Preparation method of organic acid salt catalytic polyarylene sulfide
An organic acid salt catalyzed polyarylene sulfide and polyarylene sulfide technology, which is applied in the field of polyarylene sulfide preparation, can solve the problems of difficult catalyst recovery, low solvent recovery rate, and many reaction by-products, and achieve easy processing, Effect of high glass transition temperature and low product cost
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
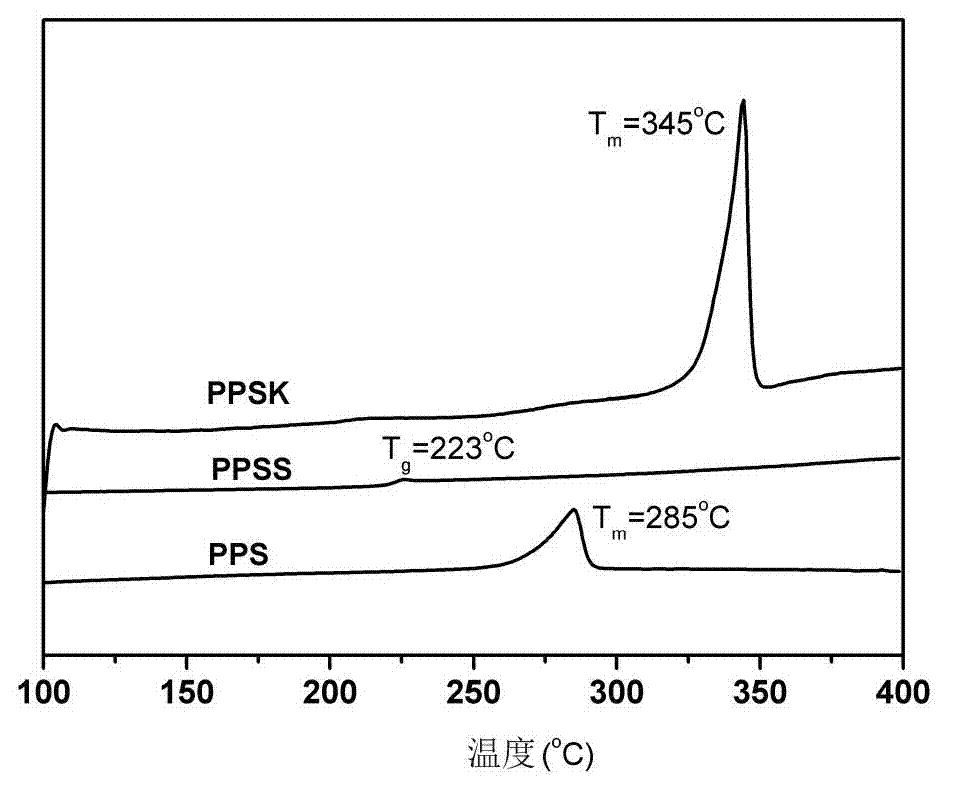
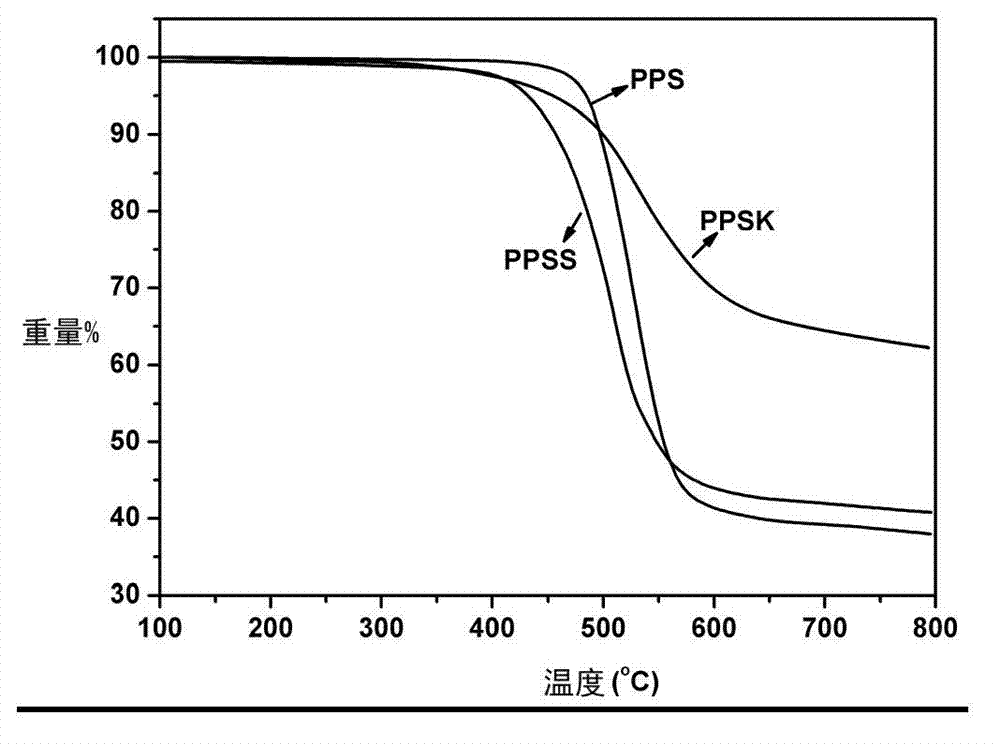
[0031] 30g calcium adipate, 10g sodium carbonate, 500g N-methylpyrrolidone and 240g Na 2 S·9H 2 O was added to the autoclave, and under the protection of nitrogen, the dehydration reaction was carried out at a temperature of 200°C for 2 hours to obtain the dehydrated Na 2 S solution, to be the above Na 2 When the temperature of the S solution drops to 190°C, add 147g of p-dichlorobenzene, react at a temperature of 200°C for 6h, then keep at a temperature of 230°C for 3h, cool down to 140°C, filter the reaction solution, and filter the polyphenylene sulfide ( PPS) was poured into water, the crude product was separated, the crude product was washed, dried, purified with deionized water and acetone, and dried at a temperature of 120°C and a vacuum of 0.095 MPa for 1 hour to obtain polyphenylene sulfide resin (PPS). Its yield is 93%, intrinsic viscosity number [η]=0.32, solvent recovery rate is 93%, and melt index is 56g / 10min. For infrared characterization see figure 1 Shown,...
Embodiment 2
[0033] 50g of zinc p-ethylbenzenesulfonate, 20g of potassium formate, 400g of N,N-dimethylpropenyl urea and 166g of Na 2 S·5H 2 O was added to the autoclave, and under the protection of nitrogen, the dehydration reaction was carried out at a temperature of 190°C for 1 hour to obtain the