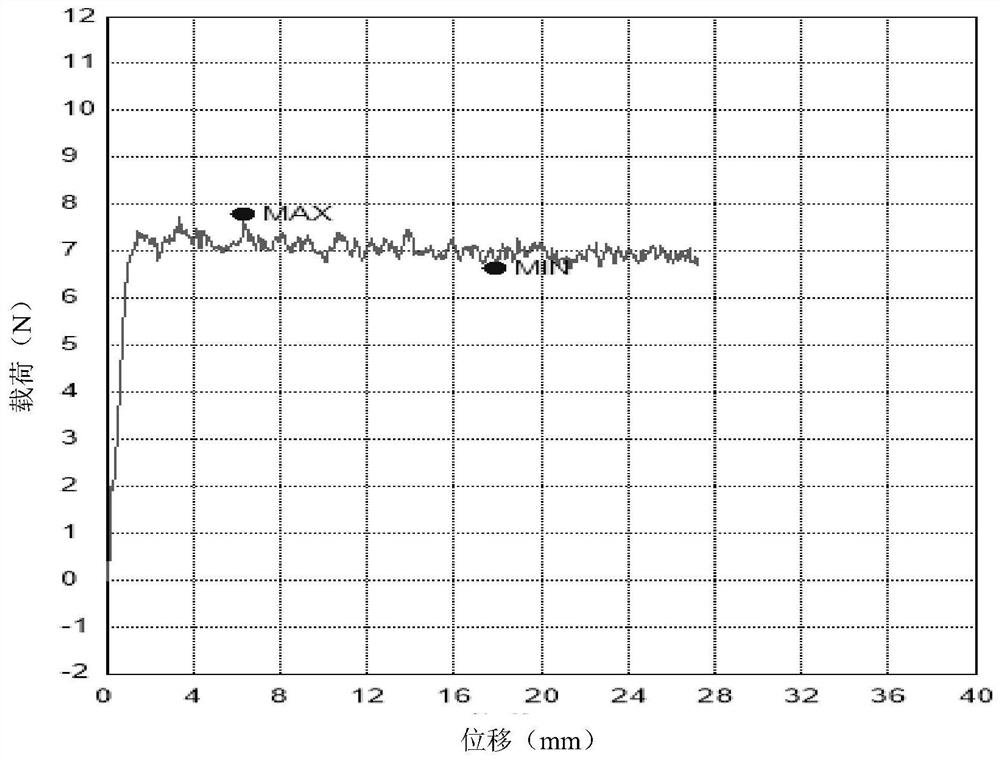
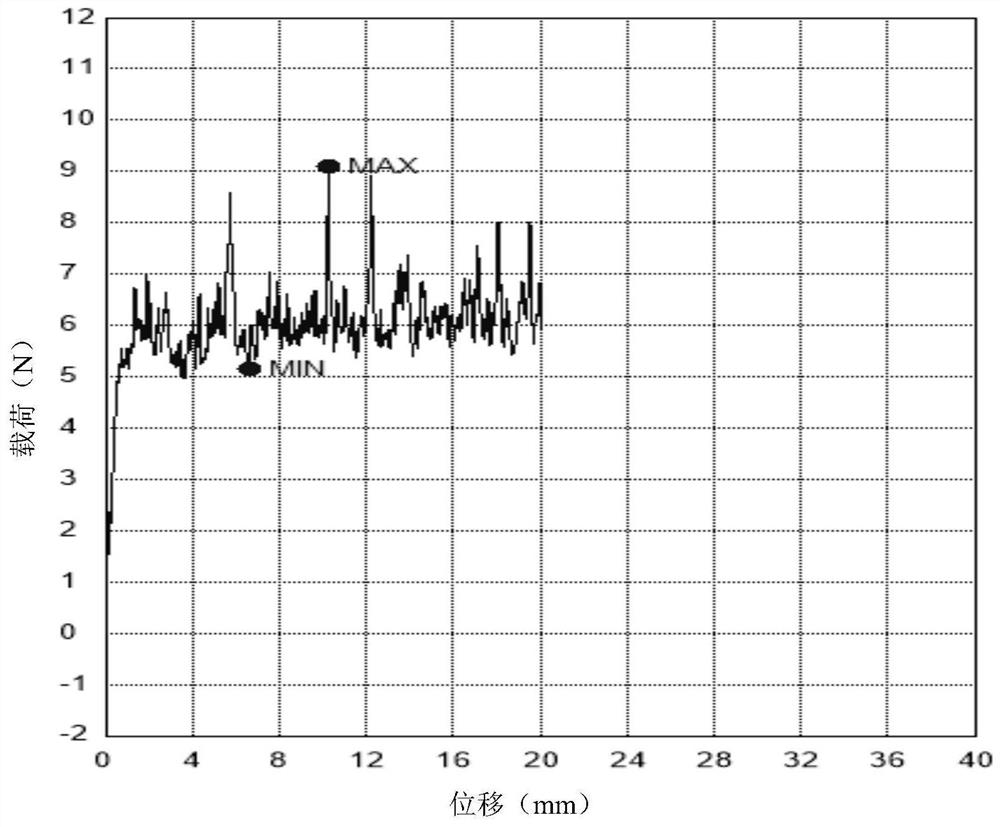
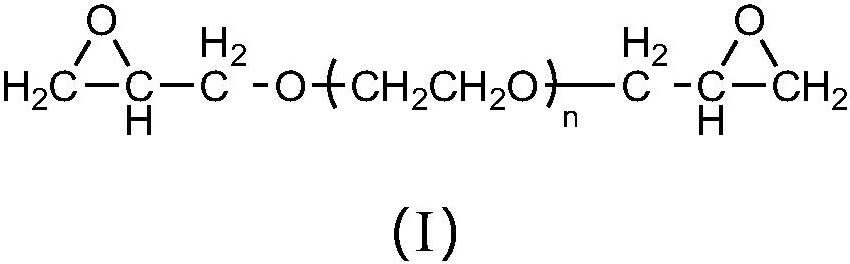Sodium hyaluronate gel cross-linked with polyglycol epoxy derivatives for injection and preparation method thereof
A sodium hyaluronate, alcohol epoxy technology, applied in drug delivery, tissue regeneration, pharmaceutical formulations, etc., can solve the problems of small pushing force, long retention time in the body, and good injectability.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0098] Embodiment 1: synthetic tetraethylene glycol diglycidyl ether (Ia)
[0099] Synthesize tetraethylene glycol diglycidyl ether of following structure:
[0100]
[0101] Add tetraethylene glycol (0.1mol), tetrahydrofuran (100mL) and potassium hydroxide (0.4mol) into the three-necked flask, stir in a water bath, then add epichlorohydrin (0.6mol) dropwise to the reaction system, and control the reaction temperature not to exceed React overnight at room temperature at 35°C. After the reaction, the reaction solution was filtered, and the filter residue was washed with dichloromethane, then the filtrate was collected, and the dichloromethane was removed by rotary evaporation to obtain a crude product. The crude product was molecularly distilled to obtain pure tetraethylene glycol diglycidyl ether.
[0102] 1 H-NMR (DMSO-d6): 2.52-2.55 (m, 2H), 2.70-2.73 (m, 2H), 3.07-3.11 (m, 2H), 3.22-3.28 (m, 2H), 3.52-3.56 (m , 16H), 3.68-3.73(m, 2H);
[0103] HPLC detection: the pro...
Embodiment 2
[0105] Embodiment 2: Synthesis of dodecaethylene glycol diglycidyl ether (Ib)
[0106] Synthesize dodecaethylene glycol diglycidyl ether of following structure:
[0107]
