Medicinal atorvastatin calcium composition capsules for treating coronary heart disease
A technology of atorvastatin calcium and composition, applied in the field of medicine, can solve the problems of high water content of sulfobutyl-β-cyclodextrin, degradation of atorvastatin calcium, low bioavailability and the like, and achieve water and impurities. Low content, reduced myalgia side effects, improved solubility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0034] Example 1: Preparation of Atorvastatin Calcium Crystals
[0035] Dissolving atorvastatin calcium in a mixed solvent of ethanol and N-methylpyrrolidone whose volume is 10 times the weight of atorvastatin calcium at 35°C, the volume ratio of ethanol and N-methylpyrrolidone is 3:1, After the dissolution is complete, add a chloroform solvent whose volume is 8 times the weight of atorvastatin calcium, lower the temperature to room temperature within 2 hours, keep it at room temperature for 3 hours, and then further lower the temperature to -5°C, and then - After standing at 5°C for 3 hours, crystals were precipitated to obtain atorvastatin calcium crystals.
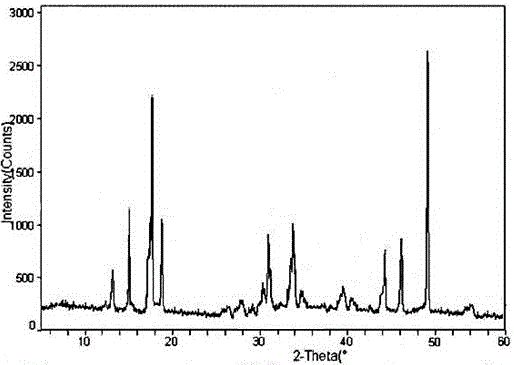
[0036] The prepared atorvastatin calcium crystals are obtained by measuring the X-ray powder diffraction pattern using Cu-Kα rays as shown in figure 1 shown.
Embodiment 2
[0037] Example 2: Preparation of Atorvastatin Calcium Capsules
[0038] Prescription: 1 part by weight of atorvastatin calcium crystal compound obtained in Example 1, 15 parts by weight of starch, 0.4 part by weight of hypromellose, 0.25 part by weight of dioctyl sodium sulfosuccinate, 95% ethanol 3.1 parts by weight, 0.7 parts by weight of talcum powder;
[0039] Preparation:
[0040] (1) Processing of raw and auxiliary materials: sieve atorvastatin calcium to 100 mesh;
[0041] (2) Weighing: Weighing according to the process prescription;
[0042] (3) Preparation of adhesive: Dissolve the prescribed amount of hypromellose and dioctyl sodium sulfosuccinate in 95% ethanol and set aside;
[0043] (4) Mixing and granulation: Add atorvastatin calcium and starch to the wet mixing granulator, turn on the stirring motor and dry mix for 10 minutes; add the prepared binder, wet mix and cut, and use a 16-mesh sieve to soften material;
[0044] (5) Drying: Add the wet granules obta...
Embodiment 3
[0048] Example 3: Preparation of Atorvastatin Calcium Capsules
[0049] Prescription: 1 part by weight of atorvastatin calcium crystal compound obtained in Example 1, 18 parts by weight of starch, 0.5 part by weight of hypromellose, 0.3 part by weight of dioctyl sodium sulfosuccinate, 95% ethanol 3.2 parts by weight, 0.8 parts by weight of talcum powder;
[0050] Preparation:
[0051] (1) Processing of raw and auxiliary materials: sieve atorvastatin calcium to 100 mesh;
[0052] (2) Weighing: Weighing according to the process prescription;
[0053] (3) Preparation of adhesive: Dissolve the prescribed amount of hypromellose and dioctyl sodium sulfosuccinate in 95% ethanol and set aside;
[0054] (4) Mixing and granulation: Add atorvastatin calcium and starch to the wet mixing granulator, turn on the stirring motor and dry mix for 10 minutes; add the prepared binder, wet mix and cut, and use a 16-mesh sieve to soften material;
[0055] (5) Drying: Add the wet granules obt...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More