dehydrated Na 2 S solution, to be the above Na 2 When the temperature of the S solution drops to 180°C, add 218g of 4,4'-difluorobenzophenone, react at a temperature of 240°C for 3h, then keep at a temperature of 280°C for 0.5h, cool down to 120°C, filter the reaction solution, and Pour the finished polyphenylene sulfide ketone (PPSK) into water, separate the crude product, wash the crude product, dry it, purify it with deionized water and acetone, and dry it at a temperature of 110°C and a vacuum of 0.09 MPa for 5 hours to obtain polyphenylene sulfide Etherketone resin (PPSK). The yield was 92%, the intrinsic viscosity [η]=0.52, and the solvent recovery was 95%. For infrared characterizati...
Embodiment 3
[0035] 5 g tin p-sulfosalicylate, 0.1 g potassium hydroxide, 400 g 2,4-dimethylsulfolane and 130 g Na 2 S·3H 2 O was added to the autoclave, and under the protection of nitrogen, the dehydration reaction was carried out at a temperature of 190°C for 1.5h to obtain the dehydrated Na 2 S solution, to be the above Na 2 When the temperature of the S solution drops to 180°C, add 287g of 4,4'-dichlorodiphenyl sulfone, react at a temperature of 220°C for 3 hours, then keep at a temperature of 230°C for 1 hour, cool down to 110°C, and filter the reaction solution. Pour polyphenylene sulfide sulfone (PPSS) into water, separate the crude product, wash the crude product, dry it, purify it with deionized water and acetone, and dry it for 8 hours at a temperature of 110°C and a vacuum of 0.085 MPa to obtain polyphenylene sulfide sulfone Resin (PPSS). Its yield is 92%, intrinsic viscosity number [η]=0.61, and solvent recovery rate is 90%. For infrared characterization see figure 1 Show...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Melt index | aaaaa | aaaaa |
| Melt index | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More