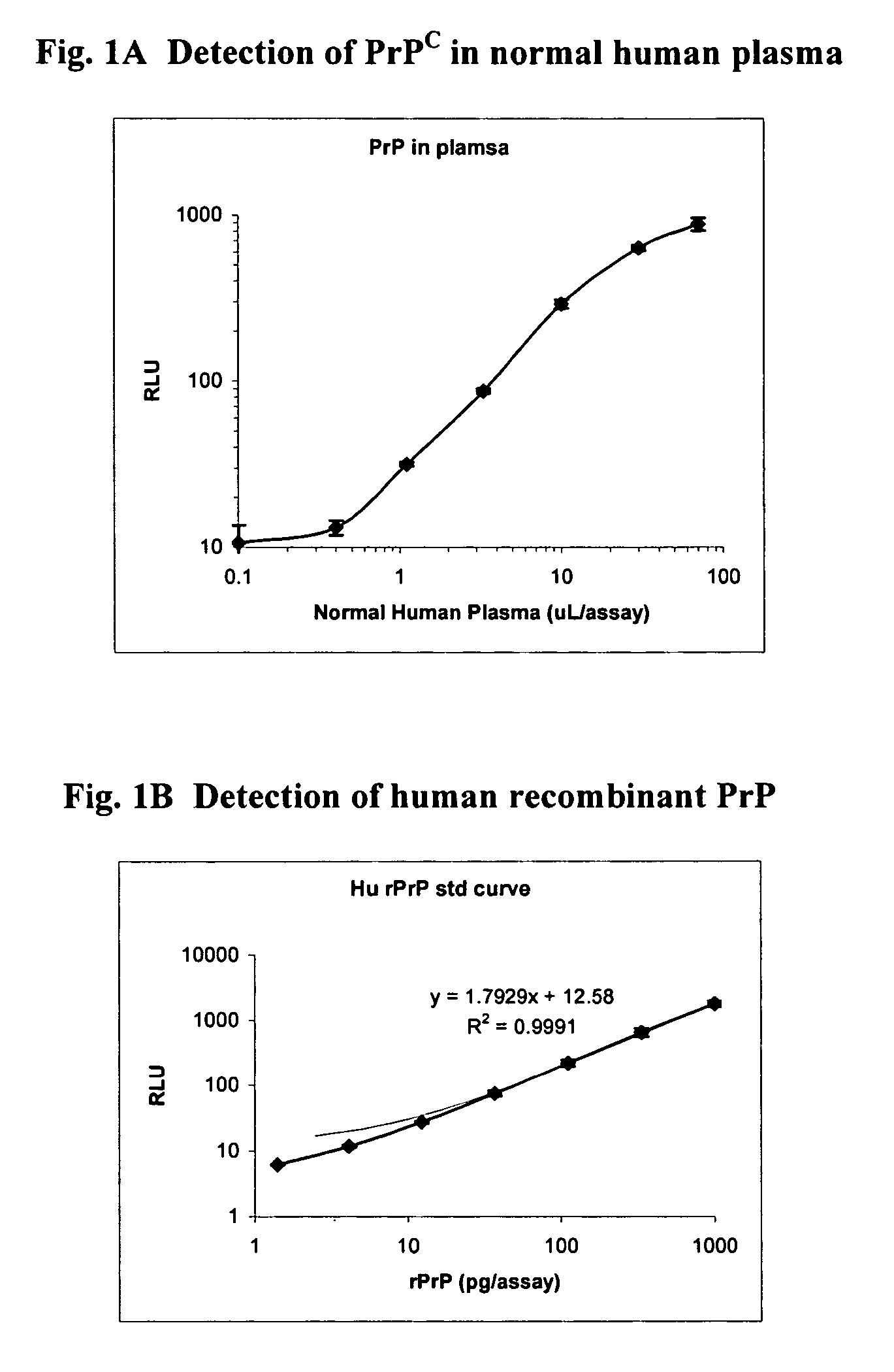
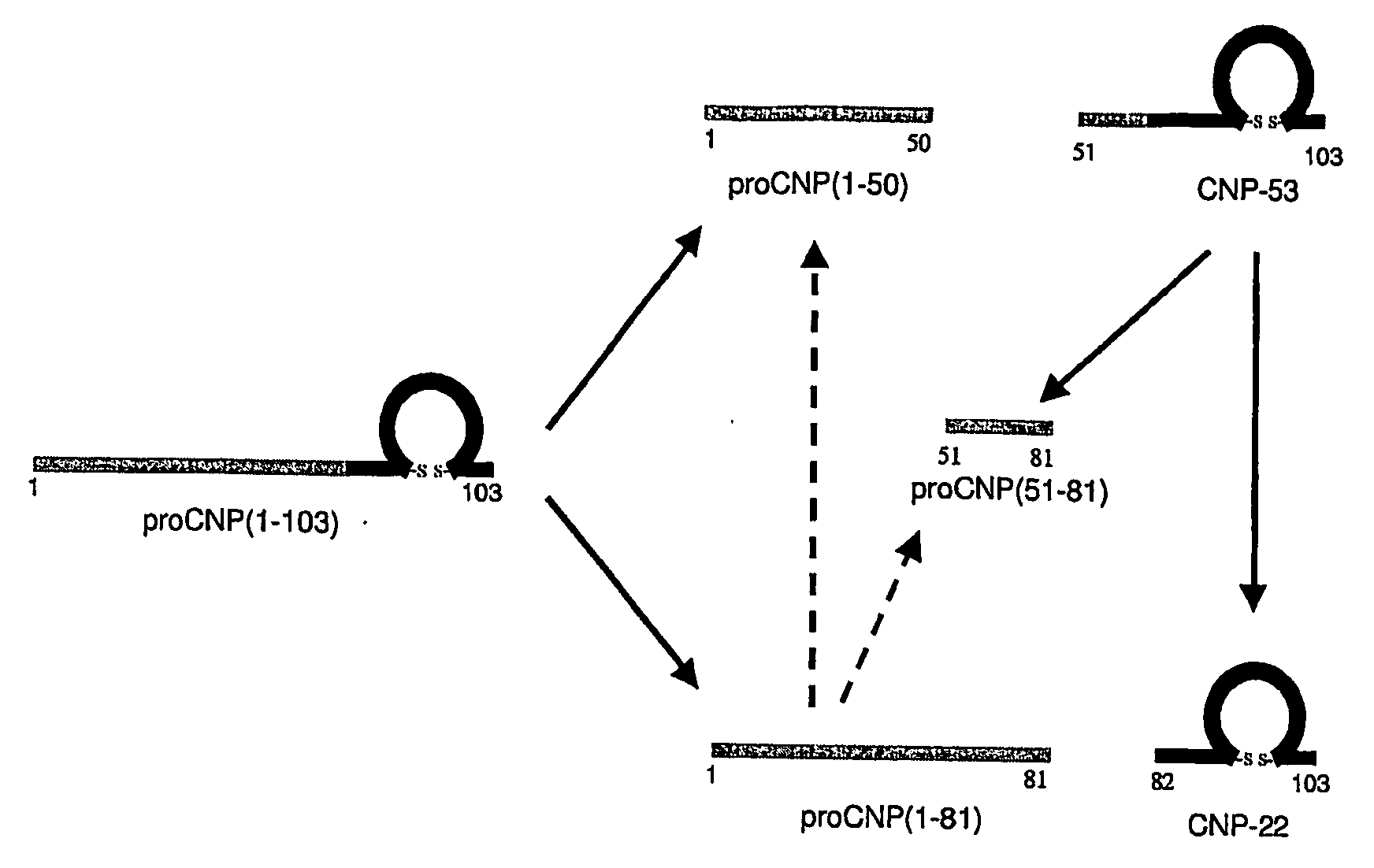
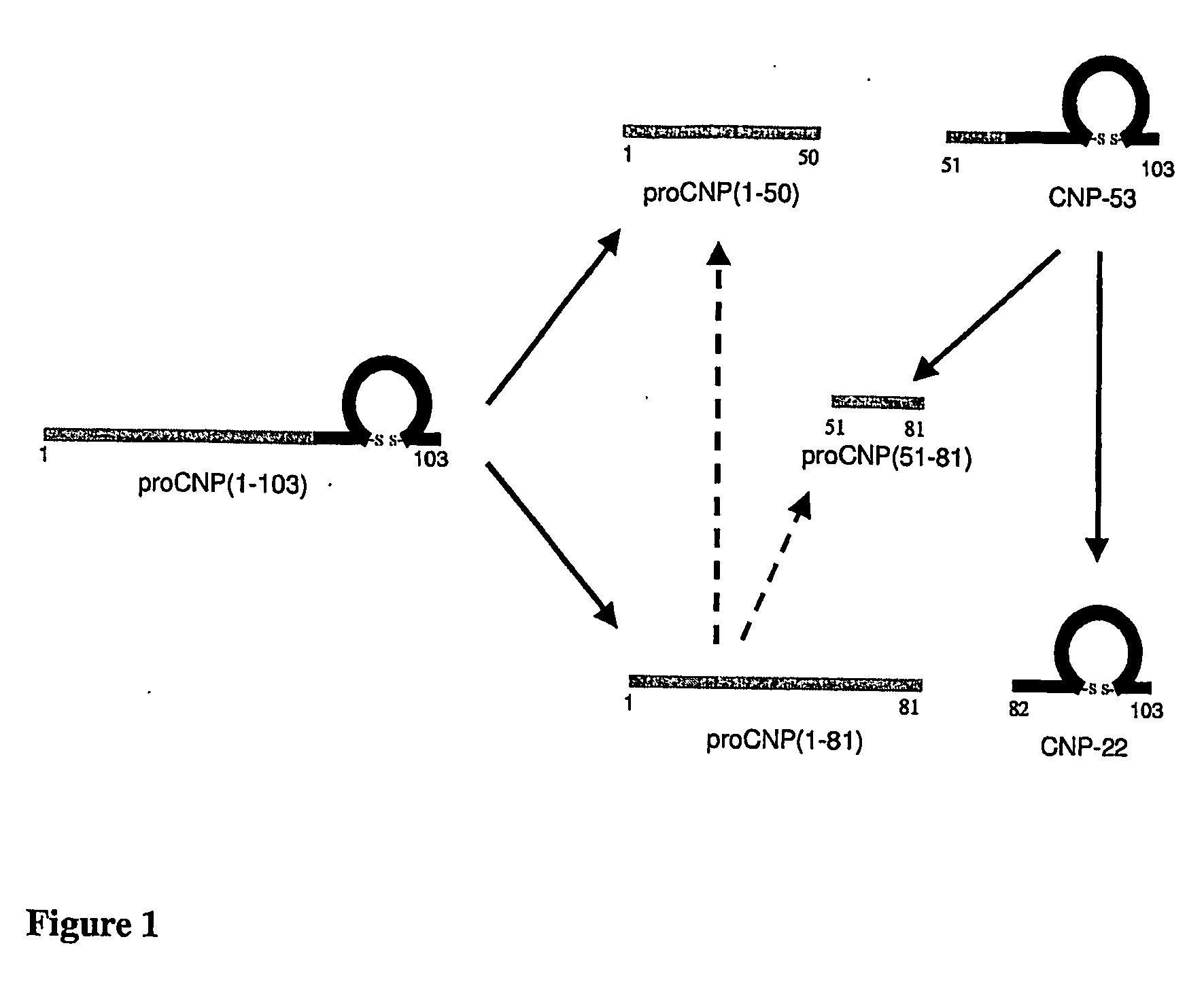
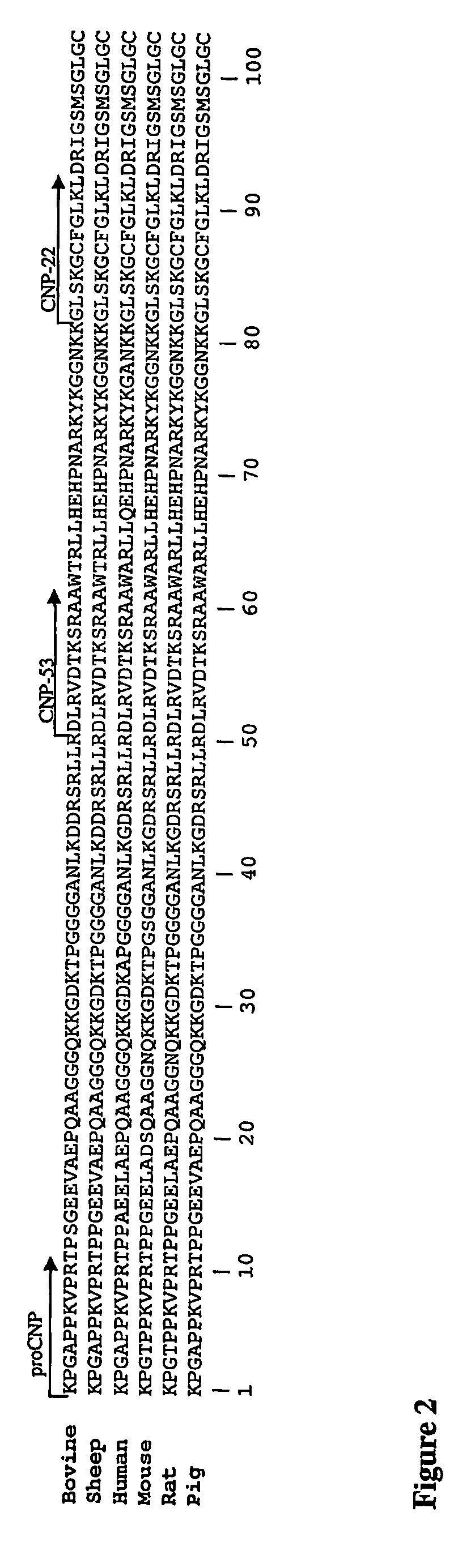
Assessment of Skeletal Growth Using Measurements of Nt-Cnp Peptides
InactiveUS20070292966A1Minimize adverse effectsIncreasing and decreasing skeletal growthSkeletal disorderDisease diagnosisDiseaseC-type natriuretic peptide
A means of measuring and monitoring skeletal growth rate using a plasma analyte, amino-terminal C-type natriuretic peptide (NT-CNP). Plasma NT-CNP concentrations reflect the potential for further growth of the immature skeleton. Measurement of plasma NT-CNP in a subject, when related to the mean of an appropriate age and gender matched set of control subjects, provides