Immunity test strip based on fluorescent microsphere for detecting infectious pleural pneumonia antibody of goat as well as preparation and detection method
A fluorescent microsphere, pleuropneumonia technology, applied in the field of immunological detection, can solve the problems of low sensitivity, poor repeatability or specificity, and need laboratory instruments, and achieves the effect of high sensitivity and shortened time.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
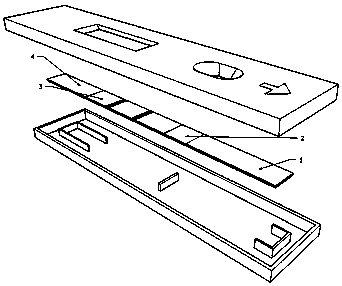
[0025] Embodiment 1, the preparation of test paper card
[0026] 1. Preparation of binding pads
[0027] (1) Cleaning: Take 50 μL of fluorescent microspheres into a 1.5 mL centrifuge tube, add 1 mL of 0.01M MES buffer, shake and mix, centrifuge at 15,000 r / min for 15 min, discard the supernatant, add 1 mL of 0.01M MES buffer, and ultrasonically Loose microspheres; repeat this step three times, the purpose of cleaning the microspheres has been achieved, and finally the microsphere liquid after cleaning is obtained;
[0028] (2) Activation: Add 250 μL of EDC solution to 1 mL of microsphere solution after washing, activate in the dark for 2 h, centrifuge at 15,000 r / min for 15 min, discard the supernatant, add 1 mL of 0.01M MES buffer, and disperse the microspheres by ultrasonication, 15,000 r / min Centrifuge for 15min, discard the supernatant;
[0029] (3) Labeling: Add 1 mL of 0.05M borate buffer, disperse by ultrasonication, add 20 μL of Mycoplasma goatum recombinant protein...
Embodiment 2
[0048] Embodiment 2, detection method and comparison test of goat infectious pleuropneumonia antibody in goat serum
[0049] 1. Tear open the aluminum foil packaging bag of the test card, take out the test card, and place it on a flat and clean table;
[0050] 2. Absorb freshly collected goat serum with a matching straw, and drop 2-3 drops (about 60 μl) vertically and slowly into the sample well;
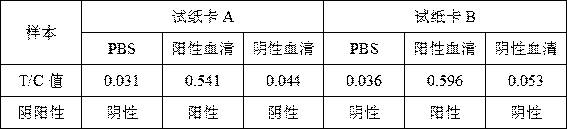
[0051] 3. Read the result in 5-10 minutes, insert the test paper card into the card slot of the fluorescence immunoassay analyzer in the direction of the arrow shown on the card cover, and read the T / C value;
[0052] 4. Judgment criteria: T / C value < 0.1 is negative; T / C value ≧ 0.1 is positive; C value is 0, invalid;
[0053] 5. Comparison with colloidal gold test strips: select 20 weakly positive samples detected by goat infectious pleuropneumonia hemagglutination diagnostic kit, test the results with colloidal gold test strips, and detect 5 positive and 15 negative; Fluorescen...
Embodiment 3
[0055] Example 3. Comparison test of the stability of colloidal gold-labeled Mycoplasma pneumoniae recombinant protein and fluorescent microsphere-labeled recombinant protein against Mycoplasma pneumoniae
[0056] 1. Place the prepared colloidal gold-antigen complex binding pad and the fluorescent microsphere-antigen complex binding pad in a 37°C blast drying oven;
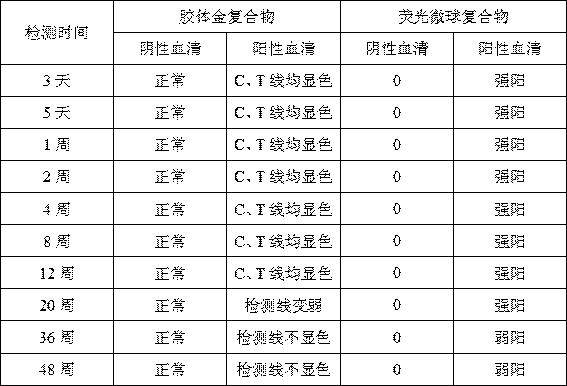
[0057] 2. On the 3rd day, 5th day, 1 week, 2 weeks, 4 weeks, 8 weeks, 12 weeks, 20 weeks, 36 weeks, and 48 weeks, test the negative serum and positive serum respectively, and observe their stability;
[0058] 3. The result is as follows
[0059]
[0060] As shown in the above table, the detection effect of the colloidal gold-labeled antigen complex decreased with the extension of the destruction test time, and the detection situation changed significantly at the 20th week; while the test strip using the fluorescent microsphere-antigen complex binding pad The positive sera showed a downward trend in the 36th we...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More