Diethylamine derivative of Cleistanine and preparation method and application thereof
A technology for cleistanone and derivatives, which is applied in the field of preparation of cleistanone derivatives, can solve the problems of non-standard treatment and management of tuberculosis patients, difficulty in tuberculosis prevention and control, and the like, and achieve good antibacterial effect.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
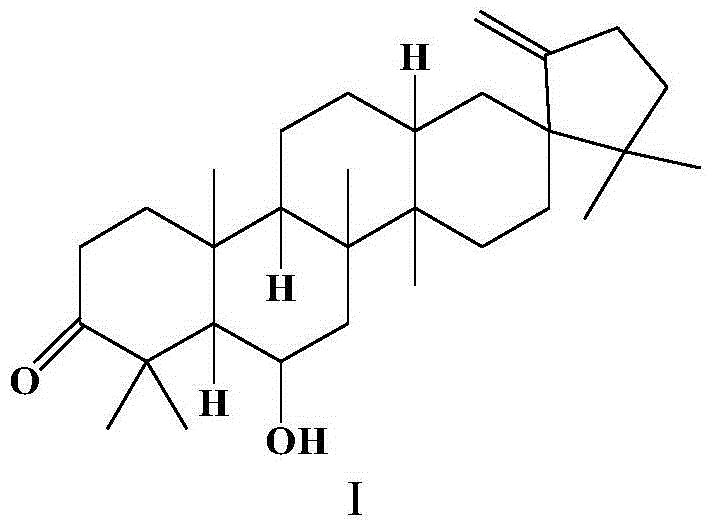
[0025] Preparation of Example 1 Compound Cleistanone
[0026] The preparation method of the compound Cleistanone (I) refers to the literature published by Van Trinh Thi Thanh et al. –4111, August 2011).
[0027]
Embodiment 2
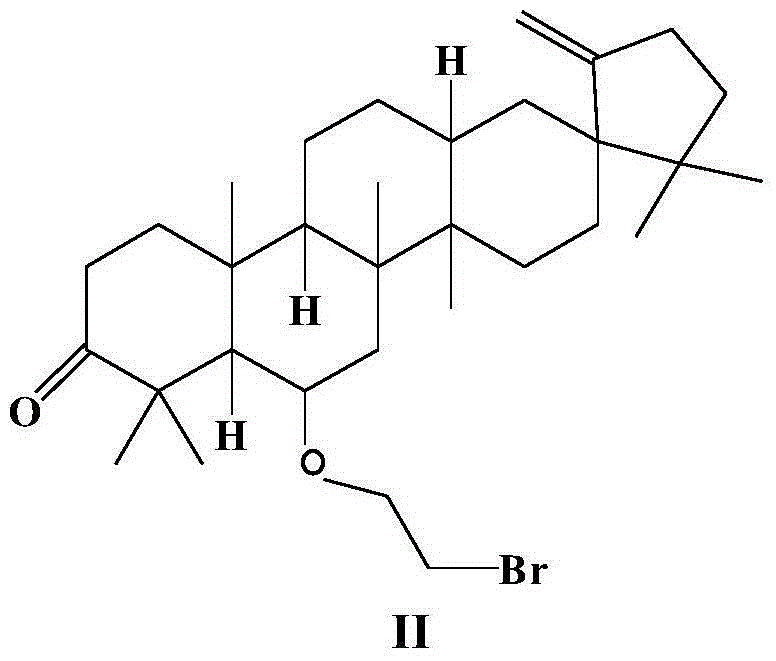
[0028] Synthesis of O-bromoethyl derivatives (II) of cleistanone Cleistanone of embodiment 2
[0029] Compound I (440 mg, 1.00 mmol) was dissolved in 10 mL of benzene, and tetrabutylammonium bromide (TBAB) (0.04 g), 1,2-dibromoethane (3.760 g, 20.00 mmol) and 6 mL of 50% sodium hydroxide solution. The mixture was stirred at 25 °C for 24 h. After 24 hours, the reaction solution was poured into ice water, extracted twice with dichloromethane immediately, and the organic phase solutions were combined. Then the organic phase solution was washed with water and saturated brine three times successively, then dried with anhydrous sodium sulfate, and finally concentrated under reduced pressure to remove the solvent to obtain a crude product. The crude product was purified by silica gel column chromatography (mobile phase: petroleum ether / acetone=100:1, v / v), and the yellow concentrated elution band was collected to obtain compound II as a yellow solid (344 mg, 63%).
[0030] 1H NMR ...
Embodiment 3
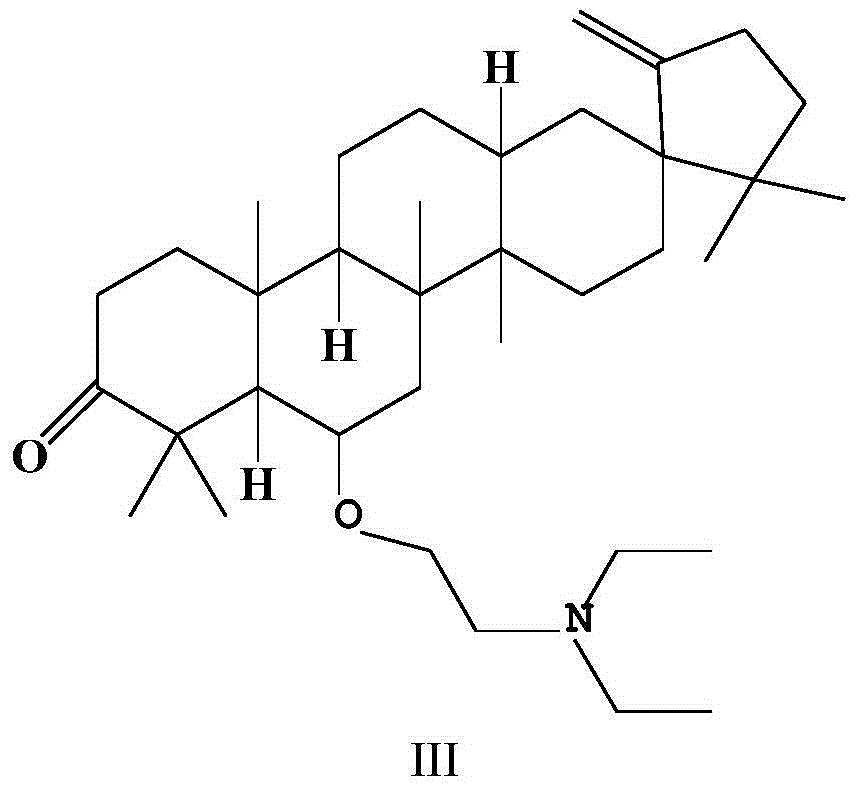
[0034] Example 3 Synthesis of O-(diethylamino)ethyl derivatives (III) of Cleistanone
[0035] Compound II (273mg, 0.5mmol) was dissolved in 20mL of acetonitrile, anhydrous potassium carbonate (345mg, 2.5mmol), potassium iodide (84mg, 0.5mmol) and diethylamine (1460mg, 20mmol) were added thereto, and the mixture was heated to reflux for 8h . After the reaction was completed, the reaction solution was poured into ice water, extracted three times with an equal amount of dichloromethane, and the organic phases were combined. The combined organic phases were successively washed with water and saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to remove the solvent to obtain a crude product. The crude product was purified by silica gel column chromatography (mobile phase: petroleum ether / acetone=100:1, v / v), and the light yellow concentrated elution band was collected to obtain compound III as a light yellow colloidal solid (169.8mg, 63% ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com