an index of the severity of a growth disorder not otherwise available and facilitates diagnosis in children with undetected osteo-chondrodysplasias or other intrinsic disorders of the growth plate. The invention also provides a means of detecting and monitoring potentially harmful effects of drugs and other factors on skeletal growth long before they are evident using current methods in clinical practice.
Owner:OTAGO INNOVATION
Intracellular delivery of small molecules, proteins, and nucleic acids
InactiveUS7166692B2Enhance cell viabilityVirus peptidesSaccharide peptide ingredientsProtein transduction domainADAMTS Proteins
An amino acid sequence Arg-Lys-Met-Leu-Lys-Ser-Thr-Arg-Arg-Gln-Arg-Arg (SEQ ID NO:1) functions as a protein transduction domain (PTD) and is capable of delivering small molecules, proteins, and nucleic acids to an intracellular compartment of a cell. An amino terminal lysine linker improves the efficiency of the PTD. A nuclear localization signal can be used to target the PTD to a cell's nucleus. The PTD can be used in PTD-cargo moiety complexes that can reversibly immortalize cells and increase cell viability in culture.
Owner:LONZA WALKERSVILLE INC
HER2 antibody composition
A composition comprising a main species HER2 antibody that binds to domain II of HER2, and an amino acid sequence variant thereof comprising an amino-terminal leader extension is disclosed. Pharmaceutical formulations comprising the composition, and therapeutic uses for the composition are also disclosed.