[0108] Add dodecaethylene glycol (0.1mol), tetrahydrofuran (100mL) and potassium hydroxide (0.4 mol) in the there-necked flask, stir in a water bath, then drop epichlorohydrin (0.6mol) in the reaction system, control the reaction temperature Over 35°C, react overnight at room temperature. After the reaction, the reaction solution was filtered, and the filter residue was washed with dichloromethane, then the filtrate was collected, and the dichloromethane was removed by rotary evaporation to obtain a crude product. The crude product was molecularly distilled to obtain pure dodecaethylene glycol diglycidyl ether.
[0109] 1 H-NMR (DMSO-d6): 2.51-2.55 (m, 2H), 2.70-2.73 (m, 2H), 3.07-3.11 (m, 2H), 3.22-3.29 (m, 2H), 3.51-3.57 (m , 48H), 3.68-3.73(m, 2H);
[0110] HPLC detection: the product purity is 99...
Embodiment 3
[0112] Embodiment 3: synthesizing Tetracethylene glycol diglycidyl ether (Ic)
[0113] Synthesize four tetraethylene glycol diglycidyl ethers of the following structure:
[0114]
[0115] Add tetracosanethylene glycol (0.1mol), tetrahydrofuran (100mL) and potassium hydroxide (0.4mol) into the three-necked flask, stir in a water bath, then add epichlorohydrin (0.6mol) dropwise to the reaction system, and control the reaction temperature Do not exceed 35°C, and react overnight at room temperature. After the reaction, the reaction solution was filtered, and the filter residue was washed with dichloromethane, then the filtrate was collected, and the dichloromethane was removed by rotary evaporation to obtain a crude product. The crude product was separated by a column to obtain pure tetracosanthyl glycol diglycidyl ether.
[0116] 1 H-NMR (DMSO-d6): 2.51-2.55 (m, 2H), 2.70-2.73 (m, 2H), 3.07-3.11 (m, 2H), 3.22-3.28 (m, 2H), 3.51-3.56 (m , 96H), 3.68-3.73(m, 2H);
[0117] H...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More