Owner:F HOFFMANN LA ROCHE & CO AG
POLYMERIC IMMUNOGLOBULIN FUSION PROTEINS THAT TARGET LOW AFFINITY FCyRECEPTORS
InactiveUS20090117133A1Small size range and conformationPromote generationSenses disorderPeptide/protein ingredientsLow affinityNACHT domain
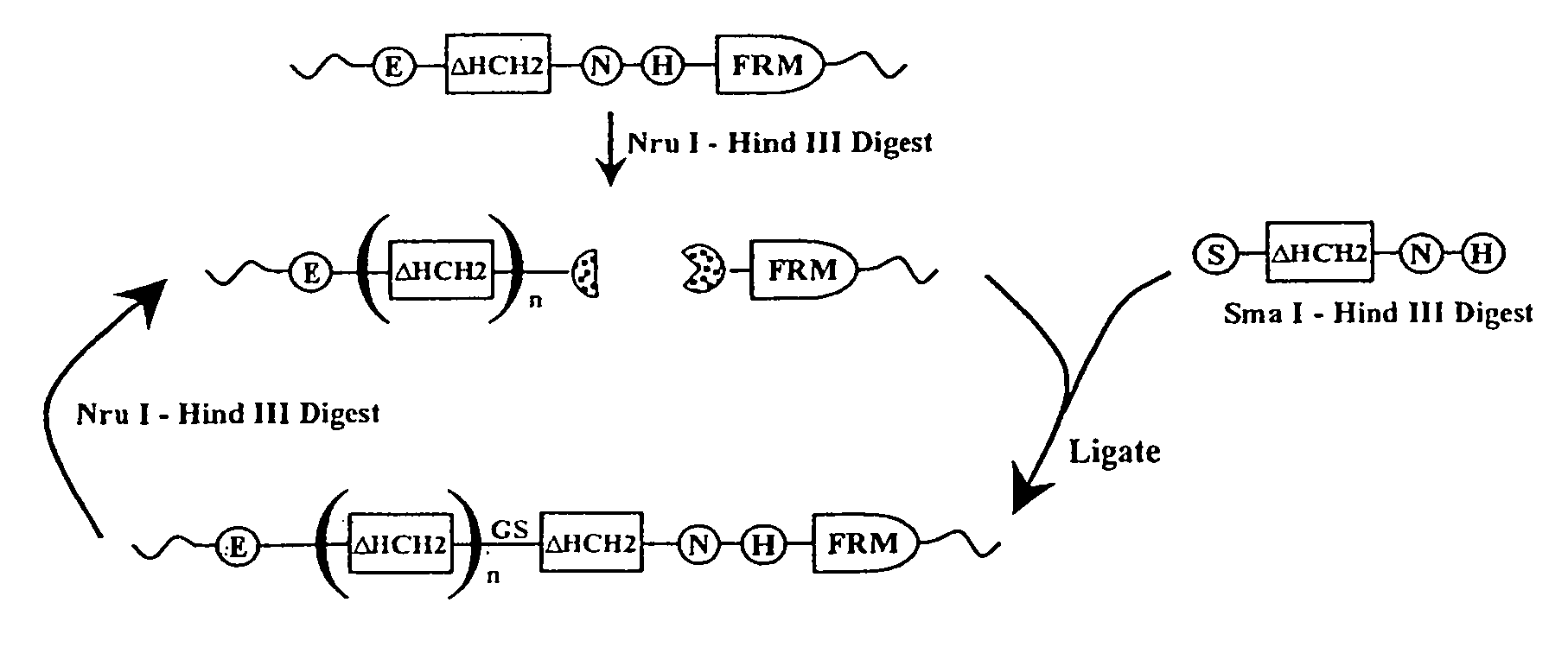
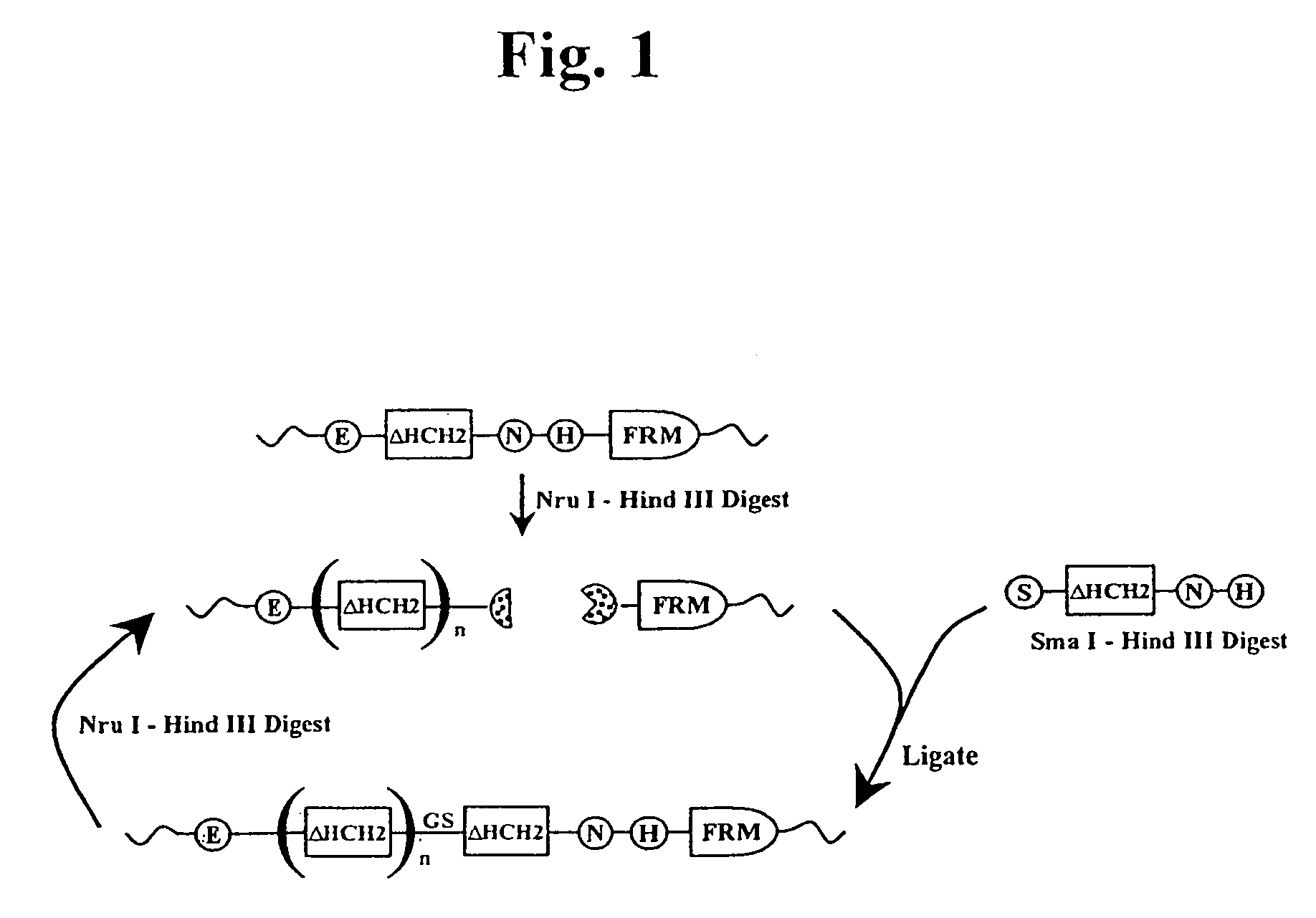
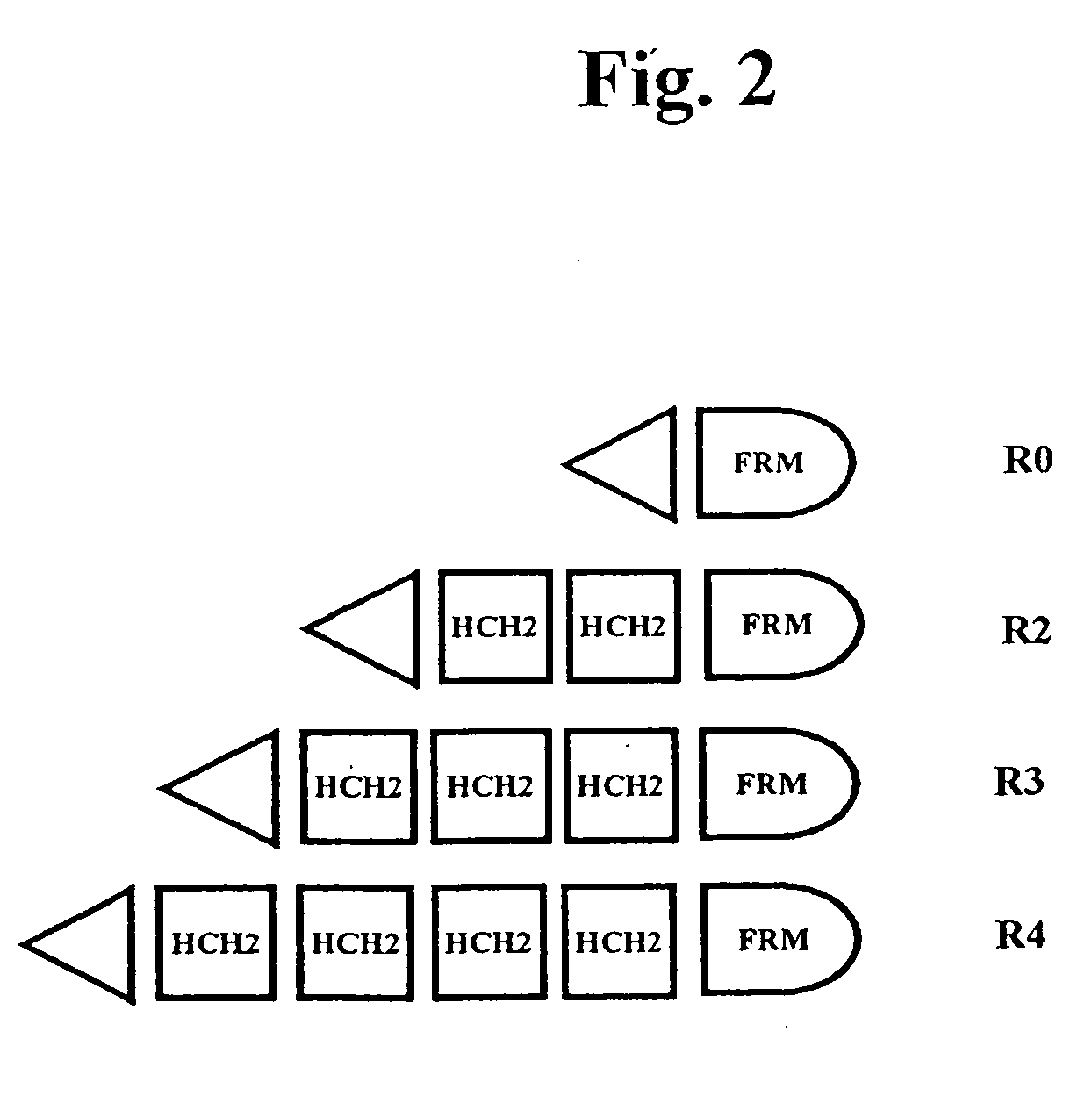
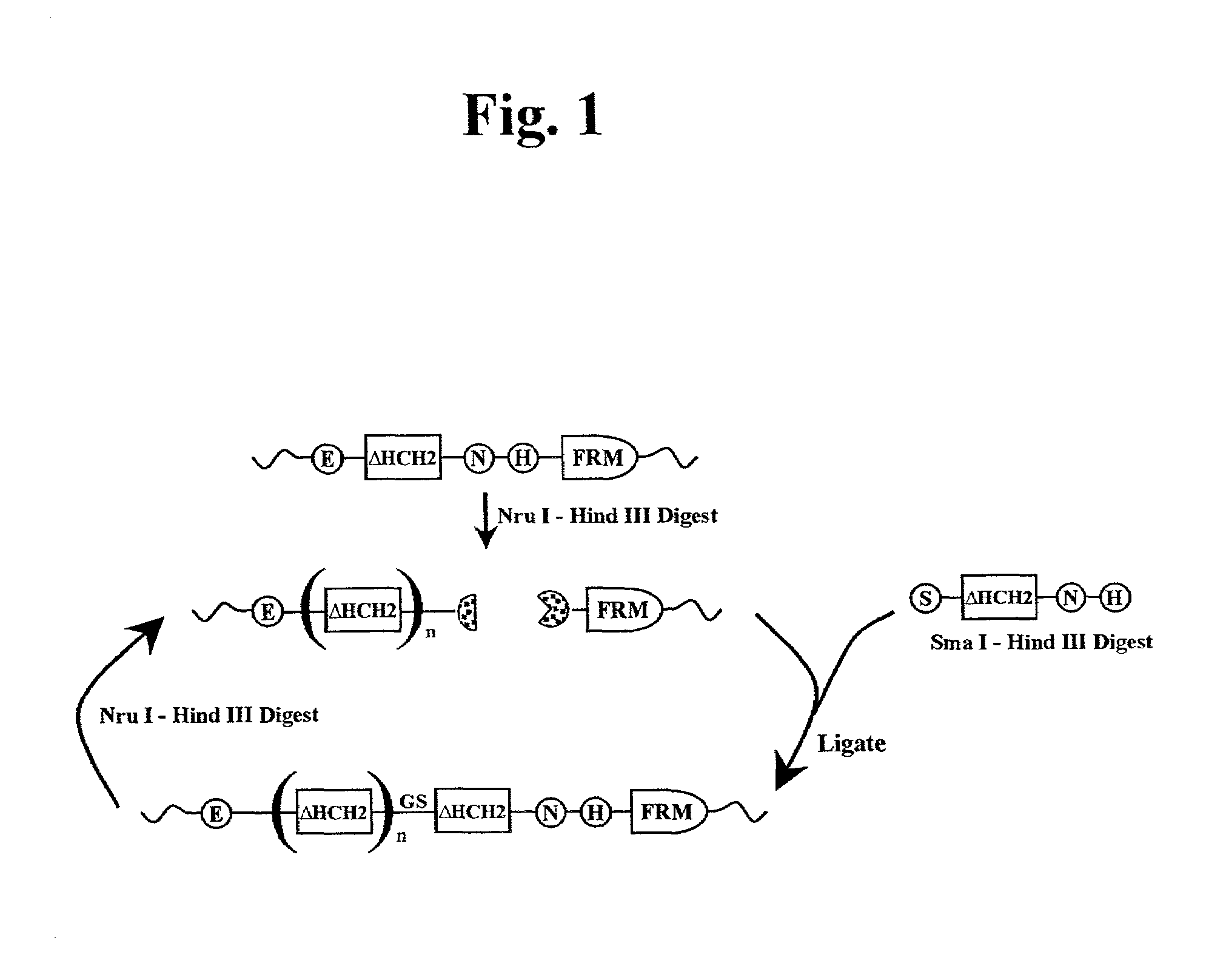
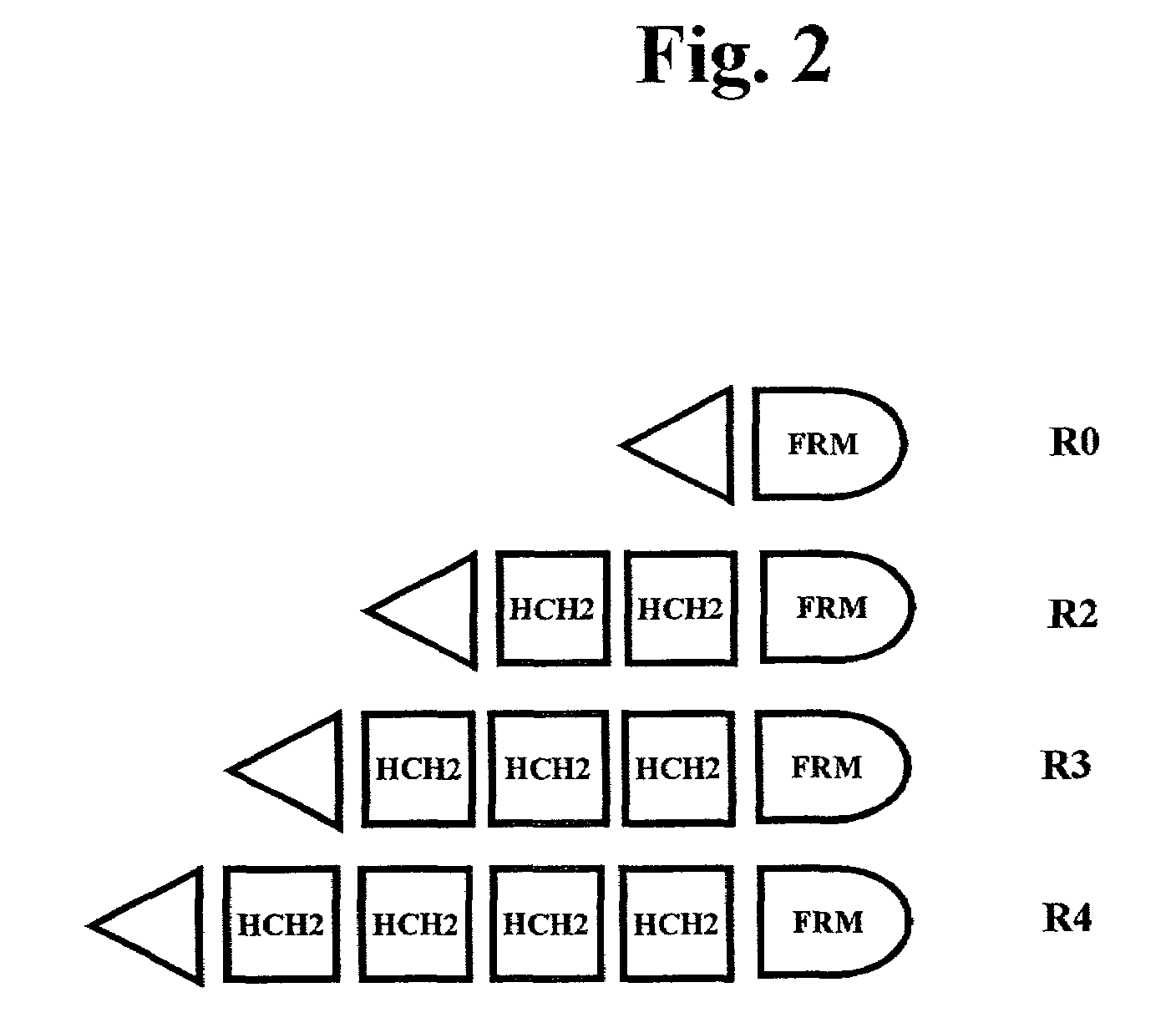
The present invention concerns a family of nucleic acids, polypeptides and cloning vectors which direct expression of fusion proteins that can mimic aggregated IgG (AIG) and immune complex function with respect to their interactions with FcγR and which allow for the inclusion and targeting of a second protein domain to cells expressing FcγR. This was accomplished by expressing multiple linear copies of the hinge and CH2 domains (HCH2) of human IgG1 fused to the framework region of human IgG1. Convenient restriction sites allow for the facile introduction of additional amino-terminal domains. Methods for treating patients using fission proteins are also disclosed. The HCH2 polymers described here represent a new strategy in the design of recombinant proteins for the therapeutic targeting of FcγR in autoimmune disorders.
Owner:ITERATIVE THERAPEUTICS
Polymeric immunoglobulin fusion proteins that target low-affinity Fcγreceptors
ActiveUS7511121B2Improve effectivenessModerating disease severitySenses disorderPeptide/protein ingredientsFusion Protein ExpressionLow affinity
The present invention concerns a family of nucleic acids, polypeptides and cloning vectors which direct expression of fusion proteins that can mimic aggregated IgG (AIG) and immune complex function with respect to their interactions with FcγR and which allow for the inclusion and targeting of a second protein domain to cells expressing FcγR. This was accomplished by expressing multiple linear copies of the hinge and CH2 domains (HCH2) of human IgG1 fused to the framework region of human IgG1. Convenient restriction sites allow for the facile introduction of additional amino-terminal domains. Methods for treating patients using fusion proteins are also disclosed. The HCH2 polymers described here represent a new strategy in the design of recombinant proteins for the therapeutic targeting of FcγR in autoimmune disorders.
Owner:ITERATIVE THERAPEUTICS
Long-acting growth hormone and methods of producing same
ActiveUS8450269B2Decreasing body fatReduce weight lossPeptide/protein ingredientsMetabolism disorderGrowth hormoneDosing Frequency
Use of a growth hormone protein and polynucleotides encoding same comprising an amino-terminal carboxy-terminal peptide (CTP) of chorionic gonadotrophin and two carboxy-terminal chorionic gonadotrophin CTPs attached to the growth hormone in methods of inducing growth or weight gain, method of increasing insulin-like growth factor (IGF-1) levels, and methods of reducing the dosing frequency of a growth hormone in a human subject are disclosed. Pharmaceutical compositions comprising the growth hormone and polynucleotides encoding the growth hormone of the invention and methods of using same are also disclosed.
Owner:OPKO BIOLOGICS
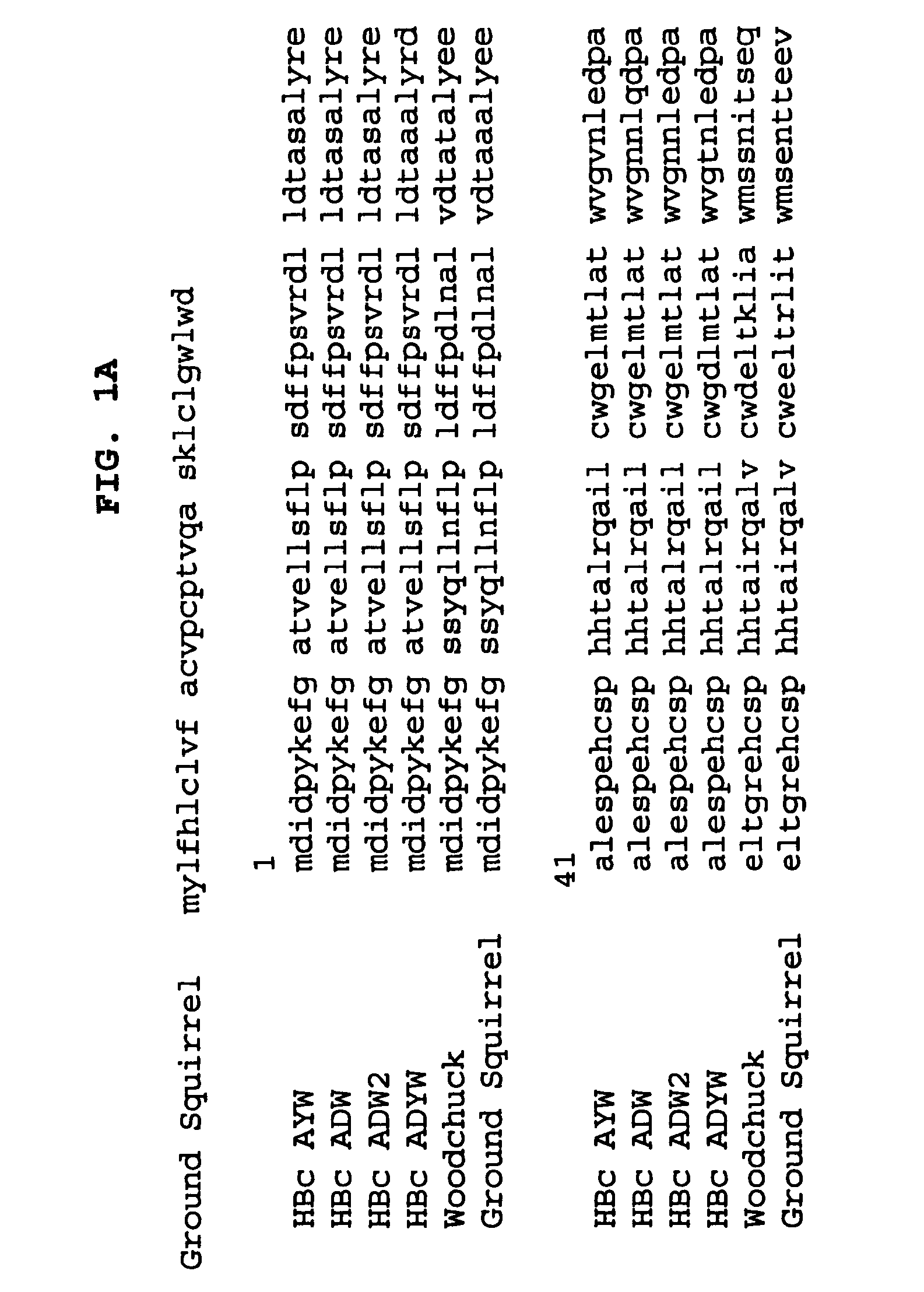
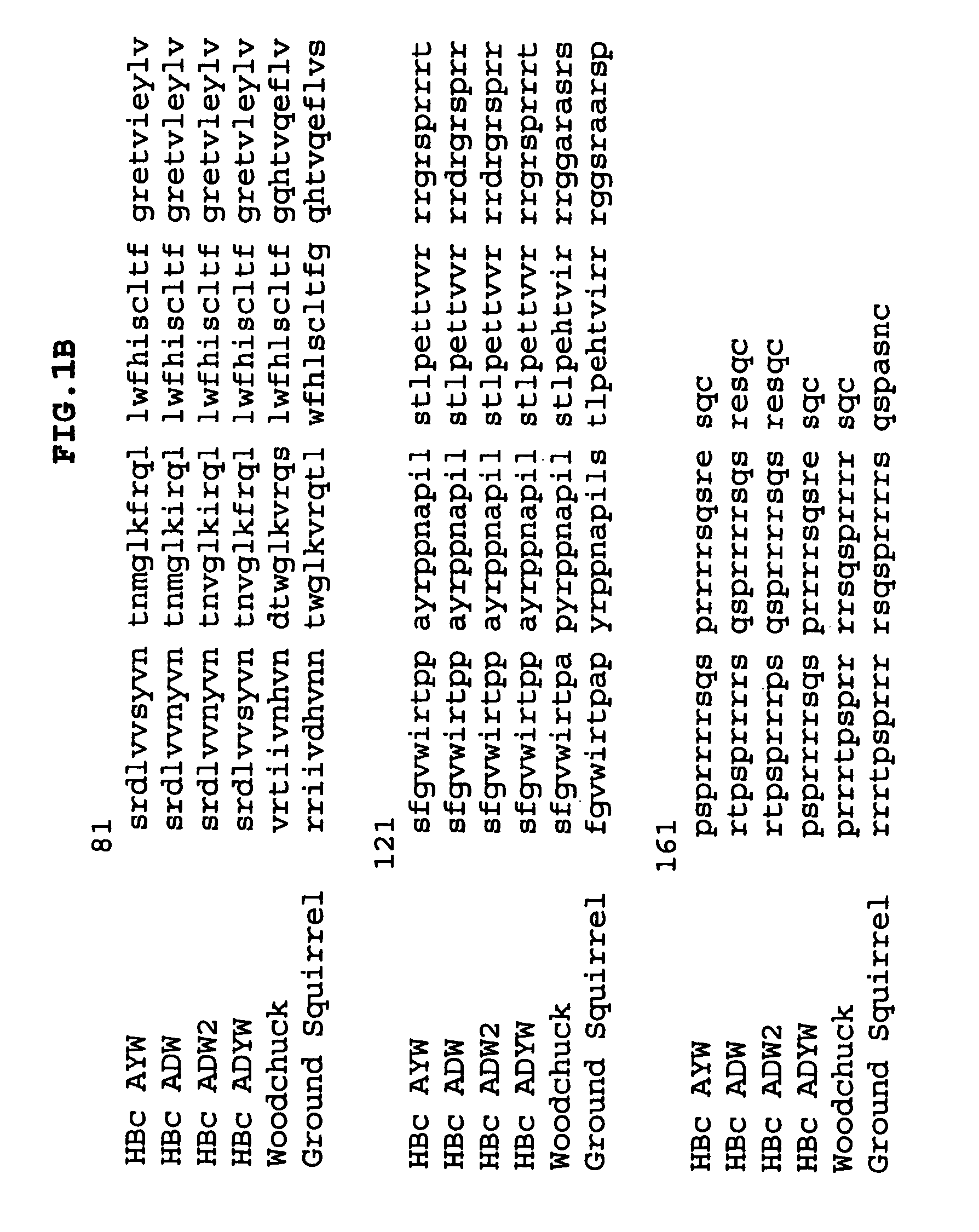
Stabilized HBc chimer particles as immunogens for chronic hepatitis
InactiveUS7351413B2Easy to prepareImprove stabilityAntibody mimetics/scaffoldsAntipyreticProtein moleculesChronic hepatitis
A method of treating chronic hepatitis B is disclosed that comprises administering a T cell-stimulating amount of a vaccine to a patient. The vaccine comprises an immunogenic amount of chimeric, carboxy-terminal truncated hepatitis B virus nucleocapsid (core) protein (HBc) that is engineered for both enhanced stability of self-assembled particles and the substantial absence of nucleic acid binding by those particles. The chimeric protein molecule can include one or more immunogenic epitopes peptide-bonded to one or more of the N-terminus, the immunogenic loop or the C-terminus of HBc. The enhanced stability of self-assembled particles is obtained by the presence of at least one heterologous cysteine residue near one or both of the amino-terminus and carboxy-terminus of the chimer molecule.
Owner:LORANTIS